Medicine:Venipuncture
In medicine, venipuncture or venepuncture is the process of obtaining intravenous access for the purpose of venous blood sampling (also called phlebotomy) or intravenous therapy. In healthcare, this procedure is performed by medical laboratory scientists, medical practitioners, some EMTs, paramedics, phlebotomists, dialysis technicians, and other nursing staff.[1] In veterinary medicine, the procedure is performed by veterinarians and veterinary technicians.
It is essential to follow a standard procedure for the collection of blood specimens to get accurate laboratory results. Any error in collecting the blood or filling the test tubes may lead to erroneous laboratory results.[2]
Venipuncture is one of the most routinely performed invasive procedures and is carried out for any of five reasons:
- to obtain blood for diagnostic purposes;
- to monitor levels of blood components;[3]
- to administer therapeutic treatments including medications, nutrition, or chemotherapy;
- to remove blood due to excess levels of iron or erythrocytes (red blood cells); or
- to collect blood for later uses, mainly transfusion either in the donor or in another person.
Blood analysis is an important diagnostic tool available to clinicians within healthcare.
Blood is most commonly obtained from the superficial veins of the upper limb.[1] The median cubital vein, which lies within the cubital fossa anterior to the elbow, is close to the surface of the skin without many large nerves positioned nearby. Other veins that can be used in the cubital fossa for venipuncture include the cephalic, basilic, and median antebrachial veins.[4]
Minute quantities of blood may be taken by fingerstick sampling and collected from infants by means of a heelprick or from scalp veins with a winged infusion needle.
Phlebotomy (incision into a vein) is also the treatment of certain diseases such as hemochromatosis and primary and secondary polycythemia.
Complications
A 1996 study of blood donors (a larger needle is used in blood donation than in routine venipuncture) found that 1 in 6,300 donors sustained a nerve injury.[5]
Among the risks and side affects are dizziness, sweating, or a drop in the patient's heart rate and blood pressure.[6]
Equipment

There are many ways in which blood can be drawn from a vein, and the method used depends on the person's age, the equipment available, and the type of tests required.
With evacuated or vacuum tubes
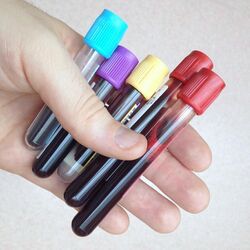
The basic Evacuated Tube System (ETS) consists of a needle, a tube holder, and the evacuated tubes. The needle is attached to the tube holder by the phlebotomist prior to collection, or may come from the manufacturer as one unit. The needle protrudes through the end of the tube holder, and has a needle on each end. After first cleaning the venipuncture site and applying a tourniquet, the phlebotomist uncaps the needle attached to the tube holder, inserts the needle into the vein, then slides evacuated tubes into the tube holder, where the tube's stopper is pierced by the back end of the needle. The vacuum in the tube then automatically draws the needed blood directly from the vein. Multiple vacuum tubes can be attached to and removed in turn from a single needle, allowing multiple samples to be obtained from a single procedure. This is possible due to the multiple sample sleeve, which is a flexible rubber fitting over the posterior end of the needle cannula which seals the needle until it is pushed out of the way. This keeps blood from freely draining out of the back of the needle inserted in the vein, as each test tube is removed and the next impaled. OSHA safety regulations require that needles or tube holders come equipped with a safety device to cover the needle after the procedure to prevent accidental needle stick injury.[7]
There are several needle gauges for a phlebotomist to choose from. The most commonly used are as follows: a 21g (green top) needle, a 22g (black top) needle, a 21g (green label) butterfly needle, a 23g (light blue label) butterfly needle, and a 25g (orange or dark blue label) butterfly needle (however this needle is only used in pediatrics or extreme cases as it is so small that it can often result in hemolyzing the blood sample). There are also a variety of tube and bottle sizes and volumes for different test requirements.[8]
Additives and order of draw
Test tubes are labeled with the additive they contain, but the stopper on each tube is color coded according to additive as well. While colors vary between manufacturers, stopper colors generally are associated with each additive as listed below. Because the additives from each tube can be left on the needle used to fill the tubes, they must be drawn in a specific order to ensure that cross contamination will not negatively affect testing of the samples if multiple tubes are to be drawn at once. The "order of draw" varies by collection method. Below in the order of draw generally required for the Evacuated Tube System (ETS) collection method are the most common tubes, listing additive and color:[9][10]
Template:Table of blood sampling tubes
In children
Use of lidocaine iontophoresis is effective for reducing pain and alleviating distress during venipuncture in children.[11] A needle-free powder lignocaine delivery system has been shown to decrease the pain of venipuncture in children.[12][13] Rapid dermal anesthesia can be achieved by local anesthetic infiltration, but it may evoke anxiety in children frightened by needles or distort the skin, making vascular access more difficult and increasing the risk of needle exposure to health care workers. Dermal anesthesia can also be achieved without needles by the topical application of local anesthetics or by lidocaine iontophoresis. By contrast, noninvasive dermal anesthesia can be established in 5–15 min without distorting underlying tissues by lidocaine iontophoresis, where a direct electric current facilitates dermal penetration of positively charged lidocaine molecules when placed under the positive electrode.
One study concluded that the iontophoretic administration of lidocaine was safe and effective in providing dermal anesthesia for venipuncture in children 6–17 years old. This technique may not be applicable to all children. Future studies may provide information on the minimum effective iontophoretic dose for dermal anesthesia in children and the comparison of the anesthetic efficacy and satisfaction of lidocaine iontophoresis with topical anesthetic creams and subcutaneous infiltration.[11]
Non-pharmacological treatments for pain associated with venipuncture in children includes hypnosis and distraction. These treatments reduced self reported pain and when combined with cognitive-behavioural therapy (CBT) the reduction of pain was even greater. Other interventions have not been found to be effective and these are suggestion, blowing out air, and distraction with parent coaching did not differ from control for pain and distress.[14]
With needle and syringe
Some health care workers prefer to use a syringe-needle technique for venipuncture. Sarstedt manufactures a blood-drawing system (S-Monovette) that uses this principle.[15] This method can be preferred on the elderly, those with cancer, severe burns, obesity, or where the veins are unreliable or fragile. Because syringes are manually operated, the amount of suction applied may be easily controlled. This is particularly helpful when veins are small which may collapse under the suction of an evacuated tube. In children or other circumstances where the quantity of blood gained may be limited it can be helpful to know how much blood can be obtained before distributing it amongst the various additives that the laboratory will require. Another alternative is drawing blood from indwelling cannulae.[16]
Blood cultures
There are times when a blood culture collection is required. The culture will determine if there are pathogens in the blood. Normally blood is sterile. When drawing blood from cultures use a sterile solution such as Betadine rather than alcohol. This is done using sterile gloves, while not wiping away the surgical solution, touching the puncture site, or in any way compromising the sterile process. It is vital that the procedure is performed in as sterile a manner as possible as the persistent presence of skin commensals in blood cultures could indicate endocarditis but they are most often found as contaminants. It is encouraged to use an abrasive method of skin preparation. This removes the upper layers of dead skin cells along with their contaminating bacteria.[17] Povidone-iodine has traditionally been used but in the UK a 2% chlorhexidine in 70% ethanol or isopropyl alcohol solution is preferred and time must be allowed for it to dry. The tops of any containers used when drawing a blood culture should also be disinfected using a similar solution. Some labs will actively discourage iodine use where iodine is thought to degrade the rubber stopper through which blood enters the bottle, thus allowing contaminates to enter the container.
Taking blood samples from animals
Blood samples from living laboratory animals may be collected using following methods:[18][19]
- Blood collection not requiring anesthesia:
- Saphenous vein (rat, mice, guinea pig)
- Dorsal pedal vein (rat, mice)
- Blood collection requiring anesthesia (local/general anesthesia):
- Tail vein (rat, mice)
- Tail snip (mice)
- Orbital sinus (rat, mice)
- Jugular vein (rat, mice)
- Temporary cannula (rat, mice)
- Blood vessel cannulation (guinea pig, ferret)
- Tarsal vein (guinea pig)
- Marginal ear vein or artery (rabbit)
- Terminal procedure:
- Cardiac puncture (rat, mice, guinea pig, rabbit, ferret)
- Orbital sinus (rat, mice)
- Posterior vena cava (rat, mice)
The volume of the blood sample collection is very important in experimental animals. All nonterminal blood collection without replacement of fluids is limited up to 10% of total circulating blood volume in healthy, normal, adult animals on a single occasion and collection may be repeated after three to four weeks. In case repeated blood samples are required at short intervals, a maximum of 0.6 ml/kg/day or 1.0% of an animal's total blood volume can be removed every 24 hours. The estimated blood volume in adult animals is 55 to 70 ml/kg body weight. Care should be taken for older and obese animals. If blood collection volume exceeds more than 10% of total blood volume, fluid replacement may be required. Lactated Ringer's solution (LRS) is recommended as the best fluid replacement by National Institutes of Health (NIH). If the volume of blood collection exceeds more than 30% of the total circulatory blood volume, adequate care should be taken so that the animal does not develop hypovolemia.[19]
Blood alcohol tests
See also
- Arterial blood is taken from an artery instead of a vein
References
- ↑ 1.0 1.1 Higgins, Dan (28 September 2004). "Venepuncture". Nursing Times 100 (39): 30–1. PMID 15500234. https://www.nursingtimes.net/clinical-archive/infection-control/venepuncture-28-09-2004/.
- ↑ Dg, Dayyal (22 May 2018). "Procedures for the collection of blood for hemotological investigations". BioScience Pakistan. ISSN 2521-5760. https://www.bioscience.com.pk/topics/hemotology/item/796-procedures-for-the-collection-of-blood-for-hemotological-investigations.
- ↑ Lavery, I; Ingram, P (August 2005). "Venepuncture: Best Practice.". Nurs Stand 19 (49): 55–65. doi:10.7748/ns2005.08.19.49.55.c3936. PMID 16134421.
- ↑ Ialongo, Cristiano; Bernardini, Sergio (2016-02-15). "Phlebotomy, a bridge between laboratory and patient". Biochemia Medica 26 (1): 17–33. doi:10.11613/BM.2016.002. ISSN 1330-0962. PMID 26981016.
- ↑ Japenga, Ann (30 May 2006). "'A Little Sting' Can Become a Debilitating Injury". The New York Times. https://www.nytimes.com/2006/05/30/health/30case.html.
- ↑ Nelson, Angela. "What Is Phlebotomy?" (in en). https://www.webmd.com/a-to-z-guides/what-is-phlebotomy.
- ↑ McCall, Ruth (2020). Phlebotomy Essentials (7th ed.). Wolters Kluwer. p. 193. ISBN 9781496387073.
- ↑ Perry, Sally (2007). "Ask the Lab: Hemolyze blood specimens". Nursing.advanceweb.com. http://nursing.advanceweb.com/Article/Ask-the-Lab-Hemolyze-blood-specimens.aspx.
- ↑ "Collection" (in en). https://www.osfhealthcare.org/lab/specimens/collection/.
- ↑ Christopher Parker (2019-01-31). "Phlebotomy Services". https://www.uncmedicalcenter.org/mclendon-clinical-laboratories/test-specifications/tube-guide/.
- ↑ 11.0 11.1 Rose, John B.; Galinkin, Jeffrey L.; Jantzen, Ellen C.; Chiavacci, Rosetta M. (2002). "A Study of Lidocaine Iontophoresis for Pediatric Venipuncture". Anesthesia & Analgesia 94 (4): 867–71, table of contents. doi:10.1097/00000539-200204000-00017. PMID 11916787.
- ↑ Lacroix, Helene (October 2008). "A needle-free powder lignocaine delivery system reduced the pain of venipuncture in children" (in en). Evidence-Based Nursing 11 (4): 106. doi:10.1136/ebn.11.4.106. PMID 18815318.
- ↑ Robertson, J (January 2006). "Liposomal lidocaine improved intravenous cannulation success rates in children.". Evid-Based Nurs 9 (1): 10. doi:10.1136/ebn.9.1.10. PMID 16437788.
- ↑ Robertson, J (July 2007). "Review: Distraction, Hypnosis, and Combined Cognitive-Behavioural Interventions Reduce Needle Related Pain and Distress in Children and Adolescents". Evid-Based Nurs 10 (3): 75. doi:10.1136/ebn.10.3.75. PMID 17596380.
- ↑ "Blood collection with the S-Monovette". https://www.sarstedt.com/fileadmin/user_upload/99_Gebrauchsanweisungen/Englisch_US_Code/644_c_PosterA3_AnleitungVenoeseBE_SafetyKanuele_GB_US_0314.pdf.
- ↑ Lesser, Finnian D; Lanham, David A; Davis, Daniel (6 May 2020). "Blood sampled from existing peripheral IV cannulae yields results equivalent to venepuncture: a systematic review". JRSM Open 11 (5). doi:10.1177/2054270419894817. PMID 32523703.
- ↑ Garcia, RA; Spitzer, ED; Beaudry, J; Beck, C; Diblasi, R; Gilleeny-Blabac, M; Haugaard, C; Heuschneider, S et al. (November 2015). "Multidisciplinary Team Review of Best Practices for Collection and Handling of Blood Cultures to Determine Effective Interventions for Increasing the Yield of True-positive Bacteremias, Reducing Contamination, and Eliminating False-positive Central Line-associated Bloodstream Infections". Am J Infect Control 43 (11): 1222–37. doi:10.1016/j.ajic.2015.06.030. PMID 26298636.
- ↑ Hoff, J. (2000). "Methods of Blood Collection in the Mouse". Lab Animal 29 (10): 47–53. http://www.lawte.org/materials/hoff.pdf. Retrieved 2013-12-29.
- ↑ 19.0 19.1 Parasuraman, S; Raveendran, R; Kesavan, R (2010). "Blood sample collection in small laboratory animals". Journal of Pharmacology and Pharmacotherapeutics 1 (2): 87–93. doi:10.4103/0976-500X.72350. PMID 21350616.
 |
