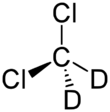
Chemistry:Deuterated dichloromethane
From HandWiki
Revision as of 08:42, 3 July 2022 by imported>TextAI2 (linkage)
|
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Dichloro(2H2)methane | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 1733318 | |||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID
|
|||
| UN number | 1593 | ||
| |||
| |||
| Properties | |||
| C2H2Cl2 | |||
| Molar mass | 86.945 g mol−1 | ||
| Density | 1.362 g cm−3 | ||
| Boiling point | 40 °C (104 °F; 313 K) | ||
| Vapor pressure | 52.6 kPa (at 20 °C) | ||
| Hazards | |||
| GHS pictograms |  
| ||
| GHS Signal word | Warning | ||
| H315, H319, H335, H351, H373 | |||
| P201, P202, P260, P261, P264, P271, P280, P281, P302+352, P304+340, P305+351+338, P308+313, P312, P314, P321, P332+313, P337+313, P362, P403+233, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Related compounds | |||
Related compounds
|
Deuterated chloroform | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Deuterated dichloromethane (CD2Cl2) is a form (called an isotopologue) of dichloromethane (DCM, CH2Cl2) in which the hydrogen atoms ("H") are replaced with deuterium (heavy hydrogen) isotope ("D").[1] Deuterated DCM is not a common solvent used in NMR spectroscopy as it is expensive compared to deuterated chloroform.
References
- ↑ Bertolini, Alessandro; Carelli, Giorgio; Moretti, Augusto; Moruzzi, Giovanni (2001). "Assignment of Fir Laser Lines of Fully Deuterated Dichloromethane". International Journal of Infrared and Millimeter Waves 22 (10): 1421–1431. doi:10.1023/A:1015082422018.
 |