Chemistry:Glyceryl diacetate
From HandWiki
| |||
| Names | |||
|---|---|---|---|
| Other names
Diacetin; Glycerol diacetate
| |||
| Identifiers | |||
| |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII |
| ||
| |||
| |||
| Properties | |||
| C7H12O5 | |||
| Molar mass | 176.168 g·mol−1 | ||
| Melting point | −30 °C (−22 °F; 243 K) | ||
| Boiling point | 280 °C (536 °F; 553 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Tracking categories (test):
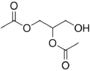
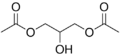
Glyceryl diacetate is a food additive with the E number E1517.[1] This diglyceride is more generally known as diacetin. It is the diester of glycerol and acetylating agents, such as acetic acid and acetic anhydride.[2] It is a colorless, viscous and odorless liquid with a high boiling point. Glycerol diacetate is typically a mixture of two isomers, 1,2-glyceryl diacetate and 1,3-glyceryl diacetate.[3]
See also
References
- ↑ "Call for food additives usage level and/or concentration data in food and beverages intended for human consumption (Batch 7)". EFSA. https://www.efsa.europa.eu/en/consultations/call/180122. Retrieved 1 October 2018.
- ↑ Kong, P. S.; Aroua, M. K.; Daud, W. M. A. W.; Lee, H. V.; Cognet, P.; Peres, Y. (2016). "Catalytic role of solid acid catalysts in glycerol acetylation for the production of bio-additives: a review". RSC Advances 6 (73): 68885–68905. doi:10.1039/C6RA10686B. Bibcode: 2016RSCAd...668885K. https://www.researchgate.net/publication/304528567.
- ↑ Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
 |