Chemistry:Solvent Yellow 7
| |
| Names | |
|---|---|
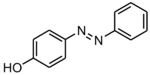
| Preferred IUPAC name
4-(Phenyldiazenyl)phenol | |
| Other names
4-Hydroxyazobenzene
Solvent Yellow 7 Simpsol Yellow | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C12H10N2O | |
| Molar mass | 198.225 g·mol−1 |
| Appearance | An orange solid[2] |
| Melting point | 155 °C (311 °F; 428 K) |
| Slightly soluble in hot water | |
| Solubility in other solvents | Soluble in ethanol, acetone |
| Acidity (pKa) | 8.2 (from the hydroxyl group) |
| Hazards | |
| Main hazards | Irritant |
| Related compounds | |
Related compounds
|
Benzenediazonium chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Solvent Yellow 7 is an aromatic organic molecule and a common azo dye with a formula of C6H5N2C6H4OH.[3] It has a phenolic hydroxyl and an azo group in the same molecule.[4]
Synthesis
Like most azobenzenes, Solvent Yellow 7 can be synthesized by the reaction of the phenyldiazonium salt with phenol. The optimal pH value for this azo coupling is 8.5-10. The reaction is carried out in water, since sodium chloride (or potassium chloride) formed in the reaction is soluble in water, while the product precipitates.[5]

As azo dyes are not usually water soluble, the effect of various solvents on them has been studied analytically, and likewise analytical methods and calculations for the color concentration developed.[6][7]
Further reactions
The molecule can be further reacted including with bromine,[8] and other halogens.[9] Other reactions include nitration.[10] The reactivity with Grignard reagents has also been studied.[11]
Toxicology
The toxicology has been extensively studied,[12] including IARC studies.[13] There have been other extensive reviews.[14]
References
- ↑ "4-PHENYLAZOPHENOL". https://www.chemicalbook.com/ChemicalProductProperty_EN_CB5366310.htm.
- ↑ Solomons, T.W Graham (2017). Organic chemistry (10 ed.). John Wiley and sons. pp. 941. ISBN 978-1119248972. OCLC 973372285.
- ↑ "4-Phenylazophenol - PubChem Compound - NCBI". https://www.ncbi.nlm.nih.gov/pccompound/?term=%221689-82-3%22.
- ↑ "4-PHENYLAZOPHENOL | 1689-82-3" (in en). https://www.chemicalbook.com/ChemicalProductProperty_EN_CB5366310.htm.
- ↑ Estibaliz, Merino. "Synthesis of azobenzenes: the coloured pieces of molecular materials". http://www.chtf.stuba.sk/~szolcsanyi/education/files/Organicka%20chemia%20II/Prednaska%202_Aminy_Reakcie/Doplnkove%20studijne%20materialy/Azobenzenes/Synthesis%20of%20azobenzenes%20-%20the%20coloured%20pieces%20of%20molecular%20materials.pdf.
- ↑ Brode, W. R. (1926-01-01). "The Effect of Solvents on the Absorption Spectrum of a Simple Azo Dye" (in en). The Journal of Physical Chemistry 30 (1): 56–69. doi:10.1021/j150259a006. ISSN 0092-7325. https://pubs.acs.org/doi/abs/10.1021/j150259a006.
- ↑ Moir, James (1922-01-01). "CLXXXV.—The calculation of the colour of the azo-dyes and related coloured substances" (in en). Journal of the Chemical Society, Transactions 121: 1555–1562. doi:10.1039/CT9222101555. ISSN 0368-1645. https://pubs.rsc.org/en/content/articlelanding/1922/ct/ct9222101555.
- ↑ Hewitt, J. T.; Aston, W. G. (1900-01-01). "LXI.—Bromination of benzeneazophenol" (in en). Journal of the Chemical Society, Transactions 77: 712–716. doi:10.1039/CT9007700712. ISSN 0368-1645. https://pubs.rsc.org/en/content/articlelanding/1900/ct/ct9007700712.
- ↑ Hodgson, Herbert H.; Turner, Gerald (1942-01-01). "82. The coupling of m-halogenophenols with diazotised aniline, and the existence of chromoisomerism among the 3-halogeno-4-benzeneazophenols" (in en). Journal of the Chemical Society (Resumed): 433–435. doi:10.1039/JR9420000433. ISSN 0368-1769. https://pubs.rsc.org/en/content/articlelanding/1942/jr/jr9420000433.
- ↑ Hewitt, J. T. (1900-01-01). "X.—Preparation of benzeneazo-o-nitrophenol" (in en). Journal of the Chemical Society, Transactions 77: 99–103. doi:10.1039/CT9007700099. ISSN 0368-1645. https://pubs.rsc.org/en/content/articlelanding/1900/ct/ct9007700099.
- ↑ Gilman, Henry; Bailie, J. Clyde (March 1937). "Relative Reactivities of Organometallic Compounds. Xvii. The Azo Linkage" (in en). The Journal of Organic Chemistry 02 (1): 84–94. doi:10.1021/jo01224a010. ISSN 0022-3263. https://pubs.acs.org/doi/abs/10.1021/jo01224a010.
- ↑ Smith, J. N.; Williams, R. T. (1951-05-01). "Studies in detoxication. 36. A note on the glucuronides of benzeneazophenol and benzeneazoresorcinol" (in en). Biochemical Journal 48 (5): 546–547. doi:10.1042/bj0480546. ISSN 0306-3283. PMID 14838898. PMC 1275371. https://portlandpress.com/biochemj/article/48/5/546/47153/Studies-in-detoxication-36-A-note-on-the.
- ↑ "4-Hydroxyazobenzene (IARC Summary & Evaluation, Volume 8, 1975)". https://inchem.org/documents/iarc/vol08/4-hydroxyazobenzene.html.
- ↑ Walker, R. (1970-01-01). "The metabolism of azo compounds: a review of the literature". Food and Cosmetics Toxicology 8 (6): 659–676. doi:10.1016/S0015-6264(70)80455-2. ISSN 0015-6264. PMID 5500003. https://www.sciencedirect.com/science/article/pii/S0015626470804552.
See also
- Benzenediazonium chloride
- Azo compound
- Solvent Yellow 1
External Websites
 |
