Medicine:EIF5A
 Generic protein structure example |
Eukaryotic translation initiation factor 5A-1 is a protein that in humans is encoded by the EIF5A gene.[1]
It is the only known protein to contain the unusual amino acid hypusine [Nε-(4-amino-2-hydroxybutyl)-lysine], which is synthesized on eIF5A at a specific lysine residue from the polyamine spermidine by two catalytic steps.[2]
EF-P is the bacterial homolog of eIF5A, which is modified post-translationally in a similar but distinct way.[3][4] Both proteins are believed to catalyze peptide bond formation and help resolve ribosomal stalls, making them elongation factors despite the "initiation factor" name originally assigned.[5]
Faundes-Banka syndrome
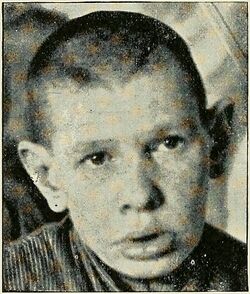
Germline deleterious heterozygous EIF5A variants cause Faundes-Banka syndrome.[6][7] This rare human disorder is characterized by variable combinations of developmental delay, microcephaly, micrognathia and dysmorphic features. It was named after Víctor Faundes and Siddharth Banka, two geneticists who discovered the condition.
See also
EIF5A2
References
- ↑ "The eukaryotic cofactor for the human immunodeficiency virus type 1 (HIV-1) rev protein, eIF-5A, maps to chromosome 17p12-p13: three eIF-5A pseudogenes map to 10q23.3, 17q25, and 19q13.2". Genomics 25 (3): 749–752. February 1995. doi:10.1016/0888-7543(95)80025-H. PMID 7759117.
- ↑ "Posttranslational synthesis of hypusine: evolutionary progression and specificity of the hypusine modification". Amino Acids 33 (2): 341–350. August 2007. doi:10.1007/s00726-007-0525-0. PMID 17476569.
- ↑ "Post-translational modification by β-lysylation is required for activity of Escherichia coli elongation factor P (EF-P)". The Journal of Biological Chemistry 287 (4): 2579–2590. January 2012. doi:10.1074/jbc.M111.309633. PMID 22128152.
- ↑ "Lys34 of translation elongation factor EF-P is hydroxylated by YfcM". Nature Chemical Biology 8 (8): 695–697. August 2012. doi:10.1038/nchembio.1001. PMID 22706199.
- ↑ "eIF5A and EF-P: two unique translation factors are now traveling the same road". Wiley Interdisciplinary Reviews. RNA 5 (2): 209–222. 2013. doi:10.1002/wrna.1211. PMID 24402910.
- ↑ "Impaired eIF5A function causes a Mendelian disorder that is partially rescued in model systems by spermidine". Nature Communications 12 (1): 833. February 2021. doi:10.1038/s41467-021-21053-2. PMID 33547280. Bibcode: 2021NatCo..12..833F.
- ↑ "OMIM Entry - # 619376 - FAUNDES-BANKA SYNDROME; FABAS". https://omim.org/entry/619376.
Further reading
- "Eukaryotic initiation factor 5A activity and HIV-1 Rev function". Biological Signals 6 (3): 124–133. 1997. doi:10.1159/000109118. PMID 9285095.
- "Roles of HIV-1 auxiliary proteins in viral pathogenesis and host-pathogen interactions". Cell Research 15 (11–12): 923–934. 2006. doi:10.1038/sj.cr.7290370. PMID 16354571.
- "Microsequences of 145 proteins recorded in the two-dimensional gel protein database of normal human epidermal keratinocytes". Electrophoresis 13 (12): 960–969. December 1992. doi:10.1002/elps.11501301199. PMID 1286667.
- "Eukaryotic initiation factor 5A: the molecular form of the hypusine-containing protein from human erythrocytes". Biochimica et Biophysica Acta (BBA) - Protein Structure and Molecular Enzymology 1076 (3): 448–451. February 1991. doi:10.1016/0167-4838(91)90490-q. PMID 1900436.
- "Sequence determination and cDNA cloning of eukaryotic initiation factor 4D, the hypusine-containing protein". The Journal of Biological Chemistry 264 (3): 1578–1583. January 1989. doi:10.1016/S0021-9258(18)94226-2. PMID 2492279.
- "Eukaryotic initiation factor 4D. Purification from human red blood cells and the sequence of amino acids around its single hypusine residue". The Journal of Biological Chemistry 261 (31): 14515–14519. November 1986. doi:10.1016/S0021-9258(18)66899-1. PMID 3095320.
- "The genomic structure encoding human initiation factor eIF-5A". Gene 144 (2): 249–252. July 1994. doi:10.1016/0378-1119(94)90385-9. PMID 7545941.
- "Isolation and structural characterization of different isoforms of the hypusine-containing protein eIF-5A from HeLa cells". Biochemistry 34 (45): 14693–14702. November 1995. doi:10.1021/bi00045a010. PMID 7578077.
- "Identification of a new member of the human eIF-5A gene family". Gene 159 (2): 283–284. July 1995. doi:10.1016/0378-1119(95)00136-T. PMID 7622067.
- "Structural features of the eIF-5A precursor required for posttranslational synthesis of deoxyhypusine". The Journal of Biological Chemistry 269 (41): 25916–25921. October 1994. doi:10.1016/S0021-9258(18)47333-4. PMID 7929297.
- "Induced gene expression of the hypusine-containing protein eukaryotic initiation factor 5A in activated human T lymphocytes". Proceedings of the National Academy of Sciences of the United States of America 91 (23): 10829–10833. November 1994. doi:10.1073/pnas.91.23.10829. PMID 7971969. Bibcode: 1994PNAS...9110829B.
- "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene 138 (1–2): 171–174. January 1994. doi:10.1016/0378-1119(94)90802-8. PMID 8125298.
- "Eukaryotic initiation factor 5A is a cellular target of the human immunodeficiency virus type 1 Rev activation domain mediating trans-activation". The Journal of Cell Biology 123 (6 Pt 1): 1309–1320. December 1993. doi:10.1083/jcb.123.6.1309. PMID 8253832.
- "Interaction of eukaryotic initiation factor 5A with the human immunodeficiency virus type 1 Rev response element RNA and U6 snRNA requires deoxyhypusine or hypusine modification". Biological Signals 6 (3): 166–174. 1997. doi:10.1159/000109123. PMID 9285100.
- "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene 200 (1–2): 149–156. October 1997. doi:10.1016/S0378-1119(97)00411-3. PMID 9373149.
- "Identification of the eukaryotic initiation factor 5A as a retinoic acid-stimulated cellular binding partner for tissue transglutaminase II". The Journal of Biological Chemistry 273 (4): 1946–1950. January 1998. doi:10.1074/jbc.273.4.1946. PMID 9442029.
- "Interaction of the HIV-1 rev cofactor eukaryotic initiation factor 5A with ribosomal protein L5". Proceedings of the National Academy of Sciences of the United States of America 95 (4): 1607–1612. February 1998. doi:10.1073/pnas.95.4.1607. PMID 9465063. Bibcode: 1998PNAS...95.1607S.
- "Complex formation between deoxyhypusine synthase and its protein substrate, the eukaryotic translation initiation factor 5A (eIF5A) precursor". The Biochemical Journal 340 (1): 273–281. May 1999. doi:10.1042/0264-6021:3400273. PMID 10229683.
 |
