Chemistry:Potassium thioacetate
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
| MeSH | C005732 |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C2H3KOS | |
| Molar mass | 114.21 |
| Appearance | white solid |
| good | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
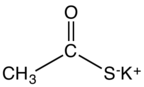
Potassium thioacetate is an organosulfur compound and a salt with the formula CH
3COS−
K+
. This white, water-soluble solid is used as a reagent for preparing thioacetate esters and other derivatives.[1]
Synthesis and reactions
Potassium thioacetate, which is commercially available, can be prepared by combining acetyl chloride and potassium hydrosulfide:
- CH
3COCl + 2 KSH → KCl + CH
3COSK + H
2S
It arises also by the neutralisation of thioacetic acid with potassium hydroxide.
Use in preparation of thiols
In a common application, potassium thioacetate is combined with alkylating agents to give thioacetate esters (X = halide):
- CH
3COSK + RX → CH
3COSR + KX
One example is ethyl thioacetate. Hydrolysis of these esters affords thiols:
- CH
3COSR + H
2O → CH
3CO
2H + RSH
The thioacetate esters can also undergo cleavage with methanethiol in the presence of stoichiometric base, as illustrated in the preparation of pent-4-yne-1-thiol:[2]
- H
3C(CH
2)
3OMs + KSAc → H
3C(CH
2)
3SAc + KOMs - H
3C(CH
2)
3SAc + HSMe → H
3C(CH
2)
3SH + MeSAc
References
- ↑ Zongjun Qiao and Xuefeng Jiang "Potassium Thioacetate" e-EROS Encyclopedia Of Reagents For Organic Synthesis, 2014. doi:10.1002/047084289X.rn01737
- ↑ Matteo Minozzi; Daniele Nanni; Piero Spagnolo (2008). "4-Pentyne-1-thiol". EEROS. doi:10.1002/047084289X.rn00855. ISBN 978-0-471-93623-7.
 |
