Chemistry:Thioacetic acid
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Ethanethioic S-acid[1] | |||
| Other names
Thioacetic S-acid
Thiolacetic acid | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties | |||
| CH 3C(O)SH | |||
| Molar mass | 76.11756 g/mol | ||
| Appearance | Transparent, colorless to light yellow liquid[2][3] | ||
| Odor | Unpleasant, strong thiol-like | ||
| Density | 1.08 g/mL | ||
| Melting point | −58 °C (−72 °F; 215 K) | ||
| Boiling point | 93 °C (199 °F; 366 K) | ||
| −38.4·10−6 cm3/mol | |||
Refractive index (nD)
|
1.465 | ||
| Hazards | |||
| Main hazards | May cause severe skin and eye burns. Highly flammable liquid. | ||
| Safety data sheet | Fischer Scientific | ||
| GHS pictograms |    
| ||
| GHS Signal word | Danger | ||
| HH225Script error: No such module "Preview warning".Category:GHS errors, HH301Script error: No such module "Preview warning".Category:GHS errors, HH302Script error: No such module "Preview warning".Category:GHS errors, HH314Script error: No such module "Preview warning".Category:GHS errors, HH317Script error: No such module "Preview warning".Category:GHS errors | |||
| PP210Script error: No such module "Preview warning".Category:GHS errors, PP233Script error: No such module "Preview warning".Category:GHS errors, PP240Script error: No such module "Preview warning".Category:GHS errors, PP241Script error: No such module "Preview warning".Category:GHS errors, PP242Script error: No such module "Preview warning".Category:GHS errors, PP243Script error: No such module "Preview warning".Category:GHS errors, PP260Script error: No such module "Preview warning".Category:GHS errors, PP261Script error: No such module "Preview warning".Category:GHS errors, PP264Script error: No such module "Preview warning".Category:GHS errors, PP264+P265Script error: No such module "Preview warning".Category:GHS errors, PP270Script error: No such module "Preview warning".Category:GHS errors, PP272Script error: No such module "Preview warning".Category:GHS errors, PP280Script error: No such module "Preview warning".Category:GHS errors, PP301+P316Script error: No such module "Preview warning".Category:GHS errors, PP301+P317Script error: No such module "Preview warning".Category:GHS errors, PP301+P330+P331Script error: No such module "Preview warning".Category:GHS errors, PP302+P352Script error: No such module "Preview warning".Category:GHS errors, PP302+P361+P354Script error: No such module "Preview warning".Category:GHS errors, PP303+P361+P353Script error: No such module "Preview warning".Category:GHS errors, PP304+P340Script error: No such module "Preview warning".Category:GHS errors, PP305+P354+P338Script error: No such module "Preview warning".Category:GHS errors, PP316Script error: No such module "Preview warning".Category:GHS errors, PP317Script error: No such module "Preview warning".Category:GHS errors, PP321Script error: No such module "Preview warning".Category:GHS errors, PP330Script error: No such module "Preview warning".Category:GHS errors | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
75 mg/kg (mouse, intraperitoneal) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
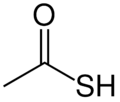
Thioacetic acid is an organosulfur compound with the molecular formula CH
3C(O)SH. It is a thioic acid: the sulfur analogue of acetic acid (CH
3C(O)OH), as implied by the thio- prefix. It is a yellow liquid with a strong thiol-like odor. It is used in organic synthesis for the introduction of thiol groups (–SH) in molecules.[4]
Synthesis and properties
Thioacetic acid is prepared by the reaction of acetic anhydride with hydrogen sulfide:[5]
- (CH
3C(O))
2O + H
2S → CH
3C(O)SH + CH
3C(O)OH
It has also been produced by the action of phosphorus pentasulfide on glacial acetic acid, followed by distillation.[6]
- CH
3C(O)OH + P
2S
5 → CH
3C(O)SH + P
2OS
4
Thioacetic acid is typically contaminated by acetic acid.
The compound exists exclusively as the thiol tautomer, consistent with the strength of the C=O double bond. Reflecting the influence of hydrogen-bonding, the boiling point (93 °C) and melting points are 20 and 75 K lower than those for acetic acid.
Reactivity
Acidity
With a pKa near 3.4, thioacetic acid is about 15 times more acidic than acetic acid.[7] The conjugate base is thioacetate:
- CH
3C(O)SH → CH
3C(O)S−
+ H+
In neutral water, thioacetic acid is fully ionized.
Reactivity of thioacetate
Most of the reactivity of thioacetic acid arises from the conjugate base, thioacetate. Salts of this anion, e.g. potassium thioacetate, are used to generate thioacetate esters.[8] Thioacetate esters undergo hydrolysis to give thiols. A typical method for preparing a thiol from an alkyl halide using thioacetic acid proceeds in four discrete steps, some of which can be conducted sequentially in the same flask:
- CH
3C(O)SH + NaOH → CH
3C(O)SNa + H
2O - CH
3C(O)SNa + RX → CH
3C(O)SR + NaX, where X = Cl, Br, I - CH
3C(O)SR + 2 NaOH → CH
3CO
2Na + RSNa + H
2O - RSNa + HCl → RSH + NaCl
In an application that illustrates the use of its radical behavior, thioacetic acid is used with AIBN in a free radical mediated nucleophilic addition to an exocyclic alkene forming a thioester:[9]
Reductive acetylation
Potassium thioacetate can be used convert nitroarenes to aryl acetamides in one step. This is particularly useful in the preparation of pharmaceuticals, e.g., paracetamol from 4-nitrophenol or 4-nitroanisole.[10]
References
- ↑ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. pp. 97. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ↑ "Thioacetic acid". https://pubchem.ncbi.nlm.nih.gov/compound/Thioacetic-acid.
- ↑ "Identification of the substance/mixture and of the company/undertaking - Thioacetic acid". https://www.sigmaaldrich.com/GB/en/sds/aldrich/t30805.
- ↑ Jeannie R. Phillips "Thiolacetic Acid" Encyclopedia of Reagents for Organic Synthesis, 2001 John Wiley. doi:10.1002/047084289X.rt096
- ↑ Ellingboe, E. K. (1951). "Thiolacetic acid". Organic Syntheses 31: 105. doi:10.15227/orgsyn.031.0105.
- ↑ Schiff, Robert (1895-08-09). "Preparation of Thioacetic Acid and its Importance for Chemical-Legal Investigations". Chemical News and Journal of Industrial Science 72: 64. https://books.google.com/books?id=lSLOAAAAMAAJ&pg=PA412. Retrieved 2016-11-02.
- ↑ Matthys J. Janssen "Carboxylic Acids and Esters" in PATAI'S Chemistry of Functional Groups: Carboxylic Acids and Esters, Saul Patai, Ed. pp. 705–764, 1969. doi:10.1002/9780470771099.ch15
- ↑ Ervithayasuporn, V. (2011). "Synthesis and Characterization of Octakis(3-propyl ethanethioate)octasilsesquioxane". Organometallics 30 (17): 4475–4478. doi:10.1021/om200477a.
- ↑ Synthesis of methyl 6-deoxy-4-O-(sodium sulfonato)-α-L-talopyranoside, its C-4 epimer and both isosteric [4-C-(potassium sulfonatomethyl)] derivatives László Lázár, Magdolna Csávás, Anikó Borbás, Gyöngyi Gyémánt, and András Lipták Arkivoc 2004 (vii) 196-207 Link
- ↑ Bhattacharya, Apurba (2006). "One-step reductive amidation of nitro arenes: application in the synthesis of Acetaminophen". Tetrahedron Letters 47: 1861–1864. doi:10.1016/j.tetlet.2005.09.196. http://www.cs.gordon.edu/~ijl/_lead_papers/One%20step%20reductive%20amidation%20of%20nitroarenes%20-%20Tylenol.pdf. Retrieved 2016-11-02.
 |