Chemistry:Pinolenic acid
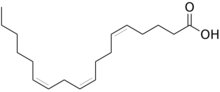
| |
| Names | |
|---|---|
| Preferred IUPAC name
(5Z,9Z,12Z)-Octadeca-5,9,12-trienoic acid | |
| Other names
Columbinic acid
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C18H30O2 | |
| Molar mass | 278.4296 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Pinolenic acid (often misspelled as pinoleic acid) is a fatty acid contained in Siberian Pine nuts, Korean Pine nuts and the seeds and xylem of other pine (Pinus) species. The highest percentage of pinolenic acid is found in Siberian pine nuts and the oil produced from them.[1]
Chemistry and biochemistry
Pinolenic acid is formally designated as all-cis-5,9,12-18:3.[2][3] Some sources also use the term columbinic acid for this substance.[3] But columbinic acid sometimes designates an E-Z isomer (trans,cis,cis delta-5,9,12/18:3) in the biologic literature.[4]
Pinolenic acid is an isomer of gamma-linolenic acid (GLA). GLA is an ω-6 essential fatty acid (EFA) but pinolenic acid is not. However, like the EFAs, it forms biologically active metabolites in the presence of cyclooxygenase or lipoxygenase. These metabolites can partially relieve some of the symptoms of EFA deficiency.[5]
Physiology
Recent research has shown its potential use in weight loss by curbing appetite.[6] Pinolenic acid causes the triggering of two hunger suppressants—cholecystokinin and glucagon-like peptide-1 (GLP-1). Pinolenic acid may have LDL-lowering properties by enhancing hepatic LDL uptake.[7]
Natural occurrence
Oftentimes, it is found in conifers together with other fatty acids (juniperonic, coniferonic, taxoleic, sciadonic acid) that have a double bond in the position 5, separated by more than one methylene group from the next double bond.[8]
References

- ↑ Takala, Rabaa; Ramji, Dipak P.; Choy, Ernest (2023). "The Beneficial Effects of Pine Nuts and Its Major Fatty Acid, Pinolenic Acid, on Inflammation and Metabolic Perturbations in Inflammatory Disorders". International Journal of Molecular Sciences 24 (2): 1171. doi:10.3390/ijms24021171. ISSN 1422-0067.
- ↑ Cyberlipid Center. "POLYENOIC FATTY ACIDS". http://www.cyberlipid.org/fa/acid0003.htm#1e. Retrieved 2007-10-24.
- ↑ 3.0 3.1 PubChem. "PubChem - CID 5312493". https://pubchem.ncbi.nlm.nih.gov/compound/5312493.
- ↑ "Methylene-interrupted double bond in polyunsaturated fatty acid is an essential structure for metabolism by the fatty acid chain elongation system of rat liver". Biochim. Biophys. Acta 1393 (2–3): 299–306. 1998. doi:10.1016/s0005-2760(98)00084-8. PMID 9748638.
- ↑ "Physiologic effects of columbinic acid and its metabolites on rat skin". Biochim. Biophys. Acta 835 (1): 158–60. 1985. doi:10.1016/0005-2760(85)90043-8. PMID 3924106.
- ↑ "Korean pine nut may offer help for obesity". https://news.yahoo.com/s/nm/20060908/hl_nm/indonesia_pinenut_dc. Retrieved 2006-09-08.
- ↑ Lee JW; Lee KW; Lee SW; Kim IH; Rhee C. (April 2004). "Selective increase in pinolenic acid (all-cis-5,9,12-18:3) in Korean pine nut oil by crystallization and its effect on LDL-receptor activity". Lipids 39 (4): 383–7. doi:10.1007/s11745-004-1242-2. PMID 15357026.
- ↑ Pédrono, Frédérique; Boulier-Monthéan, Nathalie; Boissel, Françoise; Ossemond, Jordane; Viel, Roselyne; Fautrel, Alain; Marchix, Justine; Dupont, Didier (10 April 2020). "Sciadonic acid derived from pine nuts as a food component to reduce plasma triglycerides by inhibiting the rat hepatic Δ9-desaturase" (in en). Scientific Reports 10 (1): 6223. doi:10.1038/s41598-020-63301-3. ISSN 2045-2322. PMC 7148351. https://www.nature.com/articles/s41598-020-63301-3. Retrieved 19 May 2025.
 |
