Chemistry:Tetraacetylethane
From HandWiki
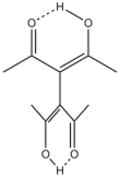
| |
| Names | |
|---|---|
| Other names
3,4-diacetyl-2,5-Hexanedione
| |
| Identifiers | |
PubChem CID
|
|
| Properties | |
| C10H14O4 | |
| Molar mass | 198.218 g·mol−1 |
| Appearance | white solid |
| Density | 1.305 g/cm3 |
| Melting point | 192–193 °C (378–379 °F; 465–466 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Tetraacetylethane is the organic compound with the nominal formula [CH(C(O)CH3)2]2. It is a white solid that has attracted interest as a precursor to heterocycles and metal complexes. It is prepared by oxidation of sodium acetylacetonate:[1]
- I2 + 2 NaCH(C(O)CH3)2 → [CH(C(O)CH3)2]2 + 2 NaI
Reminiscent of the case of acetylacetone, tetraacetylethane exists as the enol, as established by X-ray crystallography. The two C3O2H rings are twisted with a dihedral angle near 90°.[2]
Many metal complexes have been prepared from the conjugate base of this ligand. One example is diruthenium(III) derivative [Ru(acac)2]2[C(C(O)CH3)2]2, which is closely related to ruthenium(III) acetylacetonate.[3]
References
- ↑ Robert G. Charles (1959). "Tetraacetylethane". Organic Syntheses 39: 61. doi:10.15227/orgsyn.039.0061.
- ↑ Paula M. B. Piccoli, Thomas F. Koetzle, Arthur J. Schultz, Elizabeth A. Zhurova, Jernej Stare, A. Alan Pinkerton, Juergen Eckert, and Dusan Hadzi (2008). "Variable Temperature Neutron Diffraction and X-Ray Charge Density Studies of Tetraacetylethane". J. Phys. Chem. A 112 (29): 6667–6677. doi:10.1021/jp800204r. PMID 18593102. Bibcode: 2008JPCA..112.6667P.
- ↑ Koiwa, Tomohiro; Masuda, Yuki; Shono, Junpei; Kawamoto, Yuji; Hoshino, Yoshimasa; Hashimoto, Takeshi; Natarajan, Karuppannan; Shimizu, Kunio (2004). "Synthesis, Characterization, and Detailed Electrochemistry of Binuclear Ruthenium(III) Complexes Bridged by Bisacetylacetonate. Crystal and Molecular Structures of [{Ru(acac)2}2(tae)] (Acac = 2,4-Pentanedionate Ion, tae = 1,1,2,2-Tetraacetylethanate Dianion)". Inorganic Chemistry 43 (20): 6215–6223. doi:10.1021/ic030216c. PMID 15446866.
 |
