Chemistry:Tetrachloroferrate
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| 26231 | |
PubChem CID
|
|
| |
| |
| Properties | |
| Cl4Fe−1 | |
| Molar mass | 197.65 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
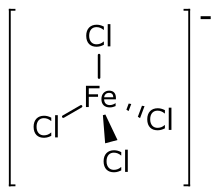
Tetrachloroferrate is the polyatomic ion having chemical formula FeCl−
4. The metallate can be formed when ferric chloride (FeCl
3) abstracts a chloride ion from various other chloride salts.[1] The resulting tetrachloroferrate salts are typically soluble in non-polar solvents. The tetrachloroferrate anion, with iron(III) in the center, has tetrahedral geometry.[2] It is useful as a non-coordinating anion comparable to perchlorate.[3] Several organoammonium salts have been studied for their novel material properties. 1-Butyl-3-methylimidazolium tetrachloroferrate is one of several ionic liquids that are magnetic.[4] Trimethylchloromethylammonium tetrachloroferrate is a plastic crystal that can behave as a molecular switch in response to several different types of inputs.[5]
References
- ↑ Cook, Charles M. Jr.; Dunn, Wendell E. Jr. (1961). "The Reaction of Ferric Chloride with Sodium and Potassium Chlorides". J. Phys. Chem. 65 (9): 1505–1511. doi:10.1021/j100905a008.
- ↑ Lutz, Martin; Huang, Yuxing; Moret, Marc-Etienne; Klein Gebbink, Robertus J. M. (2014). "Phase Transitions and Twinned Low-Temperature Structures of Tetraethylammonium Tetrachloridoferrate(III)". Acta Crystallographica Section C 70 (5): 470–476. doi:10.1107/S2053229614007955. PMID 24816016.
- ↑ Golding, Raymund M.; Harris, CM; Jessop, KJ; Tennant, William C. (1972). "Oxidation of dithiocarbamato metal complexes". Australian Journal of Chemistry 25 (12): 2567–2576. doi:10.1071/CH9722567.
- ↑ Hayashi, Satoshi; Saha, Satyen; Hamaguchi, Hiro-o (2006). "A new class of magnetic fluids: bmim[FeCl4] and nbmim[FeCl4] ionic liquids". IEEE Transactions on Magnetics 42 (1): 12–14. doi:10.1109/TMAG.2005.854875. Bibcode: 2006ITM....42...12H.
- ↑ Li, Dong; Zhao, Xue-Mei; Zhao, Hai-Xia; Long, La-Sheng; Zheng, Lan-Sun (2019). "Coexistence of Magnetic-Optic-Electric Triple Switching and Thermal Energy Storage in a Multifunctional Plastic Crystal of Trimethylchloromethyl Ammonium Tetrachloroferrate(III)". Inorg. Chem. 58 (1): 655–662. doi:10.1021/acs.inorgchem.8b02835. PMID 30576116.
External links
category:Ferrates
 |
