Biology:Mechanism of autism
The mechanisms of autism are the molecular and cellular processes believed to cause or contribute to the symptoms of autism. Multiple processes are hypothesized to explain different autistic features. These hypotheses include defects in synapse structure and function,[1][2] reduced synaptic plasticity,[3] disrupted neural circuit function, gut–brain axis dyshomeostasis,[4][5][6] neuroinflammation,[7] and altered brain structure or connectivity.[8][9][10][11] Autism symptoms stem from maturation-related changes in brain systems.[9] The mechanisms of autism are divided into two main areas: pathophysiology of brain structures and processes, and neuropsychological linkages between brain structures and behaviours, with multiple pathophysiologies linked to various autism behaviours.[10]
Evidence suggests gut–brain axis abnormalities may contribute to autism.[6][4] Studies propose that immune, gastrointestinal inflammation, autonomic nervous system dysfunction, gut microbiota alterations, and dietary metabolites may contribute to brain neuroinflammation and dysfunction.[5] Additionally, enteric nervous system abnormalities could play a role in neurological disorders by allowing disease pathways from the gut to impact the brain.[5]
Synaptic dysfunction also appears to be implicated in autism, with some mutations disrupting synaptic pathways involving cell adhesion.[2] Evidence points to teratogens affecting the early developmental stages, suggesting autism arises very early, possibly within the first eight weeks after conception.[12]
Neuroanatomical studies support that autism may involve abnormal neuronal growth and pruning, leading to brain enlargement in some areas and reduction in others.[13] Functional neuroimaging studies show reduced activation in somatosensory cortices during theory of mind tasks in autistic individuals and highlight potential imbalances in neurotransmitters like glutamate and Γ-aminobutyric acid that may underlie autism's behavioural manifestations.[14]
Pathophysiology
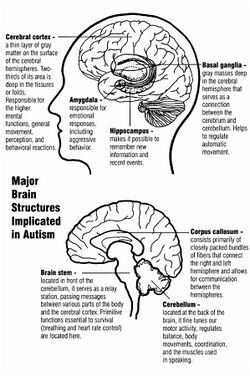
Unlike some brain disorders which have clear molecular hallmarks that can be observed in every affected individual, such as Alzheimer's disease or Parkinson's disease, autism does not have a unifying mechanism at the molecular, cellular, or systems level. The autism spectrum may comprise a small set of disorders that converge on a few common molecular pathways, or it may be a large set of disorders with diverse mechanisms.[16] Autism appears to result from developmental factors that affect many or all functional brain systems.[17] Some factors may disturb the timing of brain development rather than the final product.[15]
Listed below are some characteristic findings in ASD brains on molecular and cellular levels regardless of the specific genetic variation or mutation contributing to autism in a particular individual:
- Limbic system with smaller neurons that are more densely packed together. Given that the limbic system is the main centre of emotions and memory in the human brain, this observation may explain social impairment in ASD.[18]
- Fewer and smaller Purkinje neurons in the cerebellum. New research suggest a role of the cerebellum in emotional processing and language.[18]
- Increased number of astrocytes and microglia in the cerebral cortex. These cells provide metabolic and functional support to neurons and act as immune cells in the nervous system, respectively.[18]
- Increased brain size in early childhood causing macrocephaly in 15–20% of ASD individuals. The brain size however normalizes by mid-childhood. This variation in brain size in not uniform in the ASD brain with some parts like the frontal and temporal lobes being larger, some like the parietal and occipital lobes being normal sized, and some like cerebellar vermis, corpus callosum, and basal ganglia being smaller than neurotypical individuals.[18]
- Cell adhesion molecules that are essential to formation and maintenance of connections between neurons, neuroligins found on postsynaptic neurons that bind presynaptic cell adhesion molecules, and proteins that anchor cell adhesion molecules to neurons are all found to be mutated in ASD.[18]
- Loss of function (LoF) mutations in genes relating to the function and development of the synapse.[19] Some of those implicated include SHANK3, SCN2A, and PTEN.[19]
Brain growth
Neuroanatomical studies and the association between autism and teratogens strongly suggest that autism affects brain development soon after conception.[12] This anomaly appears to start a cascade of pathological events in the brain that are significantly influenced by environmental factors.[20] Just after birth, the brains of children with autism tend to grow faster than usual, followed by normal or relatively slower growth in childhood.[21] It is unknown whether early brain overgrowth occurs in all children with autism. It appears to be most prominent in the frontal and temporal lobes, which are associated with higher cognitive specializations such as social cognition, and language development.[22] Hypotheses for the cellular and molecular bases of pathological early overgrowth include an excess of neurons that causes local overconnectivity in key brain regions,[21] and disturbed neuronal migration during early gestation.[23][24]
Synapse dysfunction
Synapse and dendritic spine growth may be disrupted in autism due to impaired neurexin–neuroligin cell-adhesion signaling[25] or dysregulated synthesis of synaptic proteins.[26][27] Disrupted synaptic development may also contribute to epilepsy, which may explain why the two conditions are associated.[28]Studies have suggested that excitatory–inhibitory networks may be imbalanced in autism.[24]
Neurotransmitters such as serotonin, dopamine, and glutamate have been implicated in autism.[1] Fragile X, the most common genetic cause of autism, is linked to dysfunction of group I metabotropic glutamate receptors (mGluR), leading some to consider their potential role in autism.[29]
Altered circuit connectivity
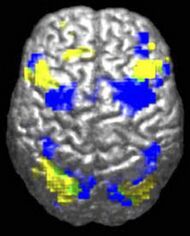
The underconnectivity theory of autism posits that autistic people tend to have fewer high-level neural connections and less global synchronization, along with an excess of low-level processes.[31] Functional connectivity studies have found both hypo- and hyperconnectivity in brains of autistic people.[32] Hypoconnectivity is commonly observed for interhemispheric (e.g. lower neuron density in corpus callosum)[33] and cortico-cortical functional connectivity.[34] Some studies have found local overconnectivity in the cerebral cortex and weak functional connections between the frontal lobe and the rest of the cortex.[35] Abnormal default mode network (task-negative) connectivity is often observed. Toggling between task-negative network activation and task-positive network activation (consisting of the dorsal attention network and salience network) may be less efficient, possibly reflecting a disturbance of self-referential thought.[36] Such patterns of low function and aberrant activation in the brain may depend on whether the brain is performing social or nonsocial tasks.[37]
Some studies have suggested that autism is a disorder of the association cortex.[38] Event-related potentials with respect to attention, orientation to auditory and visual stimuli, novelty detection, language and face processing, and information storage are altered in autistic individuals; several studies have found a preference for nonsocial stimuli.[39] Magnetoencephalography studies have observed delayed processing of auditory signals in autistic children.[40]
The mirror neuron system (MNS) theory of autism hypothesizes that disrupted development of the MNS impairs autistic people's ability to imitate others, leading to core autistic features of social impairment and communication difficulties. In animals, the MNS activates when an animal performs an action or observes another animal perform the same action. The MNS may contribute to an individual's understanding of other people by enabling the modeling of their behavior via embodied simulation of their actions, intentions, and emotions.[41][42] Several studies have tested this hypothesis by demonstrating structural abnormalities in MNS regions of individuals with ASD, delay in the activation in the core circuit for imitation in individuals with ASD, and a correlation between reduced MNS activity and severity of the syndrome in children with ASD.[43] However, individuals with autism also have abnormal brain activation in many circuits outside the MNS[44] and the MNS theory does not explain the normal performance of children with autism on imitation tasks that involve a goal or object.[45]
Common copy number variation associations have suggested similarities between the mechanisms of autism and schizophrenia. For loci such as 16p11.2, 16p13.1, 22p11, and 22q13, deletion is associated with autism whereas duplication is associated with schizophrenia. Conversely, 1q21.1 and 22p11.2 duplication is associated with autism and deletion with schizophrenia.[46]
It has been observed that people with ASD tend to have preferential processing of information on the left hemisphere compared to the right. The left hemisphere is associated with processing information related to details whereas the right hemisphere is associated with processing information in a more global and integrated sense that is essential for pattern recognition. For example, visual information like face recognition is normally processed by the right hemisphere which tends to integrate all information from an incoming sensory signal, whereas an ASD brain preferentially processes visual information in the left hemisphere where information tends to be processed for local details of the face rather than the overall configuration of the face. This left lateralization negatively impacts both facial recognition and spatial skills.[33][47]
Inflammation
The immune system is thought to play an important role in autism. Children with autism have been found by researchers to have inflammation of both the peripheral and central immune systems as indicated by increased levels of pro-inflammatory cytokines and significant activation of microglia.[48][49][7] Biomarkers of abnormal immune function have also been associated with increased impairments in behaviors that are characteristic of the core features of autism such as, deficits in social interactions and communication.[49] Interactions between the immune system and the nervous system begin early during the embryonic stage of life, and successful neurodevelopment depends on a balanced immune response. It is thought that activation of a pregnant mother's immune system such as from environmental toxicants or infection can contribute to causing autism through causing a disruption of brain development.[50][51][52] This is supported by recent studies that have found that infection during pregnancy is associated with an increased risk of autism.[53][54]
Some evidence suggests that gut–brain axis abnormalities may be involved by means of impaired serotonin signaling and inflammation.[6] A 2015 review proposed that immune dysregulation, gastrointestinal inflammation, autonomic nervous system malfunction, gut microbiota alterations, and food metabolites may cause brain neuroinflammation and dysfunction.[4] A 2016 review concluded that enteric nervous system abnormalities might play a role in neurological disorders such as autism.[5]
Metabolism
Some data suggests neuronal overgrowth observed in autism may be caused by an increase in several growth hormones[55] or impaired regulation of growth factor receptors. Some inborn errors of metabolism are associated with autism, but probably account for less than 5% of cases,[56] although the precedence of this varies internationally with levels of consanguinity[57] and it is thought to present its own endophenotype. Medical evidence has shown that autistic patients often present with abnormalities in glutathione-dependant redox metabolism,[58] mitochondrial disorders, glucose 6 phosphate deficiency, Phenylketonuria and Propionic Acidemia among other metabolic abnormalities.[57][59] Commonalities between both the genetic and epigenetic factors causing these metabolic disorders, and autism, are considered to be a potential reason for the high levels of comorbid presentation of autism in patients with a metabolic disorder. Many of the metabolic disorders implicated in autism also share a high degree of comorbidity to learning disabilities and other behavioural and neuropsychiatric concerns, most notably, catatonia (see also, catatonic autism), psychosis (see also, schizophrenia and autism) and depression.[57]
Brain connectivity
Brains of autistic individuals have been observed to have abnormal connectivity and the degree of these abnormalities directly correlates with the severity of autism. Following are some observed abnormal connectivity patterns in autistic individuals:[33][18]
- Decreased connectivity between different specialized regions of the brain (e.g. lower neuron density in corpus callosum) and relative over-connectivity within specialized regions of the brain by adulthood. Connectivity between different regions of the brain ('long-range' connectivity) is important for integration and global processing of information and comparing incoming sensory information with the existing model of the world within the brain. Connections within each specialized regions ('short-range' connections) are important for processing individual details and modifying the existing model of the world within the brain to more closely reflect incoming sensory information. In infancy, children at high risk for autism that were later diagnosed with autism were observed to have abnormally high long-range connectivity which then decreased through childhood to eventual long-range under-connectivity by adulthood.[33]
- Abnormal preferential processing of information by the left hemisphere of the brain vs. preferential processing of information by right hemisphere in neurotypical individuals. The left hemisphere is associated with processing information related to details whereas the right hemisphere is associated with processing information in a more global and integrated sense that is essential for pattern recognition. For example, visual information like face recognition is normally processed by the right hemisphere which tends to integrate all information from an incoming sensory signal, whereas an ASD brain preferentially processes visual information in the left hemisphere where information tends to be processed for local details of the face rather than the overall configuration of the face. This left lateralization negatively impacts both facial recognition and spatial skills.[33][60]
- Increased functional connectivity within the left hemisphere which directly correlates with severity of autism. This observation also supports preferential processing of details of individual components of sensory information over global processing of sensory information in an ASD brain.[33]
- Prominent abnormal connectivity in the frontal and occipital regions. In autistic individuals low connectivity in the frontal cortex was observed from infancy through adulthood. This is in contrast to long-range connectivity which is high in infancy and low in adulthood in ASD.[33] Abnormal neural organization is also observed in the Broca's area which is important for speech production.[18]
Gut-immune-brain axis
46% to 84% of autistic individuals have gastrointestinal-related problems like reflux, diarrhea, constipation, inflammatory bowel disease, and food allergies.[61] It has been observed that the makeup of gut bacteria in autistic people is different than that of non-autistic individuals which has raised the question of influence of gut bacteria on ASD development via inducing an inflammatory state.[62] Listed below are some research findings on the influence of gut bacteria and abnormal immune responses on brain development:[62]
- Some studies on rodents have shown gut bacteria influencing emotional functions and neurotransmitter balance in the brain, both of which are impacted in ASD.[18]
- The immune system is thought to be the intermediary that modulates the influence of gut bacteria on the brain. Some ASD individuals have a dysfunctional immune system with higher numbers of some types of immune cells, biochemical messengers and modulators, and autoimmune antibodies. Increased inflammatory biomarkers correlate with increased severity of ASD symptoms and there is some evidence to support a state of chronic brain inflammation in ASD.[62]
- More pronounced inflammatory responses to bacteria were found in ASD individuals with an abnormal gut microbiota. Additionally, immunoglobulin A antibodies that are central to gut immunity were also found in elevated levels in ASD populations. Some of these antibodies may attack proteins that support myelination of the brain, a process that is important for robust transmission of neural signal in many nerves.[62]
- Activation of the maternal immune system during pregnancy (by gut bacteria, bacterial toxins, an infection, or non-infectious causes) and gut bacteria in the mother that induce increased levels of Th17, a pro-inflammatory immune cell, have been associated with an increased risk of autism. Some maternal IgG antibodies that cross the placenta to provide passive immunity to the fetus can also attack the fetal brain.[62]
- It is proposed that inflammation within the brain promoted by inflammatory responses to harmful gut microbiome impacts brain development.[62]
- Pro-inflammatory cytokines IFN-γ, IFN-α, TNF-α, IL-6 and IL-17 have been shown to promote autistic behaviors in animal models. Giving anti-IL-6 and anti-IL-17 along with IL-6 and IL-17, respectively, have been shown to negate this effect in the same animal models.[62]
- Some gut proteins and microbial products can cross the blood–brain barrier and activate mast cells in the brain. Mast cells release pro-inflammatory factors and histamine which further increase blood–brain barrier permeability and help set up a cycle of chronic inflammation.[62]
Social brain interconnectivity
A number of discrete brain regions and networks among regions that are involved in dealing with other people have been discussed together under the rubric of the social brain. As of 2012[update], there is a consensus that autism spectrum is likely related to problems with interconnectivity among these regions and networks, rather than problems with any specific region or network.[63]
Temporal lobe
Functions of the temporal lobe are related to many of the deficits observed in individuals with ASDs, such as receptive language, social cognition, joint attention, action observation, and empathy. The temporal lobe also contains the superior temporal sulcus and the fusiform face area, which may mediate facial processing. It has been argued that dysfunction in the superior temporal sulcus underlies the social deficits that characterize autism. Compared to neurotypical individuals, one study found that individuals with high-functioning autism had reduced activity in the fusiform face area when viewing pictures of faces.[64]
Mitochondria
ASD could be linked to mitochondrial disease, a basic cellular abnormality with the potential to cause disturbances in a wide range of body systems.[65] A 2012 meta-analysis study, as well as other population studies show that approximately 5% of autistic children meet the criteria for classical mitochondrial dysfunction.[66] It is unclear why this mitochondrial disease occurs, considering that only 23% of children with both ASD and mitochondrial disease present with mitochondrial DNA abnormalities.[66]
Serotonin
Serotonin is a major neurotransmitter in the nervous system and contributes to formation of new neurons (neurogenesis), formation of new connections between neurons (synaptogenesis), remodelling of synapses, and survival and migration of neurons, processes that are necessary for a developing brain and some also necessary for learning in the adult brain. 45% of ASD individuals have been found to have increased blood serotonin levels.[18] Abnormalities in the serotonin transporter have also been found in ASD individuals. It has been hypothesized that increased activity of serotonin in the developing brain may facilitate the onset of ASD, with an association found in six out of eight studies between the use of selective serotonin reuptake inhibitors (SSRIs) by the pregnant mother and the development of ASD in the child exposed to SSRI in the antenatal environment.[67]
The study could not definitively conclude SSRIs caused the increased risk for ASD due to the biases found in those studies, and the authors called for more definitive, better conducted studies.[68] Confounding by indication has since then been shown to be likely.[69] However, it is also hypothesized that SSRIs may help reduce symptoms of ASD and even positively affect brain development in some ASD patients.[18]
Reduced NMDA‐receptor function
Reduced NMDA receptor function has been linked to reduced social interactions, locomotor hyperactivity, self-injury, prepulse inhibition (PPI) deficits, and sensory hypersensitivity, among others. Results suggest that NMDA dysregulation could contribute to core ASD symptoms.[70]
Abnormal folate metabolism
Several lines of evidence indicate abnormalities of folate metabolism in ASD. These abnormalities can lead to a decrease in 5-methyltetrahydrofolate production, alter the production of folate metabolites and reduce folate transport across the blood-brain barrier and in neurons.[71] The most significant abnormalities of folate metabolism associated with ASD may be autoantibodies to the alpha folate receptor (FRα). These autoantibodies have been associated with cerebral folate deficiency. Autoantibodies can bind to FRα and greatly impair its function.[72][73]
In 2013, one study reported that 60% and 44% of 93 children with ASD were positive for FRα-blocking and binding autoantibodies, respectively. This high rate of anti-FRα autoantibody positivity was confirmed by Ramaekers et al. who compared 75 children with ASD to 30 non-autistic "controls". These controls were children who had a developmental delay, but did not have ASD. FRα-blocking autoantibodies were positive in 47% of children with ASD, but only in 3% of children without ASD.[74] The authors of this study (RE Frye, JM Sequeira, EV Quadros, SJ James and DA Rossignol) have contributed to other such studies on FRα autoantibodies with similar results, [75]however are considered to have a conflict of interest by the publishers.[76]
Many children with ASD and cerebral folate deficiency have marked improvements in their clinical status when taking folinic acid.[72]
Five children with cerebral folate deficiency and low functioning autism with neurological deficits found a complete reduction of ASD symptoms with the use of folinic acid in a child and substantial improvements in communication in two other children.[77][78][79]
Abnormal redox metabolism
An imbalance in glutathione-dependent redox metabolism has been shown to be associated with autism spectrum disorder (ASD). [58]Glutathione synthesis and intracellular redox balance are related to folate metabolism and methylation, metabolic pathways that have also been shown to be abnormal in ASD. Together, these metabolic abnormalities define a distinct endophenotype of TSA closely associated with genetic, epigenetic and mitochondrial abnormalities, as well as environmental factors related to ASD. [58]Glutathione is involved in neuroprotection against oxidative stress and neuroinflammation by improving the antioxidant stress system.
In autistic children, studies have shown that glutathione metabolism can be improved[80]
- Subcutaneously by injection of methylcobalamin, a form of B12[81]
- Oral folinic acid[72]
- A vitamin and mineral supplement that includes antioxidants, coenzyme Q10 [81] and vitamins B
- Tetrahydrobiopterin[82]
Interestingly, recent DBPC studies have shown that N-acetyl-1-cysteine, a glutathione precursor supplement, is effective in improving the symptoms and behaviours associated with ASD.[83] However, glutathione was not measured in these studies.
Small, medium and large DPBC trials and open small and medium-sized clinical trials demonstrate that new treatments for children with ASD for oxidative stress are associated with improvements in baseline symptoms of ASD, sleep, gastrointestinal symptoms, hyperactivity, seizures and parental impression, sensory and motor symptoms. These new treatments include N-acetyl-l-cysteine, methylcobalamin with and without oral folinic acid, vitamin C, and a vitamin and mineral supplement that includes antioxidants, enzyme Q10, and B vitamins.
Several other treatments that have antioxidant properties, including carnosine, have also been reported to significantly improve ASD behaviours, suggesting that treatment of oxidative stress could be beneficial for children with ASD. Many antioxidants can also help improve mitochondrial function, suggesting that clinical improvements with antioxidants could occur through a reduction in oxidative stress and an improvement in mitochondrial function.
Some of these treatments can have frequent serious side effects such as bronchospasm.[77][84][85]
Neuropsychology
Two major types of cognitive theories have been proposed to explain links between autistic brains and behaviour.
Social cognition theories
Social cognition theories focuses on deficits in social cognition. Simon Baron-Cohen's empathizing–systemizing theory postulates that autistic individuals can systemize, by developing internal rules of operation to handle events inside the brain, but are less effective at empathizing, by handling events generated by other agents. An extension, the extreme male brain theory, hypothesizes that autism is an extreme case of the male brain, defined psychometrically as individuals where systemizing is better than empathizing.[86] These theories are somewhat related to Baron-Cohen's earlier theory of mind approach, which hypothesizes that autistic behaviour arises from an inability to ascribe mental states to oneself and others. The theory of mind hypothesis is supported by the atypical responses of children with autism to the Sally–Anne test for reasoning about others' motivations,[86] and the mirror neuron system theory of autism described in Pathophysiology maps well to the hypothesis.[43] However, most studies have found no evidence of impairment in autistic individuals' ability to understand other people's basic intentions or goals; instead, data suggests that impairments are found in understanding more complex social emotions or in considering others' viewpoints.[87]
Nonsocial cognition theories
Nonsocial cognition theories focuses on nonsocial or general processing: the executive functions such as working memory, planning, and inhibition. In his review, Kenworthy states that "the claim of executive dysfunction as a causal factor in autism is controversial", however, "it is clear that executive dysfunction plays a role in the social and cognitive deficits observed in individuals with autism".[88] Tests of core executive processes such as eye movement tasks indicate improvement from late childhood to adolescence, but performance never reaches typical adult levels.[89] A strength of this theory is predicting stereotyped behavior and narrow interests;[90] two weaknesses are that executive function is hard to measure[88] and that executive function deficits have not been found in young children with autism.[91]
Weak central coherence theory
Weak central coherence theory hypothesizes that a limited ability to see the big picture underlies the central disturbance in autism. One strength of this theory is predicting special talents and peaks in performance in autistic people.[92] Another theory, enhanced perceptual functioning, focuses more on the superiority of locally oriented and perceptual operations in autistic individuals.[93] Yet another theory, monotropism, posits that autism stems from a different cognitive style, tending to focus attention and processing resources intensely, to the exclusion of other stimuli.[94] These theories map well from the underconnectivity theory of autism.
Issues with theories
No single type of theory is satisfactory on its own; social cognition theories poorly address autism's rigid and repetitive behaviors, while most of the nonsocial theories have difficulty explaining autism's social impairment and communication difficulties.[95] A combined theory based on multiple deficits may prove to be more useful.[96]
See also
References
- ↑ 1.0 1.1 "Autism". Lancet 374 (9701): 1627–1638. November 2009. doi:10.1016/S0140-6736(09)61376-3. PMID 19819542.
- ↑ 2.0 2.1 "The emerging role of synaptic cell-adhesion pathways in the pathogenesis of autism spectrum disorders". Trends in Neurosciences 32 (7): 402–412. July 2009. doi:10.1016/j.tins.2009.04.003. PMID 19541375.
- ↑ "Autism and brain development". Cell 135 (3): 396–400. October 2008. doi:10.1016/j.cell.2008.10.015. PMID 18984148.
- ↑ 4.0 4.1 4.2 "Gastrointestinal symptoms and autism spectrum disorder: links and risks - a possible new overlap syndrome". Pediatric Health, Medicine and Therapeutics 6: 153–166. 2015. doi:10.2147/PHMT.S85717. PMID 29388597.
- ↑ 5.0 5.1 5.2 5.3 "The bowel and beyond: the enteric nervous system in neurological disorders". Nature Reviews. Gastroenterology & Hepatology 13 (9): 517–528. September 2016. doi:10.1038/nrgastro.2016.107. PMID 27435372.
- ↑ 6.0 6.1 6.2 "Serotonin as a link between the gut-brain-microbiome axis in autism spectrum disorders". Pharmacological Research 132: 1–6. June 2018. doi:10.1016/j.phrs.2018.03.020. PMID 29614380.
- ↑ 7.0 7.1 "Evidence linking oxidative stress, mitochondrial dysfunction, and inflammation in the brain of individuals with autism". Frontiers in Physiology 5: 150. 2014. doi:10.3389/fphys.2014.00150. PMID 24795645.
- ↑ "A Unifying Theory for Autism: The Pathogenetic Triad as a Theoretical Framework". Frontiers in Psychiatry 12. November 2021. doi:10.3389/fpsyt.2021.767075. PMID 34867553.
- ↑ 9.0 9.1 "Neurobiological correlates of autism: a review of recent research". Child Neuropsychology 12 (1): 57–79. February 2006. doi:10.1080/09297040500253546. PMID 16484102.
- ↑ 10.0 10.1 "The role of the neurobiologist in redefining the diagnosis of autism". Brain Pathology 17 (4): 408–411. October 2007. doi:10.1111/j.1750-3639.2007.00103.x. PMID 17919126.
- ↑ "Diagnosis of autism". BMJ 327 (7413): 488–493. August 2003. doi:10.1136/bmj.327.7413.488. PMID 12946972.
- ↑ 12.0 12.1 "The teratology of autism". International Journal of Developmental Neuroscience 23 (2–3): 189–199. 2005. doi:10.1016/j.ijdevneu.2004.11.001. PMID 15749245.
- ↑ "Neurobiology and Genetics of Autism: A Developmental Perspective". The development of autism: perspectives from theory and research. Mahwah, N.J.: L. Erlbaum. 2001. pp. 73–92. ISBN 978-0-8058-3245-7. OCLC 806185029.
- ↑ "Maternal immune activation: Implications for neuropsychiatric disorders". Science 353 (6301): 772–777. August 2016. doi:10.1126/science.aag3194. PMID 27540164. Bibcode: 2016Sci...353..772E.
- ↑ 15.0 15.1 "Neuroanatomy of autism". Trends in Neurosciences 31 (3): 137–145. March 2008. doi:10.1016/j.tins.2007.12.005. PMID 18258309.
- ↑ "Autism: many genes, common pathways?". Cell 135 (3): 391–395. October 2008. doi:10.1016/j.cell.2008.10.016. PMID 18984147.
- ↑ "The study of autism as a distributed disorder". Mental Retardation and Developmental Disabilities Research Reviews 13 (1): 85–95. 2007. doi:10.1002/mrdd.20141. PMID 17326118.
- ↑ 18.00 18.01 18.02 18.03 18.04 18.05 18.06 18.07 18.08 18.09 "The emerging picture of autism spectrum disorder: genetics and pathology". Annual Review of Pathology 10: 111–144. 2015. doi:10.1146/annurev-pathol-012414-040405. PMID 25621659.
- ↑ 19.0 19.1 "Synaptic, transcriptional and chromatin genes disrupted in autism". Nature 515 (7526): 209–215. November 2014. doi:10.1038/nature13772. PMID 25363760. Bibcode: 2014Natur.515..209..
- ↑ "The neuropathology of autism". Brain Pathology 17 (4): 422–433. October 2007. doi:10.1111/j.1750-3639.2007.00100.x. PMID 17919128.
- ↑ 21.0 21.1 "Mapping early brain development in autism". Neuron 56 (2): 399–413. October 2007. doi:10.1016/j.neuron.2007.10.016. PMID 17964254.
- ↑ "Advances in autism". Annual Review of Medicine 60: 367–380. 2009. doi:10.1146/annurev.med.60.053107.121225. PMID 19630577.
- ↑ "The neuropathology of autism: where do we stand?". Neuropathology and Applied Neurobiology 34 (1): 4–11. February 2008. doi:10.1111/j.1365-2990.2007.00872.x. PMID 17971078.
- ↑ 24.0 24.1 "Searching for ways out of the autism maze: genetic, epigenetic and environmental clues". Trends in Neurosciences 29 (7): 349–358. July 2006. doi:10.1016/j.tins.2006.05.010. PMID 16808981.
- ↑ "Neuroligins and neurexins link synaptic function to cognitive disease". Nature 455 (7215): 903–911. October 2008. doi:10.1038/nature07456. PMID 18923512. Bibcode: 2008Natur.455..903S.
- ↑ "The autistic neuron: troubled translation?". Cell 135 (3): 401–406. October 2008. doi:10.1016/j.cell.2008.10.017. PMID 18984149.
- ↑ "Fragile X: translation in action". Neuropsychopharmacology 33 (1): 84–87. January 2008. doi:10.1038/sj.npp.1301610. PMID 17940551.
- ↑ "Convulsing toward the pathophysiology of autism". Brain & Development 31 (2): 95–103. February 2009. doi:10.1016/j.braindev.2008.09.009. PMID 19006654.
- ↑ "Correction of fragile X syndrome in mice". Neuron 56 (6): 955–962. December 2007. doi:10.1016/j.neuron.2007.12.001. PMID 18093519.
- ↑ "Opening a window to the autistic brain". PLOS Biology 2 (8). August 2004. doi:10.1371/journal.pbio.0020267. PMID 15314667.
- ↑ "Functional and anatomical cortical underconnectivity in autism: evidence from an FMRI study of an executive function task and corpus callosum morphometry". Cerebral Cortex 17 (4): 951–961. April 2007. doi:10.1093/cercor/bhl006. PMID 16772313.
- ↑ "Neuropsychologic functioning in children with autism: further evidence for disordered complex information-processing". Child Neuropsychology 12 (4–5): 279–298. August 2006. doi:10.1080/09297040600681190. PMID 16911973.
- ↑ 33.0 33.1 33.2 33.3 33.4 33.5 33.6 "Is functional brain connectivity atypical in autism? A systematic review of EEG and MEG studies". PLOS ONE 12 (5). 2017. doi:10.1371/journal.pone.0175870. PMID 28467487. Bibcode: 2017PLoSO..1275870O.
- ↑ "Characteristics of Brains in Autism Spectrum Disorder: Structure, Function and Connectivity across the Lifespan". Experimental Neurobiology 24 (4): 273–284. December 2015. doi:10.5607/en.2015.24.4.273. PMID 26713076.
- ↑ "Resting state cortical connectivity reflected in EEG coherence in individuals with autism". Biological Psychiatry 62 (3): 270–273. August 2007. doi:10.1016/j.biopsych.2006.11.012. PMID 17336944.
- ↑ "Default-mode brain dysfunction in mental disorders: a systematic review". Neuroscience and Biobehavioral Reviews 33 (3): 279–296. March 2009. doi:10.1016/j.neubiorev.2008.09.002. PMID 18824195.
- ↑ "Functional brain correlates of social and nonsocial processes in autism spectrum disorders: an activation likelihood estimation meta-analysis". Biological Psychiatry 65 (1): 63–74. January 2009. doi:10.1016/j.biopsych.2008.09.022. PMID 18996505.
- ↑ "The new neurobiology of autism: cortex, connectivity, and neuronal organization". Archives of Neurology 64 (7): 945–950. July 2007. doi:10.1001/archneur.64.7.945. PMID 17620483.
- ↑ "Event related potentials in the understanding of autism spectrum disorders: an analytical review". Journal of Autism and Developmental Disorders 39 (3): 495–510. March 2009. doi:10.1007/s10803-008-0652-9. PMID 18850262.
- ↑ "Electrophysiological signatures: magnetoencephalographic studies of the neural correlates of language impairment in autism spectrum disorders". International Journal of Psychophysiology 68 (2): 149–160. May 2008. doi:10.1016/j.ijpsycho.2008.01.012. PMID 18336941.
- ↑ "Self-other relations in social development and autism: multiple roles for mirror neurons and other brain bases". Autism Research 1 (2): 73–90. April 2008. doi:10.1002/aur.15. PMID 19360654.
- ↑ "A mirror up to nature". Current Biology 18 (1): R13–R18. January 2008. doi:10.1016/j.cub.2007.11.004. PMID 18177704. Bibcode: 2008CBio...18..R13D.
- ↑ 43.0 43.1 "The mirror neuron system and the consequences of its dysfunction". Nature Reviews. Neuroscience 7 (12): 942–951. December 2006. doi:10.1038/nrn2024. PMID 17115076.
- ↑ "Development and neurophysiology of mentalizing". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences 358 (1431): 459–473. March 2003. doi:10.1098/rstb.2002.1218. PMID 12689373.
- ↑ "Emulation and mimicry for social interaction: a theoretical approach to imitation in autism". Quarterly Journal of Experimental Psychology 61 (1): 101–115. January 2008. doi:10.1080/17470210701508798. PMID 18038342.
- ↑ "Evolution in health and medicine Sackler colloquium: Comparative genomics of autism and schizophrenia". Proceedings of the National Academy of Sciences of the United States of America 107 (Suppl 1): 1736–1741. January 2010. doi:10.1073/pnas.0906080106. PMID 19955444. Bibcode: 2010PNAS..107.1736C.
- ↑ "Autism spectrum disorder - Symptoms and causes" (in en). https://www.mayoclinic.org/diseases-conditions/autism-spectrum-disorder/symptoms-causes/syc-20352928.
- ↑ "Immune Dysregulation in Autism Spectrum Disorder". Neurobiology of Autism. International Review of Neurobiology. 113. 2013. pp. 269–302. doi:10.1016/B978-0-12-418700-9.00009-5. ISBN 978-0-12-418700-9.
- ↑ 49.0 49.1 "The role of immune dysfunction in the pathophysiology of autism". Brain, Behavior, and Immunity 26 (3): 383–392. March 2012. doi:10.1016/j.bbi.2011.08.007. PMID 21906670.
- ↑ "Maternal infection and immune involvement in autism". Trends in Molecular Medicine 17 (7): 389–394. July 2011. doi:10.1016/j.molmed.2011.03.001. PMID 21482187.
- ↑ "Autism risk factors: genes, environment, and gene-environment interactions". Dialogues in Clinical Neuroscience 14 (3): 281–292. September 2012. doi:10.31887/DCNS.2012.14.3/pchaste. PMID 23226953.
- ↑ "The immune response in autism: a new frontier for autism research". Journal of Leukocyte Biology 80 (1): 1–15. July 2006. doi:10.1189/jlb.1205707. PMID 16698940. http://www.jleukbio.org/cgi/content/full/80/1/1.
- ↑ "Maternal hospitalization with infection during pregnancy and risk of autism spectrum disorders". Brain, Behavior, and Immunity 44: 100–105. February 2015. doi:10.1016/j.bbi.2014.09.001. PMID 25218900.
- ↑ "Maternal infection requiring hospitalization during pregnancy and autism spectrum disorders". Journal of Autism and Developmental Disorders 40 (12): 1423–1430. December 2010. doi:10.1007/s10803-010-1006-y. PMID 20414802.
- ↑ "Update on autism: a review of 1300 reports published in 2008". Epilepsy & Behavior 16 (4): 569–589. December 2009. doi:10.1016/j.yebeh.2009.09.023. PMID 19896907.
- ↑ "Autism and metabolic diseases". Journal of Child Neurology 23 (3): 307–314. March 2008. doi:10.1177/0883073807308698. PMID 18079313.
- ↑ 57.0 57.1 57.2 Ghaziuddin, Mohammad; Al-Owain, Mohammed (2013-10-01). "Autism Spectrum Disorders and Inborn Errors of Metabolism: An Update". Pediatric Neurology 49 (4): 232–236. doi:10.1016/j.pediatrneurol.2013.05.013. ISSN 0887-8994. https://www.sciencedirect.com/science/article/pii/S0887899413003433.
- ↑ 58.0 58.1 58.2 Frye, R. E.; James, S. J. (2014). "Metabolic Pathology of Autism in Relation to Redox Metabolism". Biomarkers in medicine 8, 2014 (3). https://www.tandfonline.com/doi/full/10.2217/bmm.13.158?needAccess=true.
- ↑ Das, Undurti N. (2013-10-01). "Autism as a disorder of deficiency of brain-derived neurotrophic factor and altered metabolism of polyunsaturated fatty acids". Nutrition 29 (10): 1175–1185. doi:10.1016/j.nut.2013.01.012. ISSN 0899-9007. https://www.sciencedirect.com/science/article/pii/S0899900713000543.
- ↑ "Autism spectrum disorder - Symptoms and causes" (in en). https://www.mayoclinic.org/diseases-conditions/autism-spectrum-disorder/symptoms-causes/syc-20352928.
- ↑ "Autism medical comorbidities" (in English). World Journal of Clinical Pediatrics 10 (3): 15–28. May 2021. doi:10.5409/wjcp.v10.i3.15. PMID 33972922. "Gastrointestinal (GI) disorders are significantly more common in children with ASD; they occur in 46% to 84% of them.".
- ↑ 62.0 62.1 62.2 62.3 62.4 62.5 62.6 62.7 "A systematic review of gut-immune-brain mechanisms in Autism Spectrum Disorder". Developmental Psychobiology 61 (5): 752–771. July 2019. doi:10.1002/dev.21803. PMID 30523646.
- ↑ "The social brain in psychiatric and neurological disorders". Trends in Cognitive Sciences 16 (11): 559–572. November 2012. doi:10.1016/j.tics.2012.09.006. PMID 23047070.
- ↑ "Developmental deficits in social perception in autism: the role of the amygdala and fusiform face area". International Journal of Developmental Neuroscience 23 (2–3): 125–141. 2005. doi:10.1016/j.ijdevneu.2004.12.012. PMID 15749240.
- ↑ "Mitochondrial disease: a practical approach for primary care physicians". Pediatrics 120 (6): 1326–1333. December 2007. doi:10.1542/peds.2007-0391. PMID 18055683.
- ↑ 66.0 66.1 "Mitochondrial dysfunction in autism spectrum disorders: a systematic review and meta-analysis". Molecular Psychiatry 17 (3): 290–314. March 2012. doi:10.1038/mp.2010.136. PMID 21263444.
- ↑ "Autism" (in en). https://www.who.int/news-room/fact-sheets/detail/autism-spectrum-disorders.
- ↑ "Prenatal antidepressant exposure and the risk of autism spectrum disorders in children. Are we looking at the fall of Gods?". Journal of Affective Disorders 182: 132–137. August 2015. doi:10.1016/j.jad.2015.04.048. PMID 25985383.
- ↑ "Association of Antidepressant Use With Adverse Health Outcomes: A Systematic Umbrella Review". JAMA Psychiatry 76 (12): 1241–1255. December 2019. doi:10.1001/jamapsychiatry.2019.2859. PMID 31577342.
- ↑ "Mice with reduced NMDA receptor expression: more consistent with autism than schizophrenia?". Genes, Brain and Behavior 11 (6): 740–750. August 2012. doi:10.1111/j.1601-183X.2012.00816.x. PMID 22726567.
- ↑ Rossignol, Daniel; frye, Richard (2 November 2021). "Cerebral Folate Deficiency, Folate Receptor Alpha Autoantibodies and Leucovorin (Folinic Acid) Treatment in Autism Spectrum Disorders: A Systematic Review and Meta-Analysis". Journal of Personalised Medicine 11 (11). https://www.mdpi.com/2075-4426/11/11/1141.
- ↑ 72.0 72.1 72.2 Frye, Richard E; Slattery, John C; Quadros, Edward V (August 2017). "Folate Metabolism Abnormalities in Autism: Potential Biomarkers" (in en). Biomarkers in Medicine 11 (8): 687–699. doi:10.2217/bmm-2017-0109. ISSN 1752-0363. https://www.tandfonline.com/doi/full/10.2217/bmm-2017-0109.
- ↑ Quadros, Edward V.; Sequeira, Jeffrey M.; Brown, W. Ted; Mevs, Clifford; Marchi, Elaine; Flory, Michael; Jenkins, Edmund C.; Velinov, Milen T. et al. (May 2018). "Folate receptor autoantibodies are prevalent in children diagnosed with autism spectrum disorder, their normal siblings and parents" (in en). Autism Research 11 (5): 707–712. doi:10.1002/aur.1934. ISSN 1939-3792. https://onlinelibrary.wiley.com/doi/10.1002/aur.1934.
- ↑ Frye, R. E.; Sequeira, J. M.; Quadros, E. V.; James, S. J.; Rossignol, D. A. (March 2013). "Cerebral folate receptor autoantibodies in autism spectrum disorder" (in en). Molecular Psychiatry 18 (3): 369–381. doi:10.1038/mp.2011.175. ISSN 1476-5578. https://www.nature.com/articles/mp2011175.
- ↑ "Google Scholar". https://scholar.google.com/scholar?q=related:eQashocN0VsJ:scholar.google.com/&scioq=RE+Frye,+JM+Sequeira,+EV+Quadros,+SJ+James,+DA+Rossignol&hl=en&as_sdt=0,5&inst=18159524348574443978.
- ↑ Frye, R. E.; Slattery, J.; Delhey, L.; Furgerson, B.; Strickland, T.; Tippett, M.; Sailey, A.; Wynne, R. et al. (February 2018). "Folinic acid improves verbal communication in children with autism and language impairment: a randomized double-blind placebo-controlled trial" (in en). Molecular Psychiatry 23 (2): 247–256. doi:10.1038/mp.2016.168. ISSN 1476-5578. https://www.nature.com/articles/mp2016168.
- ↑ 77.0 77.1 "Treatments for biomedical abnormalities associated with autism spectrum disorder". Frontiers in Pediatrics 2: 66. 2014. doi:10.3389/fped.2014.00066. PMID 25019065.
- ↑ "Glutathione-related factors and oxidative stress in autism, a review". Current Medicinal Chemistry 19 (23): 4000–4005. 2012. doi:10.2174/092986712802002572. PMID 22708999.
- ↑ "NAC Side Effects: Common, Severe, Long Term". https://www.drugs.com/sfx/nac-side-effects.html.
- ↑ "Sage Journals: Discover world-class research" (in en). doi:10.3233/JAD-2006-9408. https://journals.sagepub.com/action/cookieAbsent.
- ↑ 81.0 81.1 Frye, Richard E.; Rossignol, Daniel A. (December 2012). "Treatments for mitochondrial dysfunction associated with autism spectrum disorders" (in en). Journal of Pediatric Biochemistry 02 (4): 241–249. doi:10.1055/s-0036-1586420. ISSN 1879-5390. http://www.thieme-connect.de/DOI/DOI?10.1055/s-0036-1586420.
- ↑ Thöny, B.; Auerbach, G.; Blau, N. (2000-04-01). "Tetrahydrobiopterin biosynthesis, regeneration and functions". The Biochemical Journal 347 Pt 1 (Pt 1): 1–16. ISSN 0264-6021. PMID 10727395. PMC 1220924. https://pubmed.ncbi.nlm.nih.gov/10727395.
- ↑ "Effectiveness of N-acetylcysteine in autism spectrum disorders: A meta-analysis of randomized controlled trials". The Australian and New Zealand Journal of Psychiatry 55 (2): 196–206. February 2021. doi:10.1177/0004867420952540. PMID 32900213.
- ↑ "Glutathione-related factors and oxidative stress in autism, a review". Current Medicinal Chemistry 19 (23): 4000–4005. 2012. doi:10.2174/092986712802002572. PMID 22708999.
- ↑ "NAC Side Effects: Common, Severe, Long Term". https://www.drugs.com/sfx/nac-side-effects.html.
- ↑ 86.0 86.1 "Autism: the empathizing-systemizing (E-S) theory". Annals of the New York Academy of Sciences 1156 (1): 68–80. March 2009. doi:10.1111/j.1749-6632.2009.04467.x. PMID 19338503. Bibcode: 2009NYASA1156...68B.
- ↑ "Goals, intentions and mental states: challenges for theories of autism". Journal of Child Psychology and Psychiatry, and Allied Disciplines 50 (8): 881–892. August 2009. doi:10.1111/j.1469-7610.2009.02098.x. PMID 19508497.
- ↑ 88.0 88.1 "Understanding executive control in autism spectrum disorders in the lab and in the real world". Neuropsychology Review 18 (4): 320–338. December 2008. doi:10.1007/s11065-008-9077-7. PMID 18956239.
- ↑ "Neurodevelopment and executive function in autism". Development and Psychopathology 20 (4): 1103–1132. 2008. doi:10.1017/S0954579408000527. PMID 18838033.
- ↑ "Executive dysfunction in autism". Trends in Cognitive Sciences 8 (1): 26–32. January 2004. doi:10.1016/j.dr.2004.01.001. PMID 14697400. https://research.gold.ac.uk/2560/1/hill_devrev04_GRO.pdf.
- ↑ "Autism from developmental and neuropsychological perspectives". Annual Review of Clinical Psychology 2: 327–355. 2006. doi:10.1146/annurev.clinpsy.2.022305.095210. PMID 17716073.
- ↑ "The weak coherence account: detail-focused cognitive style in autism spectrum disorders". Journal of Autism and Developmental Disorders 36 (1): 5–25. January 2006. doi:10.1007/s10803-005-0039-0. PMID 16450045.
- ↑ "Enhanced perceptual functioning in autism: an update, and eight principles of autistic perception". Journal of Autism and Developmental Disorders 36 (1): 27–43. January 2006. doi:10.1007/s10803-005-0040-7. PMID 16453071.
- ↑ "Attention, monotropism and the diagnostic criteria for autism". Autism 9 (2): 139–156. May 2005. doi:10.1177/1362361305051398. PMID 15857859. http://www.autismusundcomputer.de/english//139.pdf. Retrieved 18 March 2018.
- ↑ "Time to give up on a single explanation for autism". Nature Neuroscience 9 (10): 1218–1220. October 2006. doi:10.1038/nn1770. PMID 17001340.
- ↑ "Cognitive theories of autism". Dev Rev 27 (2): 224–60. 2007. doi:10.1016/j.dr.2007.02.001. https://strathprints.strath.ac.uk/5154/7/strathprints008124.pdf.
 |
