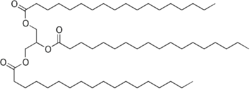
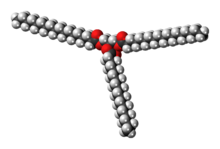
Chemistry:Stearin
| |
| |

| |
| Names | |
|---|---|
| Systematic IUPAC name
Propane-1,2,3-triyl tri(octadecanoate) | |
| Other names
Tristearin; Trioctadecanoin; Glycerol tristearate; Glyceryl tristearate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C57H110O6 | |
| Molar mass | 891.501 g·mol−1 |
| Appearance | White powder |
| Odor | Odorless |
| Density | 0.862 g/cm3 (80 °C) 0.8559 g/cm3 (90 °C)[2] |
| Melting point | 54–72.5 °C (129.2–162.5 °F; 327.1–345.6 K)[3] |
| Insoluble | |
| Solubility | Slightly soluble in C6H6, CCl4 Soluble in acetone, CHCl3 Insoluble in EtOH[2] |
Refractive index (nD)
|
1.4395 (80 °C)[2] |
| Structure | |
| Triclinic (β-form)[4] | |
| P1 (β-form)[4] | |
a = 12.0053 Å, b = 51.902 Å, c = 5.445 Å (β-form)[4] α = 73.752°, β = 100.256°, γ = 117.691°
| |
| Thermochemistry | |
Heat capacity (C)
|
1342.8 J/mol·K (β-form, 272.1 K) 1969.4 J/mol·K (346.5 K)[3][5] |
Std molar
entropy (S |
1534.7 J/mol·K (liquid)[5] |
Std enthalpy of
formation (ΔfH⦵298) |
−2344 kJ/mol[5] |
Std enthalpy of
combustion (ΔcH⦵298) |
35806.7 kJ/mol[5] |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | ≥ 300 °C (572 °F; 573 K) closed cup[6] |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
2000 mg/kg (rats, oral)[6] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Stearin /ˈstɪərɪn/, or tristearin, or glyceryl tristearate is an odourless, white powder. It is a triglyceride derived from three units of stearic acid. Most triglycerides are derived from at least two and more commonly three different fatty acids.[7] Like other triglycerides, stearin can crystallise in three polymorphs. For stearin, these melt at 54 (α-form), 65, and 72.5 °C (β-form).[3]
Note that stearin is also used to mean the solid component of an oil or fat that can be separated into components that melt at higher (the stearin) and lower (the olein) temperatures. This is the usage meant in an example such as palm stearin.
Occurrence
Stearin is obtained from animal fats created as a byproduct of processing beef. It can also be found in tropical plants such as palm. It can be partially purified by dry fractionation by pressing tallow or other fatty mixtures, leading to separation of the higher melting stearin-rich material from the liquid, which is typically enriched in fats derived from oleic acid. It can be obtained by interesterification, again exploiting its higher melting point which allows the higher melting tristearin to be removed from the equilibrated mixture. Stearin is a side product obtained during the extraction of cod liver oil removed during the chilling process at temperatures below −5 °C.
Uses
Stearin is used as a hardening agent in the manufacture of candles and soap.[8] It is mixed with a sodium hydroxide solution in water, creating a reaction which gives glycerin and sodium stearate, the main ingredient in most soap:
- C3H5(C18H35O2)3 + 3 NaOH → C3H5(OH)3 + 3 C18H35OONa
Stearin is also added to aluminium flakes to help in the grinding process in making dark aluminium powder.[clarification needed]
See also
- Candle
- Michel Eugène Chevreul
- Soap making
- Stearic acid
References
- ↑ Merck Index, 11th Edition, 9669.
- ↑ 2.0 2.1 2.2 Lide, David R., ed (2009). CRC Handbook of Chemistry and Physics (90th ed.). Boca Raton, Florida: CRC Press. ISBN 978-1-4200-9084-0.
- ↑ 3.0 3.1 3.2 Charbonnet, G. H.; Singleton, W. S. (1947). "Thermal properties of fats and oils". Journal of the American Oil Chemists' Society 24 (5): 140. doi:10.1007/BF02643296.
- ↑ 4.0 4.1 4.2 Van Langevelde, A.; Peschar, R.; Schenk, H. (2001). "Structure of β-trimyristin and β-tristearin from high-resolution X-ray powder diffraction data". Acta Crystallographica Section B 57 (3): 372–377. doi:10.1107/S0108768100019121. PMID 11373397.
- ↑ 5.0 5.1 5.2 5.3 Tristearin in Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD), http://webbook.nist.gov (retrieved 2014-06-19)
- ↑ 6.0 6.1 6.2 "MSDS of Stearin Wax". Sasol Wax North America Corp.. http://www.swna.us/MSDS/Stearin%20Wax%20MSDS.pdf. Retrieved 2014-06-19.
- ↑ Alfred Thomas (2002). Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a10_173. ISBN 978-3527306732.
- ↑ Geller, L. W. (1935). "Waxes in the candle industry". Oil & Soap 12 (11): 263–265. doi:10.1007/BF02636720.
 |

