Biology:1,8-cineole synthase
From HandWiki
| 1,8-cineole synthase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC number | 4.2.3.108 | ||||||||
| CAS number | 110637-19-9 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
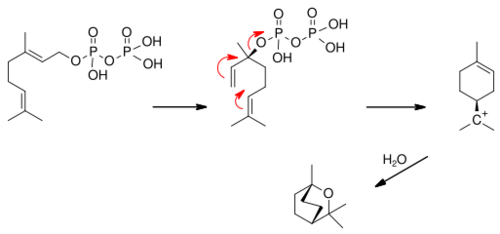
1,8-Cineole synthase (EC 4.2.3.108, 1,8-cineole cyclase, geranyl pyrophoshate:1,8-cineole cyclase, 1,8-cineole synthetase) is an enzyme with systematic name geranyl-diphosphate diphosphate-lyase (cyclizing, 1,8-cineole-forming).[1][2][3][4][5] This enzyme catalyses the following chemical reaction
This enzyme requires Mn2+ or Zn2+. Geranyl diphosphate first isomerizes to (S)-linalyl diphosphate which ionises to the alpha-terpinyl cation which reacts with water to form the product.[6]
References
- ↑ "Biosynthesis of monoterpenes: partial purification, characterization, and mechanism of action of 1,8-cineole synthase". Archives of Biochemistry and Biophysics 309 (1): 184–192. February 1994. doi:10.1006/abbi.1994.1101. PMID 8117108.
- ↑ "Monoterpene synthases from common sage (Salvia officinalis). cDNA isolation, characterization, and functional expression of (+)-sabinene synthase, 1,8-cineole synthase, and (+)-bornyl diphosphate synthase". The Journal of Biological Chemistry 273 (24): 14891–14899. June 1998. doi:10.1074/jbc.273.24.14891. PMID 9614092.
- ↑ "Alternative termination chemistries utilized by monoterpene cyclases: chimeric analysis of bornyl diphosphate, 1,8-cineole, and sabinene synthases". Archives of Biochemistry and Biophysics 417 (2): 203–211. September 2003. doi:10.1016/s0003-9861(03)00347-3. PMID 12941302.
- ↑ "Characterization of a root-specific Arabidopsis terpene synthase responsible for the formation of the volatile monoterpene 1,8-cineole". Plant Physiology 135 (4): 1956–1966. August 2004. doi:10.1104/pp.104.044388. PMID 15299125.
- ↑ "Functional and evolutionary relationships between terpene synthases from Australian Myrtaceae". Phytochemistry 71 (8–9): 844–852. June 2010. doi:10.1016/j.phytochem.2010.03.013. PMID 20399476. Bibcode: 2010PChem..71..844K.
- ↑ "A detailed view on 1,8-cineol biosynthesis by Streptomyces clavuligerus". Beilstein Journal of Organic Chemistry 12: 2317–2324. 2016-11-04. doi:10.3762/bjoc.12.225. PMID 28144299.
External links
- 1,8-cineole+synthase at the US National Library of Medicine Medical Subject Headings (MeSH)
 |