Chemistry:Gentisic acid
| |
| |
| Names | |
|---|---|
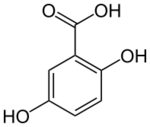
| Preferred IUPAC name
2,5-Dihydroxybenzoic acid | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties[3] | |
| C7H6O4 | |
| Molar mass | 154.121 g·mol−1 |
| Appearance | white to yellow powder |
| Melting point | 204 °C (399 °F; 477 K) |
| very soluble | |
| Solubility in ethanol | very soluble |
| Solubility in diethyl ether | very soluble |
| Solubility in acetone | soluble |
| Acidity (pKa) | 2.97[2] |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Gentisic acid is a dihydroxybenzoic acid. It is a derivative of benzoic acid and a minor (1%) product of the metabolic break down of aspirin, excreted by the kidneys.[4]
It is also found in the African tree Alchornea cordifolia and in wine.[5]
Production
Gentisic acid is produced by carboxylation of hydroquinone.[6]
- C
6H
4(OH)
2 + CO
2 → C
6H
3(CO
2H)(OH)
2
This conversion is an example of a Kolbe–Schmitt reaction.
Alternatively the compound can be synthesized from salicylic acid via Elbs persulfate oxidation.[7][8]
Metabolism
In the presence of the enzyme gentisate 1,2-dioxygenase and oxygen, gentisic acid undergoes a ring-opening reaction to give 3-maleylpyruvic acid:[9]
This reaction has been used to detect gentisic acid with a whole-cell biosensor developed from a 3-maleylpyruvic acid-inducible gene expression system and the gentisate 1,2-dioxygenase gene from chemolithoautotrophic bacterium Cupriavidus necator.[10]
Applications
As a hydroquinone, gentisic acid is readily oxidised and is used as an antioxidant excipient in some pharmaceutical preparations.[citation needed]
In the laboratory, it is used as a sample matrix in matrix-assisted laser desorption/ionization (MALDI) mass spectrometry, and has been shown to conveniently detect peptides incorporating the boronic acid moiety by MALDI.[11][12]
References
- ↑ "Gentisic acid - Compound Summary". PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/3469.
- ↑ Haynes, William M., ed (2016). "5". CRC Handbook of Chemistry and Physics (97th ed.). CRC Press. p. 91. ISBN 9781498754293. https://ia800704.us.archive.org/27/items/CRCHandbookOfChemistryAndPhysics97thEdition2016/CRC%20Handbook%20of%20Chemistry%20and%20Physics%20-%2097th%20Edition%20%282016%29.pdf?page=5-91.
- ↑ Haynes, William M., ed (2016). "3". CRC Handbook of Chemistry and Physics (97th ed.). CRC Press. p. 190. ISBN 9781498754293. https://ia800704.us.archive.org/27/items/CRCHandbookOfChemistryAndPhysics97thEdition2016/CRC%20Handbook%20of%20Chemistry%20and%20Physics%20-%2097th%20Edition%20%282016%29.pdf?page=3-190.
- ↑ Levy, G; Tsuchiya, T (1972). "Salicylate accumulation kinetics in man". New England Journal of Medicine 287 (9): 430–2. doi:10.1056/NEJM197208312870903. PMID 5044917.
- ↑ Tian, Rong-Rong; Pan, Qiu-Hong; Zhan, Ji-Cheng; Li, Jing-Ming; Wan, Si-Bao; Zhang, Qing-Hua; Huang, Wei-Dong (2009). "Comparison of Phenolic Acids and Flavan-3-ols During Wine Fermentation of Grapes with Different Harvest Times". Molecules 14 (2): 827–838. doi:10.3390/molecules14020827.
- ↑ Hudnall, Phillip M.. "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a13_499.
- ↑ Behrman, E.J. (1988). Organic Reactions, Volume 35. New York: John Wiley & Sons Inc.. p. 440. ISBN 978-0471832539. https://books.google.com/books?id=sfjdPwAACAAJ&q=0471832537.
- ↑ Schock, R. U. Jr.; Tabern, D. L. (1951). "The Persulfate Oxidation of Salicylic Acid. 2,3,5-Trihydroxybenzoic Acid". The Journal of Organic Chemistry 16 (11): 1772–1775. doi:10.1021/jo50005a018.
- ↑ Sugiyama, Shin-ich; Yano, Keiji; Komagata, Kazuo; Arima, Kei (1960). "Metabolism of Aromatic Compounds by Microbes". Bulletin of the Agricultural Chemical Society of Japan 24 (3): 243–248. doi:10.1271/bbb1924.24.243.
- ↑ Kutraite, I; Augustiniene, E; Malys,N (2024). "Maleylpyruvic Acid-Inducible Gene Expression System and Its Application for the Development of Gentisic Acid Biosensors". Analytical Chemistry 96 (47): 18727-35. doi:10.1021/acs.analchem.4c03906. PMID 39548649.
- ↑ "2,5-Dihidroxybenzoic acid: a new matrix for laser desorption-ionization mass spectrometry". Int. J. Mass Spectrom. Ion Process. 72 (111): 89–102. 1991. doi:10.1016/0168-1176(91)85050-V. Bibcode: 1991IJMSI.111...89S.
- ↑ Crumpton, J.; Zhang, W.; Santos, W. L. (2011). "Facile Analysis and Sequencing of Linear and Branched Peptide Boronic Acids by MALDI Mass Spectrometry". Analytical Chemistry 83 (9): 3548–3554. doi:10.1021/ac2002565. PMID 21449540.
 |
