Physics:Fluorine-18
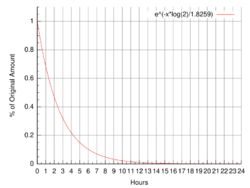 Decay over 24 hours | |
| General | |
|---|---|
| Symbol | 18F |
| Names | fluorine-18, F-18, Fluorine-18 |
| Protons | 9 |
| Neutrons | 9 |
| Nuclide data | |
| Natural abundance | Radioisotope |
| Half-life | 109.734 min[1] |
| Decay products | 18O |
| Isotope mass | 18.0009380[2] u |
| Spin | 1+ |
| Excess energy | 873.431±0.593 keV |
| Binding energy | 137369.199±0.593 keV |
| Decay modes | |
| Decay mode | Decay energy (MeV) |
| Positron emission (97%) | 0.6335[3] |
| Electron capture (3%) | 1.6555[4] |
| Isotopes of Chemistry:fluorine Complete table of nuclides | |
Fluorine-18 (18F, also called radiofluorine) is a fluorine radioisotope which is an important source of positrons. Its half-life is 109.734 minutes, less than two hours, and one of the shortest of radioisotopes with use outside research. It decays by positron emission 96.7% of the time and electron capture 3.3% of the time. Both modes of decay yield stable oxygen-18.
Natural occurrence
18F is a cosmogenic trace radioisotope produced by spallation of atmospheric argon or by reaction of protons with oxygen-18 in the air: 18O + p → 18F + n.[5]
Synthesis
In the radiopharmaceutical industry, fluorine-18 is made using either a cyclotron or linear particle accelerator to bombard a target, usually of natural or enriched [18O]water[6] with high energy protons (typically ~18 MeV). The fluorine produced is in the form of a water solution of [18F]fluoride, which is then used in a rapid chemical synthesis of various radiopharmaceuticals; this must be done after the fluorine is produced as chemical bonds would be destroyed by the production (radiolysis).
History
First published synthesis and report of properties of fluorine-18 were in 1937 by Arthur H. Snell, produced by the nuclear reaction of 20Ne(d,α)18F in the cyclotron laboratories of Ernest O. Lawrence.[7]
Chemistry
Fluorine-18 is often substituted for a hydroxyl group (–OH) in a radiotracer parent molecule, due to similar steric and electrostatic properties. This may however be problematic in certain applications due to possible changes in the molecule polarity.
Applications
Fluorine-18 is one of the early tracers used in positron emission tomography (PET), having been in use since the 1960s.[8] Its significance is due to both its short half-life and the emission of positrons when decaying. A major medical use of fluorine-18 is: in positron emission tomography (PET) to image the brain and heart; to image the thyroid gland; as a radiotracer to image bones and seeking cancers that have metastasized from other locations in the body and in radiation therapy treating internal tumors.
Tracers include sodium fluoride which can be useful for skeletal imaging as it displays high and rapid bone uptake accompanied by very rapid blood clearance, which results in a high bone-to-background ratio in a short time[9] and fluorodeoxyglucose (FDG), where the 18F substitutes a hydroxyl. New dioxaborolane chemistry enables radioactive fluoride (18F) labeling of antibodies, which allows for positron emission tomography (PET) imaging of cancer.[10] A Human-Derived, Genetic, Positron-emitting and Fluorescent (HD-GPF) reporter system uses a human protein, PSMA and non-immunogenic, and a small molecule that is positron-emitting (18F) and fluorescent for dual modality PET and fluorescence imaging of genome modified cells, e.g. cancer, CRISPR/Cas9, or CAR T-cells, in an entire mouse.[11] The dual-modality small molecule targeting PSMA was tested in humans and found the location of primary and metastatic prostate cancer, fluorescence-guided removal of cancer, and detects single cancer cells in tissue margins.[12]
References
- ↑ Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties". Chinese Physics C 45 (3). doi:10.1088/1674-1137/abddae. https://www-nds.iaea.org/amdc/ame2020/NUBASE2020.pdf.
- ↑ Template:AME2020 II
- ↑ Less than the actual decay energy by that released in annihilation.
- ↑ National Nuclear Data Center. "NuDat 2.x database". Brookhaven National Laboratory. http://www.nndc.bnl.gov/nudat2/.
- ↑ SCOPE 50 - Radioecology after Chernobyl , the Scientific Committee on Problems of the Environment (SCOPE), 1993. See table 1.9 in Section 1.4.5.2.
- ↑ Fowler J. S. and Wolf A. P. (1982). The synthesis of carbon-11, fluorine-18 and nitrogen-13 labeled radiotracers for biomedical applications. Nucl. Sci. Ser. Natl Acad. Sci. Natl Res. Council Monogr. 1982.
- ↑ Anonymous (1937-01-15). #5 shows the abstract of Arthur H. Snell about the discovery of the first produced fluorine-18.. "Minutes of the Pasadena Meeting, December 18 and 19, 1936" (in en). Physical Review 51 (2): 142–150. doi:10.1103/PhysRev.51.142. ISSN 0031-899X. Bibcode: 1937PhRv...51..142.. https://link.aps.org/doi/10.1103/PhysRev.51.142.
- ↑ Blau, Monte; Ganatra, Ramanik; Bender, Merrill A. (January 1972). "18F-fluoride for bone imaging". Seminars in Nuclear Medicine 2 (1): 31–37. doi:10.1016/S0001-2998(72)80005-9. PMID 5059349.
- ↑ Ordonez, A. A.; DeMarco, V. P.; Klunk, M. H.; Pokkali, S.; Jain, S.K. (October 2015). "Imaging Chronic Tuberculous Lesions Using Sodium [18FFluoride Positron Emission Tomography in Mice."]. Molecular Imaging and Biology 17 (5): 609–614. doi:10.1007/s11307-015-0836-6. PMID 25750032.
- ↑ Rodriguez, Erik A.; Wang, Ye; Crisp, Jessica L.; Vera, David R.; Tsien, Roger Y.; Ting, Richard (2016-04-27). "New Dioxaborolane Chemistry Enables [18F-Positron-Emitting, Fluorescent [18F]-Multimodality Biomolecule Generation from the Solid Phase"] (in EN). Bioconjugate Chemistry 27 (5): 1390–1399. doi:10.1021/acs.bioconjchem.6b00164. PMID 27064381.
- ↑ Guo, Hua; Harikrishna, Kommidi; Vedvyas, Yogindra; McCloskey, Jaclyn E; Zhang, Weiqi; Chen, Nandi; Nurili, Fuad; Wu, Amy P et al. (2019-05-23). "A fluorescent, [ 18 F-positron-emitting agent for imaging PMSA allows genetic reporting in adoptively-transferred, genetically-modified cells"] (in en). ACS Chemical Biology 14 (7): 1449–1459. doi:10.1021/acschembio.9b00160. ISSN 1554-8929. PMID 31120734.
- ↑ Aras, Omer; Demirdag, Cetin; Kommidi, Harikrishna; Guo, Hua; Pavlova, Ina; Aygun, Aslan; Karayel, Emre; Pehlivanoglu, Hüseyin et al. (March 2021). "Small Molecule, Multimodal [18F-PET and Fluorescence Imaging Agent Targeting Prostate Specific Membrane Antigen: First-in-Human Study"] (in en). Clinical Genitourinary Cancer 19 (5): 405–416. doi:10.1016/j.clgc.2021.03.011. PMID 33879400.
| Lighter: fluorine-17 |
Fluorine-18 is an isotope of fluorine |
Heavier: fluorine-19 |
| Decay product of: neon-18 |
Decay chain of fluorine-18 |
Decays to: oxygen-18 |
 |
