Chemistry:Neodymium(III) perchlorate
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
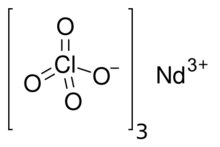
| Nd(ClO4)3 | |
| Molar mass | 442.5929 g/mol (anhydrate) 514.65402 g/mol (tetrahydrate) 523.66166 g/mol (4.5 hydrate) 550.68458 g/mol (hexahydrate) |
| Appearance | light purple crystals (anhydrous)[1] |
| Density | 3.33 g/cm³ (anhydrate)[1] |
| tan | |
| Solubility | soluble in hydrazine |
| Structure | |
| Yb(ReO4)3, α = 90°, β = 90°, γ = 120° | |
a = 0.9341 nm, b = 0.9341 nm, c = 0.5835 nm
| |
| Hazards | |
| GHS pictograms |   [2] [2]
|
| GHS Signal word | Danger[2] |
| H271, H315, H319, H335 | |
| P210, P220, P221, P264, P271, P280, P302, P352, P304, P340, P305, P351, P338, P312, P332, P313, P362, P370, P378, P501 | |
| Related compounds | |
Other cations
|
Praseodymium(III) perchlorate Samarium(III) perchlorate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Neodymium(III) perchlorate is an inorganic compound. It is a salt of neodymium and perchloric acid with the chemical formula of Nd(ClO4)3 – it is soluble in water, forming purple-pink, hydrated crystals.[citation needed]
Properties
Physical properties
Neodymium(III) perchlorate forms pale purple crystals when in its anhydrous form.[citation needed]
It is soluble in water.[citation needed]
It forms crystals Nd(ClO4)3·nH2O, where n = 4, 4.5 are purple-pink crystals, and n = 6 forms pale pink to lavender crystals.[2]
Alkaline salts
Nd(ClO4)3 can form alkaline salts, with the general formula of Nd(OH)x(ClO4)3 − x. The salt with x = 1.5 (saturated with 5 water atoms) is a light purple crystal with d = 2.88 g/cm³.[3]
Other compounds
Nd(ClO4)3 can form compounds with hydrazine, such as Nd(ClO4)3·6N2H4·4H2O which is a small white crystal that is soluble in water, methanol, ethanol and acetone, and insoluble in toluene, with density of 2,3271 g/cm³ at 20 °C.[4]
References
- ↑ 1.0 1.1 Handbook… (Pierre Villars, Karin Cenzual, Roman Gladyshevskii; Walter de Gruyter GmbH & Co KG, 24 thg 7, 2017 - 1970 trang), trang 442. Truy cập 4 tháng 2 năm 2021.
- ↑ 2.0 2.1 2.2 Neodymium(III) Perchlorate Hexahydrate – American Elements ®. Truy cập 4 tháng 2 năm 2021.
- ↑ Villars, Pierre; Cenzual, Karin; Gladyshevskii, Roman (24 July 2017) (in en). Handbook of Inorganic Substances 2017. Walter de Gruyter GmbH & Co KG. p. 3414. ISBN 978-3-11-043655-6. https://books.google.com/books?id=ycw0DwAAQBAJ&pg=PT3414.
- ↑ Izvestii︠a︡ vysshikh uchebnykh zavedeniĭ: Khimii︠a︡ i khimicheskai︠a︡ tekhnologii︠a︡, Tập 16,Số phát hành 1 (Ivanovskiĭ khimiko-tekhnologicheskiĭ in-t, 1973), trang 182. Truy cập 7 tháng 2 năm 2021.
 |

