Chemistry:Pentacyanocobaltate
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
PubChem CID
|
|
| |
| |
| Properties | |
| C5CoN53− | |
| Molar mass | 189.025 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
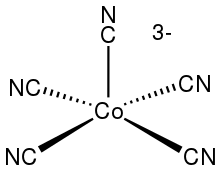
In chemistry, pentacyanocobaltate is the coordination complex with the formula [Co(CN)
5]3−. When crystallized with a quaternary ammonium cation, it can be obtained as a yellow solid. Pentacyanocobaltate attracted attention as an early example of a metal complex that reacts with hydrogen. It contains low-spin cobalt(II), with a doublet ground state.
Synthesis and structure
Aqueous solutions of pentacyanocobaltate are produced by the addition of five or more equivalents of a cyanide salt to a solution of a cobalt(II) salt. Initially this reaction produces insoluble cobalt dicyanide, but this solid dissolves in the presence of the excess cyanide. Pentacyanocobaltate forms within seconds.[1] When prepared using a quaternary ammonium (quat) cyanide, crystals can be obtained with the formula (quat)
3[Co(CN)
5]. According to X-ray crystallography, the salt features square pyamidal [Co(CN)
5]3−.[2]
Reactions
Solutions of [Co(CN)
5]3− undergo a variety of reactions. The complex attracted attention in the 1940s for its reactivity toward hydrogen, which is now understood to produce a cobalt hydride:[1]
- 2[Co(CN)
5]3− + H
2 → 2 [Co(CN)
5H]3−
When allowed to stand as a dilute solution for several minutes, the complex reacts with water to give two Co(III) derivatives:
- 2[Co(CN)
5]3− + H
2O → [Co(CN)
5H]3− + [Co(CN)
5OH]3−
In concentrated solution, the complex dimerizes:
- 2[Co(CN)
5]3− → [(NC)
5Co–Co(CN)
5]6−
With benzyl chloride and related alkylating agents, Co(III) alkyls are formed:[3]
- 2[Co(CN)
5]3− + C
6H
5CH
2Cl → [Co(CN)
5CH
2C
6H
5]3− + [Co(CN)
5Cl]3−
References
- ↑ 1.0 1.1 Kwiatek, Jack (1968). "Reactions Catalyzed by Pentacyanocobaltate(II)". Catalysis Reviews 1: 37–72. doi:10.1080/01614946808064700.
- ↑ Brown, Leo D.; Raymond, Kenneth N. (1975). "Structural Characterization of the Pentacyanocobaltate(II) Anion in the Salt Tris(diethyldiisopropylammonium) Pentacyanocobaltate(II)". Inorganic Chemistry 14 (11): 2590–2594. doi:10.1021/ic50153a002.
- ↑ Chock, Pwen Boon; Halpern, Jack (1969). "Reactions of Pentacyanocobaltate(II) with Some Organic Halides". Journal of the American Chemical Society 91 (3): 582–588. doi:10.1021/ja01031a010.
 |

