Medicine:Alzheimer's disease
| Alzheimer's disease | |
|---|---|
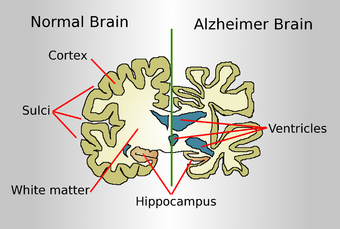 | |
| Drawing comparing a normal aged brain (left) and the brain of a person with Alzheimer's (right). Characteristics that separate the two are pointed out. | |
| Pronunciation |
|
| Specialty | Neurology |
| Symptoms | Difficulty in remembering recent events, problems with language, disorientation, mood swings[1][2] |
| Complications | Dehydration and Pneumonia in the terminal stage[3] |
| Usual onset | Over 65 years old[4] |
| Duration | Long term[2] |
| Causes | Poorly understood[1] |
| Risk factors | Genetics, head injuries, depression, hypertension[1][5] |
| Diagnostic method | Based on symptoms and cognitive testing after ruling out other possible causes[6] |
| Differential diagnosis | Normal aging[1] |
| Medication | Acetylcholinesterase inhibitors, NMDA receptor antagonists (small benefit),[7] |
| Prognosis | Life expectancy 3–9 years[8] |
| Frequency | 29.8 million (2015)[9] |
| Deaths | For all dementias 1.9 million (2015)[10] |
Alzheimer's disease (AD) is a neurodegenerative disease that usually starts slowly and progressively worsens.[2] It is the cause of 60–70% of cases of dementia.[2] The most common early symptom is difficulty in remembering recent events.[1] As the disease advances, symptoms can include problems with language, disorientation (including easily getting lost), mood swings, loss of motivation, self-neglect, and behavioral issues.[1][2] As a person's condition declines, they often withdraw from family and society.[1] Gradually, bodily functions are lost, ultimately leading to death.[11] Although the speed of progression can vary, the typical life expectancy following diagnosis is three to nine years.[8][12]
The cause of Alzheimer's disease is poorly understood.[1] There are many environmental and genetic risk factors associated with its development. The strongest genetic risk factor is from an allele of APOE.[13][14] Other risk factors include a history of head injury, clinical depression, and high blood pressure.[1] The disease process is largely associated with amyloid plaques, neurofibrillary tangles, and loss of neuronal connections in the brain.[11] A probable diagnosis is based on the history of the illness and cognitive testing with medical imaging and blood tests to rule out other possible causes.[6] Initial symptoms are often mistaken for normal aging.[1] Examination of brain tissue is needed for a definite diagnosis, but this can only take place after death.[11] Good nutrition, physical activity, and engaging socially are known to be of benefit generally in aging, and these may help in reducing the risk of cognitive decline and Alzheimer's; in 2019 clinical trials were underway to look at these possibilities.[11] There are no medications or supplements that have been shown to decrease risk.[15]
No treatments stop or reverse its progression, though some may temporarily improve symptoms.[2] Affected people increasingly rely on others for assistance, often placing a burden on the caregiver.[16] The pressures can include social, psychological, physical, and economic elements.[16] Exercise programs may be beneficial with respect to activities of daily living and can potentially improve outcomes.[17] Behavioral problems or psychosis due to dementia are often treated with antipsychotics, but this is not usually recommended, as there is little benefit and an increased risk of early death.[18][19]
As of 2015, there were approximately 29.8 million people worldwide with AD[9] with about 50 million of all forms of dementia as of 2020.[2] It most often begins in people over 65 years of age, although up to 10 per cent of cases are early-onset affecting those in their 30's to mid 60's.[11][4] Women get sick more often than men.[20] It affects about 6% of people 65 years and older.[1] In 2015, all forms of dementia resulted in about 1.9 million deaths.[10] The disease is named after German psychiatrist and pathologist Alois Alzheimer, who first described it in 1906.[21] Alzheimer's financial burden on society is large, on par with the costs of cancer and heart disease, costing 200 billion United States dollar in the US alone.[15][22]
Signs and symptoms
The course of Alzheimer's is generally described in three stages, with a progressive pattern of cognitive and functional impairment.[23][11] The three stages are described as early or mild, middle or moderate, and late or severe.[23][11] The disease is known to target the hippocampus which is associated with memory, and this is responsible for the first symptoms of memory impairment. As the disease progresses so does the degree of memory impairment.[11]
First symptoms

The first symptoms are often mistakenly attributed to aging or stress.[24] Detailed neuropsychological testing can reveal mild cognitive difficulties up to eight years before a person fulfills the clinical criteria for diagnosis of Alzheimer's disease.[25] These early symptoms can affect the most complex activities of daily living.[26] The most noticeable deficit is short term memory loss, which shows up as difficulty in remembering recently learned facts and inability to acquire new information.[25][27]
Subtle problems with the executive functions of attentiveness, planning, flexibility, and abstract thinking, or impairments in semantic memory (memory of meanings, and concept relationships) can also be symptomatic of the early stages of Alzheimer's disease.[25] Apathy and depression can be seen at this stage, with apathy remaining as the most persistent symptom throughout the course of the disease.[28][29] The preclinical stage of the disease has also been termed mild cognitive impairment (MCI).[27] This is often found to be a transitional stage between normal aging and dementia. MCI can present with a variety of symptoms, and when memory loss is the predominant symptom, it is termed amnestic MCI and is frequently seen as a prodromal stage of Alzheimer's disease.[30] Amnestic MCI has a greater than 90% likelihood of being associated with Alzheimer's.[31]
Early stage
In people with Alzheimer's disease, the increasing impairment of learning and memory eventually leads to a definitive diagnosis. In a small percentage, difficulties with language, executive functions, perception (agnosia), or execution of movements (apraxia) are more prominent than memory problems.[32] Alzheimer's disease does not affect all memory capacities equally. Older memories of the person's life (episodic memory), facts learned (semantic memory), and implicit memory (the memory of the body on how to do things, such as using a fork to eat or how to drink from a glass) are affected to a lesser degree than new facts or memories.[33][34]
Language problems are mainly characterised by a shrinking vocabulary and decreased word fluency, leading to a general impoverishment of oral and written language.[32][35] In this stage, the person with Alzheimer's is usually capable of communicating basic ideas adequately.[32][35][36] While performing fine motor tasks such as writing, drawing, or dressing, certain movement coordination and planning difficulties (apraxia) may be present, but they are commonly unnoticed.[32] As the disease progresses, people with Alzheimer's disease can often continue to perform many tasks independently, but may need assistance or supervision with the most cognitively demanding activities.[32]
Middle stage
Progressive deterioration eventually hinders independence, with subjects being unable to perform most common activities of daily living.[32] Speech difficulties become evident due to an inability to recall vocabulary, which leads to frequent incorrect word substitutions (paraphasias). Reading and writing skills are also progressively lost.[32][36] Complex motor sequences become less coordinated as time passes and Alzheimer's disease progresses, so the risk of falling increases.[32] During this phase, memory problems worsen, and the person may fail to recognise close relatives.[32] Long-term memory, which was previously intact, becomes impaired.[32]
Behavioral and neuropsychiatric changes become more prevalent. Common manifestations are wandering, irritability and emotional lability, leading to crying, outbursts of unpremeditated aggression, or resistance to caregiving.[32] Sundowning can also appear.[37] Approximately 30% of people with Alzheimer's disease develop illusionary misidentifications and other delusional symptoms.[32] Subjects also lose insight of their disease process and limitations (anosognosia).[32] Urinary incontinence can develop.[32] These symptoms create stress for relatives and carers, which can be reduced by moving the person from home care to other long-term care facilities.[32][38]
Late stage
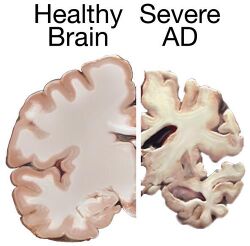
During the final stage, known as the late-stage or severe stage, the patient is completely dependent upon caregivers.[11][23][32] Language is reduced to simple phrases or even single words, eventually leading to complete loss of speech.[32][36] Despite the loss of verbal language abilities, people can often understand and return emotional signals. Although aggressiveness can still be present, extreme apathy and exhaustion are much more common symptoms. People with Alzheimer's disease will ultimately not be able to perform even the simplest tasks independently; muscle mass and mobility deteriorates to the point where they are bedridden and unable to feed themselves. The cause of death is usually an external factor, such as infection of pressure ulcers or pneumonia, not the disease itself.[32]
Causes
Alzheimer's disease is believed to occur when abnormal amounts of amyloid beta, accumulating extracellularly as amyloid plaques, and tau proteins, accumulating intracellularly as neurofibrillary tangles, form in the brain affecting neuronal functioning and connectivity, resulting in a progressive loss of brain function.[39][40] This altered protein clearance ability is age-related, regulated by brain cholesterol,[41] and associated with other neurodegenerative diseases.[42][43]
The cause for most Alzheimer's cases is still mostly unknown except for 1-2% of cases where deterministic genetic differences have been identified.[13] Several competing hypotheses exist trying to explain the cause of the disease.
Genetic
Only 1–2% of Alzheimer's cases are inherited (autosomal dominant). These types are known as early onset familial Alzheimer's disease, can have a very early onset, and a faster rate of progression.[13] Early onset familial Alzheimer's disease can be attributed to mutations in one of three genes: those encoding amyloid-beta precursor protein (APP) and presenilins PSEN1 and PSEN2.[31] Most mutations in the APP and presenilin genes increase the production of a small protein called amyloid beta (Aβ)42, which is the main component of amyloid plaques.[44] Some of the mutations merely alter the ratio between Aβ42 and the other major forms—particularly Aβ40—without increasing Aβ42 levels.[45] Two other genes associated with autosomal dominant Alzheimer's disease are ABCA7 and SORL1.[46]
Most cases of Alzheimer's are not inherited and are termed sporadic Alzheimer's disease, in which environmental and genetic differences may act as risk factors. Most cases of sporadic Alzheimer's disease in contrast to familial Alzheimer's disease are late-onset Alzheimer's disease (LOAD) developing after the age of 65 years. Less than 5% of sporadic Alzheimer's disease have an earlier onset.[13] The strongest genetic risk factor for sporadic Alzheimer's disease is APOEε4.[14] APOEε4 is one of four alleles of apolipoprotein E (APOE). APOE plays a major role in lipid-binding proteins in lipoprotein particles and the epsilon4 allele disrupts this function.[47] Between 40 and 80% of people with Alzheimer's disease possess at least one APOEε4 allele.[48] The APOEε4 allele increases the risk of the disease by three times in heterozygotes and by 15 times in homozygotes.[49] Like many human diseases, environmental effects and genetic modifiers result in incomplete penetrance. For example, certain Nigerian populations do not show the relationship between dose of APOEε4 and incidence or age-of-onset for Alzheimer's disease seen in other human populations.[50][51] Early attempts to screen up to 400 candidate genes for association with late-onset sporadic Alzheimer's disease (LOAD) resulted in a low yield.[49][52] More recent genome-wide association studies (GWAS) have found 19 areas in genes that appear to affect the risk.[53] These genes include: CASS4, CELF1, FERMT2, HLA-DRB5, INPP5D, MEF2C, NME8, PTK2B, SORL1, ZCWPW1, SLC24A4, CLU, PICALM, CR1, BIN1, MS4A, ABCA7, EPHA1, and CD2AP.[53]
Alleles in the TREM2 gene have been associated with a 3 to 5 times higher risk of developing Alzheimer's disease.[54][55] A suggested mechanism of action is that in some variants in TREM2, white blood cells in the brain are no longer able to control the amount of amyloid beta present. Many single-nucleotide polymorphisms (SNPs) are associated with Alzheimer's, with a 2018 study adding 30 SNPs by differentiating Alzheimer's disease into six categories, including memory, language, visuospatial, and executive functioning.[56]
Osaka mutation
A Japanese pedigree of familial Alzheimer's disease was found to be associated with a deletion mutation of codon 693 of APP.[57] This mutation and its association with Alzheimer's disease was first reported in 2008,[58] and is known as the Osaka mutation. Only homozygotes with this mutation develop Alzheimer's disease. This mutation accelerates Aβ oligomerization but the proteins do not form the amyloid fibrils that aggregate into amyloid plaques, suggesting that it is the Aβ oligomerization rather than the fibrils that may be the cause of this disease. Mice expressing this mutation have all the usual pathologies of Alzheimer's disease.[59]
Cholinergic hypothesis
The oldest hypothesis, on which most drug therapies are based, is the cholinergic hypothesis,[60] which proposes that Alzheimer's disease is caused by reduced synthesis of the neurotransmitter acetylcholine. The cholinergic hypothesis has not maintained widespread support, largely because medications intended to treat acetylcholine deficiency have not been very effective.[61]
Amyloid hypothesis
The 1991 amyloid hypothesis postulated that extracellular amyloid beta (Aβ) deposits are the fundamental cause of the disease.[62][63] Support for this postulate comes from the location of the gene for the amyloid precursor protein (APP) on chromosome 21, together with the fact that people with trisomy 21 (Down syndrome) who have an extra gene copy almost universally exhibit at least the earliest symptoms of Alzheimer's disease by 40 years of age.[64][65] Also, a specific isoform of apolipoprotein, APOE4, is a major genetic risk factor for Alzheimer's disease. While apolipoproteins enhance the breakdown of beta amyloid, some isoforms are not very effective at this task (such as APOE4), leading to excess amyloid buildup in the brain.[66]
Tau hypothesis

The tau hypothesis proposes that tau protein abnormalities initiate the disease cascade.[63] In this model, hyperphosphorylated tau begins to pair with other threads of tau as paired helical filaments. Eventually, they form neurofibrillary tangles inside nerve cell bodies.[67] When this occurs, the microtubules disintegrate, destroying the structure of the cell's cytoskeleton which collapses the neuron's transport system.[68] This may result first in malfunctions in biochemical communication between neurons and later in the death of the cells.[69]
Inflammatory hypothesis
A number of studies connect the misfolded amyloid beta and tau proteins associated with the pathology of Alzheimer's disease, as bringing about oxidative stress that leads to chronic inflammation.[70] Sustained inflammation (neuroinflammation) is also a feature of other neurodegenerative diseases including Parkinson's disease, and ALS.[71] Spirochete infections have also been linked to dementia.[72][73]
Sleep disturbances are seen as a possible risk factor for inflammation in Alzheimer's disease. Sleep problems have been seen as a consequence of Alzheimer's disease but studies suggest that they may instead be a causal factor. Sleep disturbances are thought to be linked to persistent inflammation.[74] A possible role of chronic periodontal infection[75] and the gut microbiota has been suggested.[76]
Other hypotheses
Cholesterol signaling hypothesis postulates that amyloid production and tau phosphorylation are regulated by cholesterol and high brain cholesterol contributes to the disease. First, the cholesterol is made in the astrocytes, the astrocytes load the cholesterol into the cholesterol carrier protein apoE, and the apoE loads the cholesterol into the neurons. Once in the neurons, cholesterol causes clustering of amyloid precursor protein (APP) with its hydrolytic enzyme gamma secretase, resulting in amyloid beta production and accumulation of amyloid plaques.[41] Cholesterol regulates amyloid beta production by substrate presentation.
A neurovascular hypothesis stating that poor functioning of the blood–brain barrier may be involved has been proposed.[77]
The cellular homeostasis of biometals such as ionic copper, iron, and zinc is disrupted in Alzheimer's disease, though it remains unclear whether this is produced by or causes the changes in proteins. These ions affect and are affected by tau, APP, and APOE,[78] and their dysregulation may cause oxidative stress that may contribute to the pathology.[79][80][81][82][83] The quality of some of these studies has been criticised,[84][85] and the link remains controversial.[86] The majority of researchers do not support a causal connection with aluminium.[85]
Smoking is a significant Alzheimer's disease risk factor.[87] Systemic markers of the innate immune system are risk factors for late-onset Alzheimer's disease.[88]
There is tentative evidence that exposure to air pollution may be a contributing factor to the development of Alzheimer's disease.[89][90]
One hypothesis posits that dysfunction of oligodendrocytes and their associated myelin during aging contributes to axon damage, which then causes amyloid production and tau hyper-phosphorylation as a side effect.[91][92]
Retrogenesis is a medical hypothesis about the development and progress of Alzheimer's disease proposed by Barry Reisberg in the 1980s.[93] The hypothesis is that just as the fetus goes through a process of neurodevelopment beginning with neurulation and ending with myelination, the brains of people with Alzheimer's disease go through a reverse neurodegeneration process starting with demyelination and death of axons (white matter) and ending with the death of grey matter.[94] Likewise the hypothesis is, that as infants go through states of cognitive development, people with Alzheimer's disease go through the reverse process of progressive cognitive impairment.[93] Reisberg developed the caregiving assessment tool known as "FAST" (Functional Assessment Staging Tool) which he says allows those caring for people with Alzheimer's disease to identify the stages of disease progression and that provides advice about the kind of care needed at each stage.[93][95]
The association with celiac disease is unclear, with a 2019 study finding no increase in dementia overall in those with CD, while a 2018 review found an association with several types of dementia including Alzheimer's disease.[96][97]
Kynurenines are a downstream metabolite of tryptophan and have the potential to be neuroactive. This may be associated with the neuropsychiatric symptoms and cognitive prognosis in mild dementia. A five-year study focused on the role of kynurenine in Alzheimer's and Lewy body disease and found its increase to be associated with more hallucinations.[98]
Pathophysiology

Neuropathology
Alzheimer's disease is characterised by loss of neurons and synapses in the cerebral cortex and certain subcortical regions. This loss results in gross atrophy of the affected regions, including degeneration in the temporal lobe and parietal lobe, and parts of the frontal cortex and cingulate gyrus.[99] Degeneration is also present in brainstem nuclei particularly the locus coeruleus in the pons.[100] Studies using MRI and PET have documented reductions in the size of specific brain regions in people with Alzheimer's disease as they progressed from mild cognitive impairment to Alzheimer's disease, and in comparison with similar images from healthy older adults.[101][102]
Both Aβ plaques and neurofibrillary tangles are clearly visible by microscopy in brains of those afflicted by Alzheimer's disease,[103] especially in the hippocampus.[104] However, Alzheimer's disease may occur without neurofibrillary tangles in the neocortex.[105] Plaques are dense, mostly insoluble deposits of beta-amyloid peptide and cellular material outside and around neurons. Tangles (neurofibrillary tangles) are aggregates of the microtubule-associated protein tau which has become hyperphosphorylated and accumulate inside the cells themselves. Although many older individuals develop some plaques and tangles as a consequence of aging, the brains of people with Alzheimer's disease have a greater number of them in specific brain regions such as the temporal lobe.[106] Lewy bodies are not rare in the brains of people with Alzheimer's disease.[107]
Biochemistry
Alzheimer's disease has been identified as a protein misfolding disease, a proteopathy, caused by the accumulation of abnormally folded amyloid beta protein into amyloid plaques, and tau protein into neurofibrillary tangles in the brain.[108] Plaques are made up of small peptides, 39–43 amino acids in length, called amyloid beta (Aβ). Amyloid beta is a fragment from the larger amyloid-beta precursor protein (APP) a transmembrane protein that penetrates the neuron's membrane. APP is critical to neuron growth, survival, and post-injury repair.[109][110] In Alzheimer's disease, gamma secretase and beta secretase act together in a proteolytic process which causes APP to be divided into smaller fragments.[111] One of these fragments gives rise to fibrils of amyloid beta, which then form clumps that deposit outside neurons in dense formations known as amyloid plaques.[103][112]
Alzheimer's disease is also considered a tauopathy due to abnormal aggregation of the tau protein. Every neuron has a cytoskeleton, an internal support structure partly made up of structures called microtubules. These microtubules act like tracks, guiding nutrients and molecules from the body of the cell to the ends of the axon and back. A protein called tau stabilises the microtubules when phosphorylated, and is therefore called a microtubule-associated protein. In Alzheimer's disease, tau undergoes chemical changes, becoming hyperphosphorylated; it then begins to pair with other threads, creating neurofibrillary tangles and disintegrating the neuron's transport system.[113] Pathogenic tau can also cause neuronal death through transposable element dysregulation.[114]
Disease mechanism
Exactly how disturbances of production and aggregation of the beta-amyloid peptide give rise to the pathology of Alzheimer's disease is not known.[115][116] The amyloid hypothesis traditionally points to the accumulation of beta-amyloid peptides as the central event triggering neuron degeneration. Accumulation of aggregated amyloid fibrils, which are believed to be the toxic form of the protein responsible for disrupting the cell's calcium ion homeostasis, induces programmed cell death (apoptosis).[117] It is also known that Aβ selectively builds up in the mitochondria in the cells of Alzheimer's-affected brains, and it also inhibits certain enzyme functions and the utilisation of glucose by neurons.[118]
Various inflammatory processes and cytokines may also have a role in the pathology of Alzheimer's disease. Inflammation is a general marker of tissue damage in any disease, and may be either secondary to tissue damage in Alzheimer's disease or a marker of an immunological response.[119] There is increasing evidence of a strong interaction between the neurons and the immunological mechanisms in the brain. Obesity and systemic inflammation may interfere with immunological processes which promote disease progression.[120]
Alterations in the distribution of different neurotrophic factors and in the expression of their receptors such as the brain-derived neurotrophic factor (BDNF) have been described in Alzheimer's disease.[121][122]
Diagnosis

Alzheimer's disease is usually diagnosed based on the person's medical history, history from relatives, and behavioral observations. The presence of characteristic neurological and neuropsychological features and the absence of alternative conditions is supportive.[123][124] Advanced medical imaging with computed tomography (CT) or magnetic resonance imaging (MRI), and with single-photon emission computed tomography (SPECT) or positron emission tomography (PET) can be used to help exclude other cerebral pathology or subtypes of dementia.[125] Moreover, it may predict conversion from prodromal stages (mild cognitive impairment) to Alzheimer's disease.[126] Approved radiopharmaceutical diagnostic agents used in PET for patients with Alzheimer's disease: florbetapir (2012), flutemetamol (2013), florbetaben (2014), flortaucipir (2020).[127]
Assessment of intellectual functioning including memory testing can further characterise the state of the disease.[24] Medical organizations have created diagnostic criteria to ease and standardise the diagnostic process for practising physicians. The diagnosis can be confirmed with very high accuracy post-mortem when brain material is available and can be examined histologically.[128]
Criteria
The National Institute of Neurological and Communicative Disorders and Stroke (NINCDS) and the Alzheimer's Disease and Related Disorders Association (ADRDA, now known as the Alzheimer's Association) established the most commonly used NINCDS-ADRDA Alzheimer's Criteria for diagnosis in 1984,[128] extensively updated in 2007.[129] These criteria require that the presence of cognitive impairment, and a suspected dementia syndrome, be confirmed by neuropsychological testing for a clinical diagnosis of possible or probable Alzheimer's disease. A histopathologic confirmation including a microscopic examination of brain tissue is required for a definitive diagnosis. Good statistical reliability and validity have been shown between the diagnostic criteria and definitive histopathological confirmation.[130] Eight intellectual domains are most commonly impaired in AD—memory, language, perceptual skills, attention, motor skills, orientation, problem solving and executive functional abilities. These domains are equivalent to the NINCDS-ADRDA Alzheimer's Criteria as listed in the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV-TR) published by the American Psychiatric Association.[131][132]
Techniques

Neuropsychological tests including cognitive tests such as the Mini–Mental State Examination (MMSE) are widely used to evaluate the cognitive impairments needed for diagnosis. More comprehensive test arrays are necessary for high reliability of results, particularly in the earliest stages of the disease.[133][134] Neurological examination in early Alzheimer's disease will usually provide normal results, except for obvious cognitive impairment, which may not differ from that resulting from other diseases processes, including other causes of dementia.
Further neurological examinations are crucial in the differential diagnosis of Alzheimer's disease and other diseases.[24] Interviews with family members are also utilised in the assessment of the disease. Caregivers can supply important information on the daily living abilities, as well as on the decrease, over time, of the person's mental function.[135] A caregiver's viewpoint is particularly important, since a person with Alzheimer's disease is commonly unaware of their deficits.[136] Many times, families also have difficulties in the detection of initial dementia symptoms and may not communicate accurate information to a physician.[137]
Supplemental testing provides extra information on some features of the disease or is used to rule out other diagnoses. Blood tests can identify other causes for dementia than AD[24]—causes which may, in rare cases, be reversible.[138] It is common to perform thyroid function tests, assess B12, rule out syphilis, rule out metabolic problems (including tests for kidney function, electrolyte levels and for diabetes), assess levels of heavy metals (e.g., lead, mercury) and anaemia. (It is also necessary to rule out delirium).
Psychological tests for depression are employed, since depression can either be concurrent with Alzheimer's disease (see Depression of Alzheimer disease), an early sign of cognitive impairment,[139] or even the cause.[140][141]
Due to low accuracy, the C-PIB-PET scan is not recommended to be used as an early diagnostic tool or for predicting the development of Alzheimer's disease when people show signs of mild cognitive impairment (MCI).[142] The use of 18F-FDG PET scans, as a single test, to identify people who may develop Alzheimer's disease is also not supported by evidence.[143]
Prevention

There is no evidence that supports any particular measure as being effective in preventing Alzheimer's disease.[15] Global studies of measures to prevent or delay the onset of Alzheimer's disease have often produced inconsistent results. Epidemiological studies have proposed relationships between certain modifiable factors, such as diet, cardiovascular risk, pharmaceutical products, or intellectual activities, among others, and a population's likelihood of developing Alzheimer's disease. Only further research, including clinical trials, will reveal whether these factors can help to prevent Alzheimer's disease.[15]
Medication
Cardiovascular risk factors, such as hypercholesterolaemia, hypertension, diabetes, and smoking, are associated with a higher risk of onset and worsened course of Alzheimer's disease.[144][145] Blood pressure medications may decrease the risk.[146] A review found that the use of statins, which lower cholesterol may be of benefit in Alzheimer's and other dementias but not in vascular dementia.[147]
Long-term usage of non-steroidal anti-inflammatory drugs (NSAIDs) were thought in 2007 to be associated with a reduced likelihood of developing Alzheimer's disease.[148] Evidence also suggested the notion that NSAIDs could reduce inflammation related to amyloid plaques, but trials were suspended due to high adverse events.[15] No prevention trial has been completed.[15] They do not appear to be useful as a treatment, but as of 2011[update] were thought to be candidates as presymptomatic preventives.[149] Hormone replacement therapy in menopause, although previously used, may increase risk of dementia.[150]
Lifestyle
Evidence suggests that higher education and occupational attainment, and participation in leisure activities show a reduced risk of developing Alzheimer's,[151] or of delaying the onset of symptoms. This is compatible with the cognitive reserve theory, which states that some life experiences result in more efficient neural functioning providing the individual a cognitive reserve that delays the onset of dementia manifestations.[151] Education delays the onset of Alzheimer's disease syndrome without changing the duration of the disease.[152] Learning a second language even later in life seems to delay the onset of Alzheimer's disease.[153]
Physical exercise is associated with decreased rate of dementia.[154] Physical exercise is also effective in reducing symptom severity in those with Alzheimer's disease.[155]
Diet
Diet is seen to be a modifiable risk factor for the development of dementia. The Mediterranean diet, and the DASH diet are both associated with less cognitive decline. A different approach has been to incorporate elements of both of these diets into one known as the MIND diet.[156] These diets are generally low in saturated fats while providing a good source of carbohydrates, mainly those that help stabilize blood sugar and insulin levels.[157] Those who eat a diet high in saturated fats and simple carbohydrates (mono- and disaccharide) have a higher risk.[158]
Raised blood sugar levels over a long time, can damage nerves and cause memory problems if they are not managed.[159] Nutritional factors associated with the proposed diets for reducing dementia risk, include unsaturated fatty acids, antioxidants vitamin E, vitamin C, and flavonoids, vitamin B, and vitamin D.[160][161]
The MIND diet may be more protective but further studies are needed. The Mediterranean diet seems to be more protective against Alzheimer's than DASH but there are no consistent findings against dementia in general. The role of olive oil needs further study as it may be one of the most important components in reducing the risk of cognitive decline and dementia.[156][162]
In those with celiac disease or non-celiac gluten sensitivity, a strict gluten-free diet may relieve the symptoms given a mild cognitive impairment.[163][97] Once dementia is advanced no evidence suggests that a gluten free diet is useful.[163]
Conclusions on dietary components have been difficult to ascertain as results have differed between population-based studies and randomised controlled trials.[164] There is limited evidence that light to moderate use of alcohol, particularly red wine, is associated with lower risk of Alzheimer's disease.[164] There is tentative evidence that caffeine may be protective.[165] A number of foods high in flavonoids such as cocoa, red wine, and tea may decrease the risk of Alzheimer's disease.[166][167] A number of studies have looked at the possible role of minerals such as selenium,[168] zinc, and copper.[169][170] Omega 3 fatty acid supplements from plants and fish, and dietary docosahexaenoic acid (DHA), do not appear to benefit people with mild to moderate Alzheimer's disease.[171][172]
Curcumin as of 2010[update] had not shown benefit in people even though there is tentative evidence in animals.[173] There is growing evidence (2020) for the neuroprotection offered by the use of cannabinoids in Alzheimer's and other neurodegenerative disorders. However, further population studies are recommended to see this use beyond experimental.[174]
Management
There is no cure for Alzheimer's disease; available treatments offer relatively small symptomatic benefits but remain palliative in nature. Current treatments can be divided into pharmaceutical, psychosocial, and caregiving.
Medications


Medications used to treat the cognitive problems of Alzheimer's disease include: four acetylcholinesterase inhibitors (tacrine, rivastigmine, galantamine, and donepezil) and memantine, an NMDA receptor antagonist. The benefit from their use is small.[175][176][177]
Reduction in the activity of the cholinergic neurons is a well-known feature of Alzheimer's disease.[178] Acetylcholinesterase inhibitors are employed to reduce the rate at which acetylcholine (ACh) is broken down, thereby increasing the concentration of ACh in the brain and combating the loss of ACh caused by the death of cholinergic neurons.[179] There is evidence for the efficacy of these medications in mild to moderate Alzheimer's disease,[180][176][175] and some evidence for their use in the advanced stage.[175] The use of these drugs in mild cognitive impairment has not shown any effect in a delay of the onset of Alzheimer's disease.[181] The most common side effects are nausea and vomiting, both of which are linked to cholinergic excess. These side effects arise in approximately 10–20% of users, are mild to moderate in severity, and can be managed by slowly adjusting medication doses.[182] Less common secondary effects include muscle cramps, decreased heart rate (bradycardia), decreased appetite and weight, and increased gastric acid production.[180]
Glutamate is an excitatory neurotransmitter of the nervous system, although excessive amounts in the brain can lead to cell death through a process called excitotoxicity which consists of the overstimulation of glutamate receptors. Excitotoxicity occurs not only in Alzheimer's disease, but also in other neurological diseases such as Parkinson's disease and multiple sclerosis.[183] Memantine is a noncompetitive NMDA receptor antagonist first used as an anti-influenza agent. It acts on the glutamatergic system by blocking NMDA receptors and inhibiting their overstimulation by glutamate.[183][184] Memantine has been shown to have a small benefit in the treatment of moderate to severe Alzheimer's disease.[185] Reported adverse events with memantine are infrequent and mild, including hallucinations, confusion, dizziness, headache and fatigue.[186] The combination of memantine and donepezil has been shown to be "of statistically significant but clinically marginal effectiveness".[187]
An extract of Ginkgo biloba known as EGb 761 has been widely used for treating Alzheimer's and other neuropsychiatric disorders.[188] Its use is approved throughout Europe.[189] The World Federation of Biological Psychiatry guidelines lists EGb 761 with the same weight of evidence (level B) given to acetylcholinesterase inhibitors, and memantine. EGb 761 is the only one that showed improvement of symptoms in both Alzheimer's disease and vascular dementia. EGb 761 is seen as being able to play an important role either on its own or as an add-on particularly when other therapies prove ineffective.[188] EGb 761 is seen to be neuroprotective; it is a free radical scavenger, improves mitochondrial function, and modulates serotonin and dopamine levels. Many studies of its use in mild to moderate dementia have shown it to significantly improve cognitive function, activities of daily living, and neuropsychiatric symptoms. However, its use has not been shown to prevent the progression to dementia.[188]
Atypical antipsychotics are modestly useful in reducing aggression and psychosis in people with Alzheimer's disease, but their advantages are offset by serious adverse effects, such as stroke, movement difficulties or cognitive decline.[190] When used in the long-term, they have been shown to associate with increased mortality.[191] Stopping antipsychotic use in this group of people appears to be safe.[192]
Psychosocial intervention
Psychosocial interventions are used as an adjunct to pharmaceutical treatment and can be classified within behavior-, emotion-, cognition- or stimulation-oriented approaches. Research on efficacy is unavailable and rarely specific to Alzheimer's disease, focusing instead on dementia in general.[193]
Behavioral interventions attempt to identify and reduce the antecedents and consequences of problem behaviors. This approach has not shown success in improving overall functioning,[194] but can help to reduce some specific problem behaviors, such as incontinence.[195] There is a lack of high quality data on the effectiveness of these techniques in other behavior problems such as wandering.[196][197] Music therapy is effective in reducing behavioral and psychological symptoms.[198]
Emotion-oriented interventions include reminiscence therapy, validation therapy, supportive psychotherapy, sensory integration, also called snoezelen, and simulated presence therapy. A Cochrane review has found no evidence that this is effective.[199] Supportive psychotherapy has received little or no formal scientific study, but some clinicians find it useful in helping mildly impaired people adjust to their illness.[193] Reminiscence therapy (RT) involves the discussion of past experiences individually or in group, many times with the aid of photographs, household items, music and sound recordings, or other familiar items from the past. A 2018 review of the effectiveness of RT found that effects were inconsistent, small in size and of doubtful clinical significance, and varied by setting.[200] Simulated presence therapy (SPT) is based on attachment theories and involves playing a recording with voices of the closest relatives of the person with Alzheimer's disease. There is partial evidence indicating that SPT may reduce challenging behaviors.[201] Finally, validation therapy is based on acceptance of the reality and personal truth of another's experience, while sensory integration is based on exercises aimed to stimulate senses. There is no evidence to support the usefulness of these therapies.[202][203]
The aim of cognition-oriented treatments, which include reality orientation and cognitive retraining, is the reduction of cognitive deficits. Reality orientation consists of the presentation of information about time, place, or person to ease the understanding of the person about its surroundings and his or her place in them. On the other hand, cognitive retraining tries to improve impaired capacities by exercising mental abilities. Both have shown some efficacy improving cognitive capacities,[204] although in some studies these effects were transient and negative effects, such as frustration, have also been reported.[193]
Stimulation-oriented treatments include art, music and pet therapies, exercise, and any other kind of recreational activities. Stimulation has modest support for improving behavior, mood, and, to a lesser extent, function. Nevertheless, as important as these effects are, the main support for the use of stimulation therapies is the change in the person's routine.[193]
Caregiving
Since Alzheimer's has no cure and it gradually renders people incapable of tending to their own needs, caregiving is essentially the treatment and must be carefully managed over the course of the disease.
During the early and moderate stages, modifications to the living environment and lifestyle can increase patient safety and reduce caretaker burden.[205][206] Examples of such modifications are the adherence to simplified routines, the placing of safety locks, the labeling of household items to cue the person with the disease or the use of modified daily life objects.[193][207][208] If eating becomes problematic, food will need to be prepared in smaller pieces or even puréed.[209] When swallowing difficulties arise, the use of feeding tubes may be required. In such cases, the medical efficacy and ethics of continuing feeding is an important consideration of the caregivers and family members.[210][211] The use of physical restraints is rarely indicated in any stage of the disease, although there are situations when they are necessary to prevent harm to the person with Alzheimer's disease or their caregivers.[193]
As the disease progresses, different medical issues can appear, such as oral and dental disease, pressure ulcers, malnutrition, hygiene problems, or respiratory, skin, or eye infections. Careful management can prevent them, while professional treatment is needed when they do arise.[212][213] During the final stages of the disease, treatment is centred on relieving discomfort until death, often with the help of hospice.[214]
Prognosis

The early stages of Alzheimer's disease are difficult to diagnose. A definitive diagnosis is usually made once cognitive impairment compromises daily living activities, although the person may still be living independently. The symptoms will progress from mild cognitive problems, such as memory loss through increasing stages of cognitive and non-cognitive disturbances, eliminating any possibility of independent living, especially in the late stages of the disease.[32]
Life expectancy of people with Alzheimer's disease is reduced.[215] The normal life expectancy for 60 to 70 years old is 23 to 15 years; for 90 years old it is 4.5 years.[216] Following Alzheimer's disease diagnosis it ranges from 7 to 10 years for those in their 60s and early 70s (a loss of 13 to 8 years), to only about 3 years or less (a loss of 1.5 years) for those in their 90s.[215] It is about 50% life expectancy with Alzheimer's disease.
Fewer than 3% of people live more than fourteen years.[217] Disease features significantly associated with reduced survival are an increased severity of cognitive impairment, decreased functional level, history of falls, and disturbances in the neurological examination. Other coincident diseases such as heart problems, diabetes or history of alcohol abuse are also related with shortened survival.[218][219][220] While the earlier the age at onset the higher the total survival years, life expectancy is particularly reduced when compared to the healthy population among those who are younger.[221] Men have a less favourable survival prognosis than women.[217][3]
Pneumonia and dehydration are the most frequent immediate causes of death brought by Alzheimer's disease, while cancer is a less frequent cause of death than in the general population.[3]
Epidemiology
Two main measures are used in epidemiological studies: incidence and prevalence. Incidence is the number of new cases per unit of person-time at risk (usually number of new cases per thousand person-years); while prevalence is the total number of cases of the disease in the population at any given time.
Regarding incidence, cohort longitudinal studies (studies where a disease-free population is followed over the years) provide rates between 10 and 15 per thousand person-years for all dementias and 5–8 for Alzheimer's disease,[222][223] which means that half of new dementia cases each year are Alzheimer's disease. Advancing age is a primary risk factor for the disease and incidence rates are not equal for all ages: every five years after the age of 65, the risk of acquiring the disease approximately doubles, increasing from 3 to as much as 69 per thousand person years.[222][223] There are also sex differences in the incidence rates, women having a higher risk of developing Alzheimer's disease particularly in the population older than 85.[223][224] In the United States , the risk of dying from Alzheimer's disease is 26% higher among the non-Hispanic white population than among the non-Hispanic black population, whereas the Hispanic population has a 30% lower risk than the non-Hispanic white population.[225]
The prevalence of Alzheimer's disease in populations is dependent upon different factors including incidence and survival. Since the incidence of Alzheimer's disease increases with age, it is particularly important to include the mean age of the population of interest. In the United States, Alzheimer's prevalence was estimated to be 1.6% in 2000 both overall and in the 65–74 age group, with the rate increasing to 19% in the 75–84 group and to 42% in the greater than 84 groups.[226] Prevalence rates in less developed regions are lower.[227] The World Health Organization estimated that in 2005, 0.379% of people worldwide had dementia, and that the prevalence would increase to 0.441% in 2015 and to 0.556% in 2030.[228] Other studies have reached similar conclusions.[227] Another study estimated that in 2006, 0.40% of the world population (range 0.17–0.89%; absolute number 26.6 million, range 11.4–59.4 million) were afflicted by Alzheimer's disease, and that the prevalence rate would triple and the absolute number would quadruple by 2050.[229]
History

The ancient Greek and Roman philosophers and physicians associated old age with increasing dementia.[21] It was not until 1901 that German psychiatrist Alois Alzheimer identified the first case of what became known as Alzheimer's disease, named after him, in a fifty-year-old woman he called Auguste D. He followed her case until she died in 1906 when he first reported publicly on it.[230] During the next five years, eleven similar cases were reported in the medical literature, some of them already using the term Alzheimer's disease.[21] The disease was first described as a distinctive disease by Emil Kraepelin after suppressing some of the clinical (delusions and hallucinations) and pathological features (arteriosclerotic changes) contained in the original report of Auguste D.[231] He included Alzheimer's disease, also named presenile dementia by Kraepelin, as a subtype of senile dementia in the eighth edition of his Textbook of Psychiatry, published on 15 July, 1910.[232]
For most of the 20th century, the diagnosis of Alzheimer's disease was reserved for individuals between the ages of 45 and 65 who developed symptoms of dementia. The terminology changed after 1977 when a conference on Alzheimer's disease concluded that the clinical and pathological manifestations of presenile and senile dementia were almost identical, although the authors also added that this did not rule out the possibility that they had different causes.[233] This eventually led to the diagnosis of Alzheimer's disease independent of age.[234] The term senile dementia of the Alzheimer type (SDAT) was used for a time to describe the condition in those over 65, with classical Alzheimer's disease being used to describe those who were younger. Eventually, the term Alzheimer's disease was formally adopted in medical nomenclature to describe individuals of all ages with a characteristic common symptom pattern, disease course, and neuropathology.[235]
Society and culture
Social costs
Dementia, and specifically Alzheimer's disease, may be among the most costly diseases for society in Europe and the United States,[236][237] while their costs in other countries such as Argentina,[238] and South Korea,[239] are also high and rising. These costs will probably increase with the aging of society, becoming an important social problem. AD-associated costs include direct medical costs such as nursing home care, direct nonmedical costs such as in-home day care, and indirect costs such as lost productivity of both patient and caregiver.[237] Numbers vary between studies but dementia costs worldwide have been calculated around $160 billion,[240] while costs of Alzheimer's disease in the United States may be $100 billion each year.[237]
The greatest origin of costs for society is the long-term care by health care professionals and particularly institutionalisation, which corresponds to 2/3 of the total costs for society.[236] The cost of living at home is also very high,[236] especially when informal costs for the family, such as caregiving time and caregiver's lost earnings, are taken into account.[241]
Costs increase with dementia severity and the presence of behavioral disturbances,[242] and are related to the increased caregiving time required for the provision of physical care.[241] Therefore, any treatment that slows cognitive decline, delays institutionalisation or reduces caregivers' hours will have economic benefits. Economic evaluations of current treatments have shown positive results.[237]
Caregiving burden
The role of the main caregiver is often taken by the spouse or a close relative.[243] Alzheimer's disease is known for placing a great burden on caregivers which includes social, psychological, physical or economic aspects.[16][244][245] Home care is usually preferred by people with Alzheimer's disease and their families.[246] This option also delays or eliminates the need for more professional and costly levels of care.[246][247] Nevertheless, two-thirds of nursing home residents have dementias.[193]
Dementia caregivers are subject to high rates of physical and mental disorders.[248] Factors associated with greater psychosocial problems of the primary caregivers include having an affected person at home, the carer being a spouse, demanding behaviors of the cared person such as depression, behavioral disturbances, hallucinations, sleep problems or walking disruptions and social isolation.[249][250] Regarding economic problems, family caregivers often give up time from work to spend 47 hours per week on average with the person with Alzheimer's disease, while the costs of caring for them are high. Direct and indirect costs of caring for somebody with Alzheimer's average between $18,000 and $77,500 per year in the United States, depending on the study.[241][243]
Cognitive behavioral therapy and the teaching of coping strategies either individually or in group have demonstrated their efficacy in improving caregivers' psychological health.[16][251]
Media
Alzheimer's disease has been portrayed in films such as: Iris (2001), based on John Bayley's memoir of his wife Iris Murdoch;[252] The Notebook (2004), based on Nicholas Sparks' 1996 novel of the same name;[253] A Moment to Remember (2004); Thanmathra (2005);[254] Memories of Tomorrow (Ashita no Kioku) (2006), based on Hiroshi Ogiwara's novel of the same name;[255] Away from Her (2006), based on Alice Munro's short story "The Bear Came over the Mountain";[256] Still Alice (2014), about a Columbia University professor who has early onset Alzheimer's disease, based on Lisa Genova's 2007 novel of the same name and featuring Julianne Moore in the title role. Documentaries on Alzheimer's disease include Malcolm and Barbara: A Love Story (1999) and Malcolm and Barbara: Love's Farewell (2007), both featuring Malcolm Pointon.[257][258][259] It has also been portrayed in music by English musician the Caretaker in releases such as Persistent Repetition of Phrases (2008), An Empty Bliss Beyond This World (2011), and Everywhere at the End of Time (2016–2019).[260][261][262] Paintings depicting the disorder include the late works by American artist William Utermohlen, who drew self-portraits from 1995 to 2000 as an experiment of showing his disease through art.[263][264]
Research directions
Treatment and prevention
In the decade 2002–2012, 244 compounds were assessed in Phase I, Phase II, or Phase III trials, and only one of these (memantine) received FDA approval (though others were still in the pipeline).[265] Solanezumab and aducanumab failed to show effectiveness in people who already had Alzheimer's symptoms.[266]
In early 2017, a trial of verubecestat, which inhibits the beta-secretase protein responsible for creating beta-amyloid protein was discontinued as an independent panel found "virtually no chance of finding a positive clinical effect".[267] In 2018 and 2019, more trials, including aducanumab which reduced amyloid beta concentrations, failed, leading some to question the validity of the amyloid hypothesis.[268][269]
The senescence-accelerated mouse (SAMP8) is an Alzheimer's disease (AD) animal model in which amyloid precursor protein (APP) is overproduced. The mice develop early memory disturbances and alterations in the blood-brain barrier, which causes a decreased expulsion of amyloid-β protein from the brain. It has a marked increase in oxidative stress in the brain. Medications that reduce oxidative stress have been shown to improve memory. Treatments that reduce amyloid-β (antisense to APP and antibodies to amyloid-β) not only improve memory but also reduce oxidative stress. It has been shown that the initial deviations in lipid peroxidative damage favor mitochondrial dysfunction as being a trigger for amyloid-β overproduction in this Alzheimer's disease mouse strain. This process begets increased amyloid-beta, which further damages mitochondria.[270]
Research on the effects of meditation on preserving memory and cognitive functions is at an early stage.[271] A 2015 review suggests that mindfulness-based interventions may prevent or delay the onset of mild cognitive impairment and Alzheimer's disease.[272]
The ketogenic diet is a very high-fat, adequate-protein, low-carbohydrate diet that is used to treat refractory epilepsy in children. Designed to mimic some of the effects of fasting, following a ketogenic diet leads to elevated blood levels of molecules called ketone bodies: a metabolic state known as ketosis. These ketone bodies have a neuroprotective effect on aging brain cells, though it is not fully understood why. Limited research in the form of preclinical trials (mice and rats), and small-scale clinical (human) trials, have explored its potential as a therapy for neurodegenerative disorders like Alzheimer's disease.[273]
Infections
The herpes simplex virus HSV-1 has been found in the same areas as amyloid plaques.[274] This suggested the possibility that Alzheimer's disease could be treated or prevented with antiviral medication.[274][275] Studies of antivirals in cell cultures have shown promising results.[276] A 2021 study of 265,172 subjects in Sweden over a 12-year period found that patients with herpes diagnoses not treated with antiviral drugs had a 50% increased risk of dementia over controls, but treatment with antiviral drugs reduced the incidence by 25%.[non-primary source needed][277]
Fungal infection of Alzheimer's disease brain has also been described.[278] This hypothesis was proposed by the microbiologist L. Carrasco when his group found statistical correlation between disseminated mycoses and Alzheimer's disease.[279] Further work revealed that fungal infection is present in different brain regions of Alzheimer's disease patients, but not in the control individuals.[280][281] A fungal infection explains the symptoms observed in Alzheimer's disease patients. The slow progression of Alzheimer's disease fits with the chronic nature of some systemic fungal infections, which can be asymptomatic and thus, unnoticed and untreated.[280] The fungal hypotheses are also compatible with some other established Alzheimer's disease hypotheses, like the amyloid hypothesis, that can be explained as an immune system response to an infection in the CNS,[282][283][284] as found by R. Moir and R. Tanzi in mouse and worm models of Alzheimer's disease.
Diagnosis
Emphasis in Alzheimer's research has been placed on diagnosing the condition before symptoms begin.[285] A number of biochemical tests have been developed to enable earlier detection. Some such tests involve the analysis of cerebrospinal fluid for beta-amyloid, total tau protein and phosphorylated tau181P protein concentrations.[286] Because drawing CSF can be painful, repeated draws are avoided. A blood test for circulatory miRNA and inflammatory biomarkers is a potential alternative indicator.[286]
A series of studies suggest that aging-related breakdown of the blood–brain barrier may be causative of Alzheimer's disease, and conclude that markers for that damage may be an early predictor of the disease.[287][288][289]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 "Alzheimer's disease". BMJ 338: b158. February 2009. doi:10.1136/bmj.b158. PMID 19196745.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 "Dementia Fact sheet". World Health Organization. September 2020. https://www.who.int/en/news-room/fact-sheets/detail/dementia.
- ↑ 3.0 3.1 3.2 "Alzheimer disease and mortality: a 15-year epidemiological study". Archives of Neurology 62 (5): 779–84. May 2005. doi:10.1001/archneur.62.5.779. PMID 15883266.
- ↑ 4.0 4.1 "Early-onset Alzheimer's disease: nonamnestic subtypes and type 2 AD". Archives of Medical Research 43 (8): 677–85. November 2012. doi:10.1016/j.arcmed.2012.11.009. PMID 23178565.
- ↑ "Alzheimer's disease". Lancet 377 (9770): 1019–31. March 2011. doi:10.1016/S0140-6736(10)61349-9. PMID 21371747.
- ↑ 6.0 6.1 "Dementia diagnosis and assessment". National Institute for Health and Care Excellence (NICE). http://pathways.nice.org.uk/pathways/dementia/dementia-diagnosis-and-assessment.pdf.
- ↑ Commission de la transparence (June 2012). "Drugs for Alzheimer's disease: best avoided. No therapeutic advantage". Prescrire International 21 (128): 150. PMID 22822592.
- ↑ 8.0 8.1 "Alzheimer's disease". The New England Journal of Medicine 362 (4): 329–44. January 2010. doi:10.1056/NEJMra0909142. PMID 20107219.
- ↑ 9.0 9.1 GBD 2015 Disease Injury Incidence Prevalence Collaborators (October 2016). "Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet 388 (10053): 1545–1602. doi:10.1016/S0140-6736(16)31678-6. PMID 27733282.
- ↑ 10.0 10.1 GBD 2015 Mortality Causes of Death Collaborators (October 2016). "Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet 388 (10053): 1459–1544. doi:10.1016/S0140-6736(16)31012-1. PMID 27733281.
- ↑ 11.0 11.1 11.2 11.3 11.4 11.5 11.6 11.7 11.8 "Alzheimer's Disease Fact Sheet" (in en). https://www.nia.nih.gov/health/alzheimers-disease-fact-sheet.
- ↑ "Survival in dementia and predictors of mortality: a review". International Journal of Geriatric Psychiatry 28 (11): 1109–24. November 2013. doi:10.1002/gps.3946. PMID 23526458.
- ↑ 13.0 13.1 13.2 13.3 "Alzheimer Disease: An Update on Pathobiology and Treatment Strategies". Cell 179 (2): 312–339. October 2019. doi:10.1016/j.cell.2019.09.001. PMID 31564456.
- ↑ 14.0 14.1 "Study reveals how APOE4 gene may increase risk for dementia" (in en). https://www.nia.nih.gov/news/study-reveals-how-apoe4-gene-may-increase-risk-dementia.
- ↑ 15.0 15.1 15.2 15.3 15.4 15.5 "Primary and Secondary Prevention Trials in Alzheimer Disease: Looking Back, Moving Forward". Current Alzheimer Research 14 (4): 426–40. 2017. doi:10.2174/1567205013666160930112125. PMID 27697063.
- ↑ 16.0 16.1 16.2 16.3 "Systematic review of information and support interventions for caregivers of people with dementia". BMC Geriatrics 7: 18. July 2007. doi:10.1186/1471-2318-7-18. PMID 17662119.
- ↑ "Exercise programs for people with dementia". The Cochrane Database of Systematic Reviews 132 (4): CD006489. April 2015. doi:10.1002/14651858.CD006489.pub4. PMID 25874613. http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1516-31802014000300195.
- ↑ National Institute for Health and Clinical Excellence. "Low-dose antipsychotics in people with dementia". National Institute for Health and Care Excellence (NICE). https://www.nice.org.uk/advice/ktt7/resources/non-guidance-lowdose-antipsychotics-in-people-with-dementia-pdf.
- ↑ "Information for Healthcare Professionals: Conventional Antipsychotics". US Food and Drug Administration. 16 June 2008. https://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm124830.htm.
- ↑ Viña, Jose; Lloret, Ana (2010). "Why women have more Alzheimer's disease than men: gender and mitochondrial toxicity of amyloid-beta peptide". Journal of Alzheimer's Disease 20 Suppl 2: S527–533. doi:10.3233/JAD-2010-100501. ISSN 1875-8908. PMID 20442496. https://pubmed.ncbi.nlm.nih.gov/20442496/#:~:text=Abstract,longevity%20of%20women%20versus%20men..
- ↑ 21.0 21.1 21.2 "Evolution in the conceptualization of dementia and Alzheimer's disease: Greco-Roman period to the 1960s". Neurobiology of Aging 19 (3): 173–89. 1998. doi:10.1016/S0197-4580(98)00052-9. PMID 9661992.
- ↑ Hurd, Michael D.; Martorell, Paco; Delavande, Adeline; Mullen, Kathleen J.; Langa, Kenneth M. (4 April 2013). "Monetary Costs of Dementia in the United States". New England Journal of Medicine 368 (14): 1326–1334. doi:10.1056/NEJMsa1204629. PMID 23550670.
- ↑ 23.0 23.1 23.2 "Alzheimer's disease – Symptoms". 10 May 2018. https://www.nhs.uk/conditions/alzheimers-disease/symptoms/.
- ↑ 24.0 24.1 24.2 24.3 "Recommendations for the diagnosis and management of Alzheimer's disease and other disorders associated with dementia: EFNS guideline". European Journal of Neurology 14 (1): e1–26. January 2007. doi:10.1111/j.1468-1331.2006.01605.x. PMID 17222085.
- ↑ 25.0 25.1 25.2 "Multiple cognitive deficits during the transition to Alzheimer's disease". Journal of Internal Medicine 256 (3): 195–204. September 2004. doi:10.1111/j.1365-2796.2004.01386.x. PMID 15324363.
- ↑ "Instrumental activities of daily living: a stepping-stone towards Alzheimer's disease diagnosis in subjects with mild cognitive impairment?". Acta Neurologica Scandinavica. Supplementum 179 (s179): 42–6. 2003. doi:10.1034/j.1600-0404.107.s179.8.x. PMID 12603250.
- ↑ 27.0 27.1 "Neuropsychological features of mild cognitive impairment and preclinical Alzheimer's disease". Acta Neurologica Scandinavica. Supplementum 179: 34–41. 2003. doi:10.1034/j.1600-0404.107.s179.7.x. PMID 12603249.
- ↑ "Behavioral and psychological symptoms in Alzheimer's dementia and vascular dementia". Handbook of Clinical Neurology 165: 5–32. 2019. doi:10.1016/B978-0-444-64012-3.00002-2. ISBN 9780444640123. PMID 31727229.
- ↑ "Depression and Psychosis in Neurological Practice". Bradley's neurology in clinical practice (6th ed.). Philadelphia, PA: Elsevier/Saunders. 2012. ISBN 978-1-4377-0434-1.
- ↑ "Practice guideline update summary: Mild cognitive impairment: Report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology". Neurology 90 (3): 126–35. January 2018. doi:10.1212/WNL.0000000000004826. PMID 29282327.
- ↑ 31.0 31.1 "The Alzheimer's Disease Clinical Spectrum: Diagnosis and Management". Med Clin North Am 103 (2): 263–293. March 2019. doi:10.1016/j.mcna.2018.10.009. PMID 30704681.
- ↑ 32.00 32.01 32.02 32.03 32.04 32.05 32.06 32.07 32.08 32.09 32.10 32.11 32.12 32.13 32.14 32.15 32.16 32.17 32.18 "Clinical features of Alzheimer's disease". European Archives of Psychiatry and Clinical Neuroscience 249 (6): 288–90. 1999. doi:10.1007/s004060050101. PMID 10653284.
- ↑ "Memory deficits in Alzheimer's patients: a comprehensive review". Neuropsychology Review 3 (2): 119–69. June 1992. doi:10.1007/BF01108841. PMID 1300219.
- ↑ "Implicit memory performance of patients with Alzheimer's disease: a brief review". International Psychogeriatrics 7 (3): 385–92. 1995. doi:10.1017/S1041610295002134. PMID 8821346.
- ↑ 35.0 35.1 "Language performance in Alzheimer's disease and mild cognitive impairment: a comparative review". Journal of Clinical and Experimental Neuropsychology 30 (5): 501–56. July 2008. doi:10.1080/13803390701550128. PMID 18569251.
- ↑ 36.0 36.1 36.2 "Effect of Alzheimer's disease on communication function". Journal of the South Carolina Medical Association 90 (9): 417–23. September 1994. PMID 7967534.
- ↑ "Sundowning and circadian rhythms in Alzheimer's disease". The American Journal of Psychiatry 158 (5): 704–11. May 2001. doi:10.1176/appi.ajp.158.5.704. PMID 11329390.
- ↑ "When home caregiving ends: a longitudinal study of outcomes for caregivers of relatives with dementia". Journal of the American Geriatrics Society 43 (1): 10–6. January 1995. doi:10.1111/j.1532-5415.1995.tb06235.x. PMID 7806732.
- ↑ Alzheimer's disease – Causes (NHS)
- ↑ "Familial Alzheimer's disease mutations at position 22 of the amyloid β-peptide sequence differentially affect synaptic loss, tau phosphorylation and neuronal cell death in an ex vivo system". PLOS ONE 15 (9): e0239584. 2020. doi:10.1371/journal.pone.0239584. PMID 32966331. Bibcode: 2020PLoSO..1539584T.
- ↑ 41.0 41.1 Wang, Hao; Kulas, Joshua A.; Wang, Chao; Holtzman, David M.; Ferris, Heather A.; Hansen, Scott B. (17 August 2021). "Regulation of beta-amyloid production in neurons by astrocyte-derived cholesterol". Proceedings of the National Academy of Sciences 118 (33): e2102191118. doi:10.1073/pnas.2102191118. PMID 34385305.
- ↑ "The role of protein clearance mechanisms in organismal ageing and age-related diseases". Nat Commun 5: 5659. December 2014. doi:10.1038/ncomms6659. PMID 25482515. Bibcode: 2014NatCo...5.5659V.
- ↑ Jacobson, M; McCarthy, N (2002). Apoptosis. Oxford, OX: Oxford University Press. p. 290. ISBN 0199638497.
- ↑ "Translating cell biology into therapeutic advances in Alzheimer's disease". Nature 399 (6738 Suppl): A23–31. June 1999. doi:10.1038/19866. PMID 10392577.
- ↑ "Familial Alzheimer's disease-linked presenilin 1 variants elevate Abeta1-42/1-40 ratio in vitro and in vivo". Neuron 17 (5): 1005–13. November 1996. doi:10.1016/S0896-6273(00)80230-5. PMID 8938131.
- ↑ Kim, JH (December 2018). "Genetics of Alzheimer's Disease.". Dementia and Neurocognitive Disorders 17 (4): 131–36. doi:10.12779/dnd.2018.17.4.131. PMID 30906402.
- ↑ "Microglia in Alzheimer's Disease in the Context of Tau Pathology". Biomolecules 10 (10): 1439. October 2020. doi:10.3390/biom10101439. PMID 33066368.
- ↑ "Apolipoprotein E4: a causative factor and therapeutic target in neuropathology, including Alzheimer's disease". Proceedings of the National Academy of Sciences of the United States of America 103 (15): 5644–51. April 2006. doi:10.1073/pnas.0600549103. PMID 16567625. Bibcode: 2006PNAS..103.5644M.
- ↑ 49.0 49.1 "Alzheimer's disease". Lancet 368 (9533): 387–403. July 2006. doi:10.1016/S0140-6736(06)69113-7. PMID 16876668.
- ↑ "Cholesterol, APOE genotype, and Alzheimer disease: an epidemiologic study of Nigerian Yoruba". Neurology 66 (2): 223–27. January 2006. doi:10.1212/01.wnl.0000194507.39504.17. PMID 16434658.
- ↑ "APOE epsilon4 is not associated with Alzheimer's disease in elderly Nigerians". Annals of Neurology 59 (1): 182–85. January 2006. doi:10.1002/ana.20694. PMID 16278853.
- ↑ "Genome-wide association studies in Alzheimer disease". Archives of Neurology 65 (3): 329–34. March 2008. doi:10.1001/archneur.65.3.329. PMID 18332245.
- ↑ 53.0 53.1 "Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer's disease". Nature Genetics 45 (12): 1452–8. December 2013. doi:10.1038/ng.2802. PMID 24162737.
- ↑ "Variant of TREM2 associated with the risk of Alzheimer's disease". The New England Journal of Medicine 368 (2): 107–16. January 2013. doi:10.1056/NEJMoa1211103. PMID 23150908.
- ↑ "TREM2 variants in Alzheimer's disease". The New England Journal of Medicine 368 (2): 117–27. January 2013. doi:10.1056/NEJMoa1211851. PMID 23150934.
- ↑ "Genetic data and cognitively defined late-onset Alzheimer's disease subgroups". Molecular Psychiatry 25 (11): 2942–51. November 2020. doi:10.1038/s41380-018-0298-8. PMID 30514930.
- ↑ Tomiyama T (2010). "Involvement of beta-amyloid in the etiology of Alzheimer's disease". Brain Nerve 62 (7): 691–699. PMID 20675873.
- ↑ Tomiyama T, Nagata T, Shimada H, Teraoka R, Fukushima A, Kanemitsu H, Takuma H, Kuwano R, Imagawa M, Ataka S, Wada Y, Yoshioka E, Nishizaki T, Watanabe Y, Mori H (2008). "A new amyloid beta variant favoring oligomerization in Alzheimer's-type dementia". Ann Neurol 63 (3): 377–387. doi:10.1002/ana.21321. PMID 18300294.
- ↑ "APP Osaka Mutation in Familial Alzheimer's Disease-Its Discovery, Phenotypes, and Mechanism of Recessive Inheritance". Int J Mol Sci 21 (4): 1413. February 2020. doi:10.3390/ijms21041413. PMID 32093100.
- ↑ "The cholinergic hypothesis of Alzheimer's disease: a review of progress". Journal of Neurology, Neurosurgery, and Psychiatry 66 (2): 137–47. February 1999. doi:10.1136/jnnp.66.2.137. PMID 10071091.
- ↑ "Beyond the cholinergic hypothesis: do current drugs work in Alzheimer's disease?". CNS Neuroscience & Therapeutics 16 (4): 235–45. August 2010. doi:10.1111/j.1755-5949.2010.00175.x. PMID 20560995.
- ↑ "Amyloid deposition as the central event in the aetiology of Alzheimer's disease". Trends in Pharmacological Sciences 12 (10): 383–88. October 1991. doi:10.1016/0165-6147(91)90609-V. PMID 1763432.
- ↑ 63.0 63.1 "Alzheimer's disease-do tauists and baptists finally shake hands?". Trends in Neurosciences 25 (1): 22–26. January 2002. doi:10.1016/S0166-2236(00)02031-2. PMID 11801334.
- ↑ "Alpha- and beta-secretase activity as a function of age and beta-amyloid in Down syndrome and normal brain". Neurobiology of Aging 28 (10): 1493–506. October 2007. doi:10.1016/j.neurobiolaging.2006.06.023. PMID 16904243.
- ↑ "Alzheimer disease and Down syndrome: factors in pathogenesis". Neurobiology of Aging 26 (3): 383–89. March 2005. doi:10.1016/j.neurobiolaging.2004.08.005. PMID 15639317.
- ↑ "Apolipoprotein E, dementia, and cortical deposition of beta-amyloid protein". The New England Journal of Medicine 333 (19): 1242–47. November 1995. doi:10.1056/NEJM199511093331902. PMID 7566000.
- ↑ "Tau proteins and neurofibrillary degeneration". Brain Pathology 1 (4): 279–86. July 1991. doi:10.1111/j.1750-3639.1991.tb00671.x. PMID 1669718.
- ↑ "Tau pathology in Alzheimer disease and other tauopathies". Biochimica et Biophysica Acta (BBA) – Molecular Basis of Disease 1739 (2–3): 198–210. January 2005. doi:10.1016/j.bbadis.2004.09.008. PMID 15615638.
- ↑ "The role of tau phosphorylation and cleavage in neuronal cell death". Frontiers in Bioscience 12: 733–56. January 2007. doi:10.2741/2097. PMID 17127334.
- ↑ "Alzheimer's Disease, Inflammation, and the Role of Antioxidants". J Alzheimers Dis Rep 4 (1): 175–183. June 2020. doi:10.3233/ADR-200171. PMID 32715278.
- ↑ "Inflammation as a central mechanism in Alzheimer's disease". Alzheimers Dement (N Y) 4: 575–590. 2018. doi:10.1016/j.trci.2018.06.014. PMID 30406177.
- ↑ "Alzheimer's disease - a neurospirochetosis. Analysis of the evidence following Koch's and Hill's criteria". Journal of Neuroinflammation 8 (1): 90. August 2011. doi:10.1186/1742-2094-8-90. PMID 21816039.
- ↑ Allen, HB (27 June 2016). "Alzheimer's Disease: Assessing the Role of Spirochetes, Biofilms, the Immune System, and Amyloid-β with Regard to Potential Treatment and Prevention.". Journal of Alzheimer's Disease 53 (4): 1271–76. doi:10.3233/JAD-160388. PMID 27372648.
- ↑ "Implications of sleep disturbance and inflammation for Alzheimer's disease dementia". Lancet Neurol 18 (3): 296–306. March 2019. doi:10.1016/S1474-4422(18)30450-2. PMID 30661858.
- ↑ "Inflammation and Alzheimer's disease: possible role of periodontal diseases". Alzheimer's & Dementia 4 (4): 242–50. July 2008. doi:10.1016/j.jalz.2007.08.004. PMID 18631974.
- ↑ "The interplay between the intestinal microbiota and the brain". Nature Reviews. Microbiology 10 (11): 735–42. November 2012. doi:10.1038/nrmicro2876. PMID 23000955.
- ↑ "Role of the blood-brain barrier in the pathogenesis of Alzheimer's disease". Current Alzheimer Research 4 (2): 191–97. April 2007. doi:10.2174/156720507780362245. PMID 17430246.
- ↑ "Interactions of metals and Apolipoprotein E in Alzheimer's disease". Frontiers in Aging Neuroscience 6: 121. 12 June 2014. doi:10.3389/fnagi.2014.00121. PMID 24971061. "Although we still do not know if the metal ion dyshomeostasis present in AD is a cause or consequence of the disease, there is a growing body of evidence showing a direct correlation between metal ions and key AD-related key proteins.".
- ↑ "Oxidative stress signaling in Alzheimer's disease". Current Alzheimer Research 5 (6): 525–32. December 2008. doi:10.2174/156720508786898451. PMID 19075578.
- ↑ "Plant metal chaperones: a novel perspective in dementia therapy". Amyloid 16 (2): 81–83. 2009. doi:10.1080/13506120902879392. PMID 20536399.
- ↑ "Aluminium and Alzheimer's disease". Alzheimer's Society. http://www.alzheimers.org.uk/Facts_about_dementia/Risk_factors/info_aluminium.htm.
- ↑ "Low levels of aluminum can lead to behavioral and morphological changes associated with Alzheimer's disease and age-related neurodegeneration". Neurotoxicology 52: 222–29. January 2016. doi:10.1016/j.neuro.2015.12.002. PMID 26687397. http://www.escholarship.org/uc/item/5c15r7xj.
- ↑ "Understanding Aspects of Aluminum Exposure in Alzheimer's Disease Development". Brain Pathology 26 (2): 139–54. March 2016. doi:10.1111/bpa.12333. PMID 26494454.
- ↑ "Occupational risk factors in Alzheimer's disease: a review assessing the quality of published epidemiological studies". Occupational and Environmental Medicine 64 (11): 723–32. November 2007. doi:10.1136/oem.2006.028209. PMID 17525096.
- ↑ 85.0 85.1 "Is the Aluminum Hypothesis dead?". Journal of Occupational and Environmental Medicine 56 (5 Suppl): S73–79. May 2014. doi:10.1097/jom.0000000000000063. PMID 24806729.
- ↑ "Role of environmental contaminants in the etiology of Alzheimer's disease: a review". Current Alzheimer Research 12 (2): 116–46. 2015. doi:10.2174/1567205012666150204121719. PMID 25654508.
- ↑ "Cigarette smoking is a risk factor for Alzheimer's Disease: an analysis controlling for tobacco industry affiliation". Journal of Alzheimer's Disease 19 (2): 465–80. 2010. doi:10.3233/JAD-2010-1240. PMID 20110594.
- ↑ "Neuroinflammation – an early event in both the history and pathogenesis of Alzheimer's disease". Neuro-Degenerative Diseases 7 (1–3): 38–41. 2010. doi:10.1159/000283480. PMID 20160456.
- ↑ "Air pollution, oxidative stress, and Alzheimer's disease". Journal of Environmental and Public Health 2012: 1–9. 2012. doi:10.1155/2012/472751. PMID 22523504.
- ↑ "Ambient Air Pollution, Noise, and Late-Life Cognitive Decline and Dementia Risk". Annu Rev Public Health 40: 203–220. April 2019. doi:10.1146/annurev-publhealth-040218-044058. PMID 30935305.
- ↑ "Alzheimer's disease as homeostatic responses to age-related myelin breakdown". Neurobiology of Aging 32 (8): 1341–71. August 2011. doi:10.1016/j.neurobiolaging.2009.08.007. PMID 19775776.
- ↑ "Oligodendrocytes and Alzheimer's disease". The International Journal of Neuroscience 126 (2): 97–104. 2016. doi:10.3109/00207454.2015.1025778. PMID 26000818.
- ↑ 93.0 93.1 93.2 "Retrogenesis: clinical, physiologic, and pathologic mechanisms in brain aging, Alzheimer's and other dementing processes". European Archives of Psychiatry and Clinical Neuroscience 249 Suppl 3 (3): 28–36. 1999. doi:10.1007/pl00014170. PMID 10654097.
- ↑ "Integrating retrogenesis theory to Alzheimer's disease pathology: insight from DTI-TBSS investigation of the white matter microstructural integrity". BioMed Research International 2015: 291658. 2015. doi:10.1155/2015/291658. PMID 25685779.
- ↑ Brenner Carson, Verna (2015). Caregiving for Alzheimer's Disease. New York: Springer New York Academy of Sciences. pp. 1–9. ISBN 978-1-4939-2406-6.
- ↑ "Treatment of Neurological Manifestations of Gluten Sensitivity and Coeliac Disease". Current Treatment Options in Neurology 21 (3): 10. February 2019. doi:10.1007/s11940-019-0552-7. PMID 30806821.
- ↑ 97.0 97.1 "Cognitive impairment in celiac disease and non-celiac gluten sensitivity: review of literature on the main cognitive impairments, the imaging and the effect of gluten free diet". Acta Neurologica Belgica 118 (1): 21–27. March 2018. doi:10.1007/s13760-017-0870-z. PMID 29247390.
- ↑ "Kynurenines, Neuropsychiatric Symptoms, and Cognitive Prognosis in Patients with Mild Dementia". Int J Tryptophan Res 12: 1178646919877883. 2019. doi:10.1177/1178646919877883. PMID 31632053.
- ↑ "Neuropathologic changes in Alzheimer's disease". The Journal of Clinical Psychiatry 64 Suppl 9: 7–10. 2003. PMID 12934968.
- ↑ "Where, when, and in what form does sporadic Alzheimer's disease begin?". Current Opinion in Neurology 25 (6): 708–14. December 2012. doi:10.1097/WCO.0b013e32835a3432. PMID 23160422.
- ↑ "Automated MRI measures identify individuals with mild cognitive impairment and Alzheimer's disease". Brain 132 (Pt 8): 2048–57. August 2009. doi:10.1093/brain/awp123. PMID 19460794.
- ↑ "MRI Software Accurately IDs Preclinical Alzheimer's Disease". Diagnostic Imaging. 20 July 2009. http://www.diagnosticimaging.com/news/display/article/113619/1428344. Retrieved 7 January 2013.
- ↑ 103.0 103.1 "The importance of neuritic plaques and tangles to the development and evolution of AD". Neurology 62 (11): 1984–89. June 2004. doi:10.1212/01.WNL.0000129697.01779.0A. PMID 15184601.
- ↑ DeTure, Michael A.; Dickson, Dennis W. (2019). "The neuropathological diagnosis of Alzheimer's disease". Molecular Neurodegeneration 14 (1): 32. doi:10.1186/s13024-019-0333-5. ISSN 1750-1326. PMID 31375134.
- ↑ "Alzheimer disease without neocortical neurofibrillary tangles: "a second look"". Neurology 62 (7): 1141–47. April 2004. doi:10.1212/01.wnl.0000118212.41542.e7. PMID 15079014.
- ↑ "Regional distribution of neurofibrillary tangles and senile plaques in the cerebral cortex of elderly patients: a quantitative evaluation of a one-year autopsy population from a geriatric hospital". Cerebral Cortex 4 (2): 138–50. 1994. doi:10.1093/cercor/4.2.138. PMID 8038565. http://doc.rero.ch/record/299447/files/4-2-138.pdf.
- ↑ "Lewy body pathology in Alzheimer's disease". Journal of Molecular Neuroscience 17 (2): 225–32. October 2001. doi:10.1385/JMN:17:2:225. PMID 11816795.
- ↑ "Role of protein aggregation in mitochondrial dysfunction and neurodegeneration in Alzheimer's and Parkinson's diseases". Neuromolecular Medicine 4 (1–2): 21–36. 2003. doi:10.1385/NMM:4:1-2:21. PMID 14528050.
- ↑ "Synapse formation and function is modulated by the amyloid precursor protein". The Journal of Neuroscience 26 (27): 7212–21. July 2006. doi:10.1523/JNEUROSCI.1450-06.2006. PMID 16822978.
- ↑ "Roles of amyloid precursor protein and its fragments in regulating neural activity, plasticity and memory". Progress in Neurobiology 70 (1): 1–32. May 2003. doi:10.1016/S0301-0082(03)00089-3. PMID 12927332.
- ↑ "Roles of proteolysis and lipid rafts in the processing of the amyloid precursor protein and prion protein". Biochemical Society Transactions 33 (Pt 2): 335–38. April 2005. doi:10.1042/BST0330335. PMID 15787600.
- ↑ "Amyloid fibrils from the viewpoint of protein folding". Cellular and Molecular Life Sciences 61 (5): 511–24. March 2004. doi:10.1007/s00018-003-3264-8. PMID 15004691.
- ↑ "Tauopathies". Cellular and Molecular Life Sciences 64 (17): 2219–33. September 2007. doi:10.1007/s00018-007-7220-x. PMID 17604998.
- ↑ "Pathogenic tau-induced piRNA depletion promotes neuronal death through transposable element dysregulation in neurodegenerative tauopathies". Nature Neuroscience 21 (8): 1038–48. August 2018. doi:10.1038/s41593-018-0194-1. PMID 30038280.
- ↑ "Current insights into molecular mechanisms of Alzheimer disease and their implications for therapeutic approaches". Neuro-Degenerative Diseases 4 (5): 349–65. 2007. doi:10.1159/000105156. PMID 17622778.
- ↑ "Alzheimer mechanisms and therapeutic strategies". Cell 148 (6): 1204–22. March 2012. doi:10.1016/j.cell.2012.02.040. PMID 22424230.
- ↑ "Neurotrophic and neurotoxic effects of amyloid beta protein: reversal by tachykinin neuropeptides". Science 250 (4978): 279–82. October 1990. doi:10.1126/science.2218531. PMID 2218531. Bibcode: 1990Sci...250..279Y.
- ↑ "Mitochondrial Abeta: a potential cause of metabolic dysfunction in Alzheimer's disease". IUBMB Life 58 (12): 686–94. December 2006. doi:10.1080/15216540601047767. PMID 17424907.
- ↑ "New therapeutic strategies and drug candidates for neurodegenerative diseases: p53 and TNF-alpha inhibitors, and GLP-1 receptor agonists". Annals of the New York Academy of Sciences 1035: 290–315. December 2004. doi:10.1196/annals.1332.018. PMID 15681814. https://zenodo.org/record/1235888.
- ↑ "Neuroinflammation in Alzheimer's disease". Lancet Neurology 14 (4): 388–405. April 2015. doi:10.1016/S1474-4422(15)70016-5. PMID 25792098. PMC 5909703. http://www.escholarship.org/uc/item/99h2f9m1.
- ↑ "New insights into brain BDNF function in normal aging and Alzheimer disease". Brain Research Reviews 59 (1): 201–20. November 2008. doi:10.1016/j.brainresrev.2008.07.007. PMID 18708092.
- ↑ "Neurotrophic factors in Alzheimer's disease: role of axonal transport". Genes, Brain, and Behavior 7 (Suppl 1): 43–56. February 2008. doi:10.1111/j.1601-183X.2007.00378.x. PMID 18184369.
- ↑ "The accurate diagnosis of early-onset dementia". International Journal of Psychiatry in Medicine 36 (4): 401–12. 2006. doi:10.2190/Q6J4-R143-P630-KW41. PMID 17407994. https://zenodo.org/record/1236421.
- ↑ "Therapeutic approaches to Alzheimer's disease". Brain 129 (Pt 11): 2840–55. November 2006. doi:10.1093/brain/awl280. PMID 17018549.
- ↑ Dementia: Quick Reference Guide. London: (UK) National Institute for Health and Clinical Excellence. November 2006. ISBN 978-1-84629-312-2. http://www.nice.org.uk/nicemedia/pdf/CG042quickrefguide.pdf. Retrieved 22 February 2008.
- ↑ "Neural correlates of Alzheimer's disease and mild cognitive impairment: a systematic and quantitative meta-analysis involving 1351 patients". NeuroImage 47 (4): 1196–206. October 2009. doi:10.1016/j.neuroimage.2009.05.037. PMID 19463961.
- ↑ Jie CVML; Treyer, V.; Schibli, R.; Mu, L. (Jan 2021). "Tauvid: The First FDA-Approved PET Tracer for Imaging Tau Pathology in Alzheimer's Disease". Pharmaceuticals (Basel) 14 (2). doi:10.3390/ph14020110. PMID 33573211.
- ↑ 128.0 128.1 "Clinical diagnosis of Alzheimer's disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer's Disease". Neurology 34 (7): 939–44. July 1984. doi:10.1212/wnl.34.7.939. PMID 6610841.
- ↑ "Research criteria for the diagnosis of Alzheimer's disease: revising the NINCDS-ADRDA criteria". The Lancet. Neurology 6 (8): 734–46. August 2007. doi:10.1016/S1474-4422(07)70178-3. PMID 17616482.
- ↑ "Reliability and validity of NINCDS-ADRDA criteria for Alzheimer's disease. The National Institute of Mental Health Genetics Initiative". Archives of Neurology 51 (12): 1198–204. December 1994. doi:10.1001/archneur.1994.00540240042014. PMID 7986174.
- ↑ American Psychiatric Association (2000). Diagnostic and statistical manual of mental disorders: DSM-IV-TR (4th Edition Text Revision ed.). Washington, DC: American Psychiatric Association. ISBN 978-0-89042-025-6.
- ↑ "[Clinical aspects of dementia]" (in ja). [Hokkaido Igaku Zasshi] the Hokkaido Journal of Medical Science 71 (3): 315–20. May 1996. PMID 8752526.
- ↑ "The mini-mental state examination: a comprehensive review". Journal of the American Geriatrics Society 40 (9): 922–35. September 1992. doi:10.1111/j.1532-5415.1992.tb01992.x. PMID 1512391.
- ↑ "Early diagnosis of dementia: neuropsychology". Journal of Neurology 246 (1): 6–15. January 1999. doi:10.1007/s004150050299. PMID 9987708.
- ↑ "The validation of a caregiver assessment of dementia: the Dementia Severity Scale". Alzheimer Disease and Associated Disorders 19 (4): 186–94. 2005. doi:10.1097/01.wad.0000189034.43203.60. PMID 16327345.
- ↑ "[Awareness of deficits and anosognosia in Alzheimer's disease]" (in fr). L'Encephale 30 (6): 570–77. 2004. doi:10.1016/S0013-7006(04)95472-3. PMID 15738860.
- ↑ "[The initial symptoms of Alzheimer disease: caregiver perception]" (in pt). Acta Medica Portuguesa 17 (6): 435–44. 2004. PMID 16197855.
- ↑ "The decreasing prevalence of reversible dementias: an updated meta-analysis". Archives of Internal Medicine 163 (18): 2219–29. October 2003. doi:10.1001/archinte.163.18.2219. PMID 14557220.
- ↑ "Amyloid-associated depression: a prodromal depression of Alzheimer disease?". Archives of General Psychiatry 65 (5): 542–50. May 2008. doi:10.1001/archpsyc.65.5.542. PMID 18458206.
- ↑ "Differential diagnosis of Alzheimer's disease". Neurology 48 (5 Suppl 6): S2–9. May 1997. doi:10.1212/WNL.48.5_Suppl_6.2S. PMID 9153154.
- ↑ "Contribution of depression to cognitive impairment and dementia in older adults". The Neurologist 13 (3): 105–17. May 2007. doi:10.1097/01.nrl.0000252947.15389.a9. PMID 17495754.
- ↑ "(11)C-PIB-PET for the early diagnosis of Alzheimer's disease dementia and other dementias in people with mild cognitive impairment (MCI)". The Cochrane Database of Systematic Reviews (7): CD010386. July 2014. doi:10.1002/14651858.CD010386.pub2. PMID 25052054.
- ↑ "18F-FDG PET for the early diagnosis of Alzheimer's disease dementia and other dementias in people with mild cognitive impairment (MCI)". The Cochrane Database of Systematic Reviews 1: CD010632. January 2015. doi:10.1002/14651858.CD010632.pub2. PMID 25629415.
- ↑ "Diagnosis and treatment of dementia: 1. Risk assessment and primary prevention of Alzheimer disease". CMAJ 178 (5): 548–56. February 2008. doi:10.1503/cmaj.070796. PMID 18299540.
- ↑ "Cardiovascular risk factors for Alzheimer's disease". The American Journal of Geriatric Cardiology 16 (3): 143–49. 2007. doi:10.1111/j.1076-7460.2007.06696.x. PMID 17483665.
- ↑ "Antihypertensive medications and risk for incident dementia and Alzheimer's disease: a meta-analysis of individual participant data from prospective cohort studies". The Lancet. Neurology 19 (1): 61–70. January 2020. doi:10.1016/S1474-4422(19)30393-X. PMID 31706889.
- ↑ "Use of statins and the risk of dementia and mild cognitive impairment: A systematic review and meta-analysis". Sci Rep 8 (1): 5804. April 2018. doi:10.1038/s41598-018-24248-8. PMID 29643479. Bibcode: 2018NatSR...8.5804C.
- ↑ NSAIDs for the chemoprevention of Alzheimer's disease. Subcellular Biochemistry. 42. 2007. pp. 229–48. doi:10.1007/1-4020-5688-5_11. ISBN 978-1-4020-5687-1.
- ↑ "Soothing the inflamed brain: effect of non-steroidal anti-inflammatory drugs on Alzheimer's disease pathology". CNS & Neurological Disorders Drug Targets 10 (1): 57–67. February 2011. doi:10.2174/187152711794488665. PMID 21143138.
- ↑ "Long-term hormone therapy for perimenopausal and postmenopausal women". The Cochrane Database of Systematic Reviews 1: CD004143. January 2017. doi:10.1002/14651858.CD004143.pub5. PMID 28093732.
- ↑ 151.0 151.1 "Cognitive reserve and Alzheimer disease". Alzheimer Disease and Associated Disorders 20 (3 Suppl 2): S69–74. July 2006. doi:10.1097/01.wad.0000213815.20177.19. PMID 16917199.
- ↑ "Systematic review of the effect of education on survival in Alzheimer's disease". International Psychogeriatrics 21 (1): 25–32. February 2009. doi:10.1017/S1041610208008053. PMID 19026089.
- ↑ Neergaard, Lauran (19 February 2011). "Speaking 2 Languages May Delay Getting Alzheimer's". Associated Press. The Denver Post. http://www.denverpost.com/ci_17427474.
- ↑ "Cognitive Reserve and the Prevention of Dementia: the Role of Physical and Cognitive Activities". Current Psychiatry Reports 18 (9): 85. September 2016. doi:10.1007/s11920-016-0721-2. PMID 27481112.
- ↑ "The effect of exercise interventions on cognitive outcome in Alzheimer's disease: a systematic review". International Psychogeriatrics 26 (1): 9–18. January 2014. doi:10.1017/S1041610213001385. PMID 23962667.
- ↑ 156.0 156.1 "Nutritional prevention of cognitive decline and dementia". Acta Biomed 89 (2): 276–290. June 2018. doi:10.23750/abm.v89i2.7401. PMID 29957766.
- ↑ "Diet Affects Markers of Alzheimer's Disease" (in en). https://www.webmd.com/alzheimers/news/20110613/diet-affects-markers-of-alzheimers-disease.
- ↑ "Western diet consumption and cognitive impairment: links to hippocampal dysfunction and obesity". Physiology & Behavior 103 (1): 59–68. April 2011. doi:10.1016/j.physbeh.2010.12.003. PMID 21167850.
- ↑ Editor (2019-01-15). "Memory loss can be caused by a number of factors, from short term causes such as low blood sugar or medication side effects to long term health issues such as dementia." (in en-GB). https://www.diabetes.co.uk/diabetes-complications/diabetes-and-memory-loss.html.
- ↑ "Dietary Patterns and Risk of Dementia: a Systematic Review and Meta-Analysis of Cohort Studies". Molecular Neurobiology 53 (9): 6144–6154. November 2016. doi:10.1007/s12035-015-9516-4. OCLC 6947867710. PMID 26553347.
- ↑ "Nutrition and Dementia: Evidence for Preventive Approaches?". Nutrients (MDPI) 8 (3): 144. March 2016. doi:10.3390/nu8030144. OCLC 8147564576. PMID 26959055.
- ↑ "Mediterranean and MIND Diets Containing Olive Biophenols Reduces the Prevalence of Alzheimer's Disease". Int J Mol Sci 20 (11): 2797. June 2019. doi:10.3390/ijms20112797. PMID 31181669.
- ↑ 163.0 163.1 "Treatment of Neurological Manifestations of Gluten Sensitivity and Coeliac Disease.". Curr Treat Options Neurol 21 (3): 10. 26 February 2019. doi:10.1007/s11940-019-0552-7. PMID 30806821.
- ↑ 164.0 164.1 "Nutrition and the risk of Alzheimer's disease". BioMed Research International 2013: 1–12. 2013. doi:10.1155/2013/524820. PMID 23865055.
- ↑ "Caffeine intake and dementia: systematic review and meta-analysis". Journal of Alzheimer's Disease 20 Suppl 1: S187–204. 2010. doi:10.3233/JAD-2010-091387. PMID 20182026.
- ↑ "The neuroprotective effects of cocoa flavanol and its influence on cognitive performance". British Journal of Clinical Pharmacology 75 (3): 716–27. March 2013. doi:10.1111/j.1365-2125.2012.04378.x. PMID 22775434.
- ↑ "[Dietary flavonoids and human health]". Annales Pharmaceutiques Françaises 69 (2): 78–90. March 2011. doi:10.1016/j.pharma.2010.11.004. PMID 21440100.
- ↑ "Selenium and Alzheimer's disease: a systematic review". Journal of Alzheimer's Disease 26 (1): 81–104. 2011. doi:10.3233/JAD-2011-110414. PMID 21593562.
- ↑ "Zinc diet and Alzheimer's disease: a systematic review". Nutritional Neuroscience 15 (5): 2–12. September 2012. doi:10.1179/1476830512Y.0000000010. PMID 22583839.
- ↑ "Zinc and Copper in Alzheimer's Disease". Journal of Alzheimer's Disease 46 (1): 89–92. 2015. doi:10.3233/JAD-150186. PMID 25835420.
- ↑ "Docosahexaenoic acid homeostasis, brain aging and Alzheimer's disease: Can we reconcile the evidence?". Prostaglandins, Leukotrienes, and Essential Fatty Acids 88 (1): 61–70. January 2013. doi:10.1016/j.plefa.2012.04.006. PMID 22575581.
- ↑ "Omega-3 fatty acids for the treatment of dementia". The Cochrane Database of Systematic Reviews 4: CD009002. April 2016. doi:10.1002/14651858.CD009002.pub3. PMID 27063583.
- ↑ "REVIEW: Curcumin and Alzheimer's disease". CNS Neuroscience & Therapeutics 16 (5): 285–97. October 2010. doi:10.1111/j.1755-5949.2010.00147.x. PMID 20406252.
- ↑ "Cannabinoids for the Neuropsychiatric Symptoms of Dementia: A Systematic Review and Meta-Analysis". Can J Psychiatry 65 (6): 365–376. June 2020. doi:10.1177/0706743719892717. PMID 31835954.
- ↑ 175.0 175.1 175.2 "Donepezil for dementia due to Alzheimer's disease". The Cochrane Database of Systematic Reviews 6: CD001190. June 2018. doi:10.1002/14651858.CD001190.pub3. PMID 29923184.
- ↑ 176.0 176.1 {{cite journal |vauthors=Birks JS, Grimley Evans J |title=Rivastigmine for Alzheimer's disease |journal=The Cochrane Database of Systematic Reviews|issue=4|page=CD001191|date=April 2015|pmid=25858345|doi=10.1002/14651858.CD001191.pub3
- ↑ "Benefits and Harms of Prescription Drugs and Supplements for Treatment of Clinical Alzheimer-Type Dementia". Annals of Internal Medicine 172 (10): 656–668. May 2020. doi:10.7326/M19-3887. PMID 32340037.
- ↑ "Cholinesterases and the pathology of Alzheimer disease". Alzheimer Disease and Associated Disorders 9 Suppl 2: 23–28. 1995. doi:10.1097/00002093-199501002-00005. PMID 8534419.
- ↑ "The new cholinesterase inhibitors for Alzheimer's disease, Part 2: illustrating their mechanisms of action". The Journal of Clinical Psychiatry 61 (11): 813–14. November 2000. doi:10.4088/JCP.v61n1101. PMID 11105732.
- ↑ 180.0 180.1 "Cholinesterase inhibitors for Alzheimer's disease". The Cochrane Database of Systematic Reviews (1): CD005593. January 2006. doi:10.1002/14651858.CD005593. PMID 16437532.
- ↑ "Cholinesterase inhibitors in mild cognitive impairment: a systematic review of randomised trials". PLOS Medicine 4 (11): e338. November 2007. doi:10.1371/journal.pmed.0040338. PMID 18044984.
- ↑ Applied therapeutics : the clinical use of drugs (10th ed.). Baltimore: Wolters Kluwer Health/Lippincott Williams & Wilkins. 2013. p. 2385. ISBN 978-1-60913-713-7.
- ↑ 183.0 183.1 "Paradigm shift in neuroprotection by NMDA receptor blockade: memantine and beyond". Nature Reviews. Drug Discovery 5 (2): 160–70. February 2006. doi:10.1038/nrd1958. PMID 16424917.
- ↑ "Memantine". US National Library of Medicine (Medline). 4 January 2004. https://www.nlm.nih.gov/medlineplus/druginfo/meds/a604006.html.
- ↑ "Memantine for dementia". The Cochrane Database of Systematic Reviews 3 (3): CD003154. May 2019. doi:10.1002/14651858.CD003154.pub6. PMID 30891742.
- ↑ "Namenda prescribing information". Forest Pharmaceuticals. http://www.frx.com/pi/namenda_pi.pdf. (primary source)
- ↑ "Effectiveness of cholinesterase inhibitors and memantine for treating dementia: evidence review for a clinical practice guideline". Annals of Internal Medicine 148 (5): 379–97. March 2008. doi:10.7326/0003-4819-148-5-200803040-00009. PMID 18316756.
- ↑ 188.0 188.1 188.2 "Treatment of dementia and mild cognitive impairment with or without cerebrovascular disease: Expert consensus on the use of Ginkgo biloba extract, EGb 761". CNS Neurosci Ther 25 (2): 288–298. February 2019. doi:10.1111/cns.13095. PMID 30648358.
- ↑ "Ginkgo Biloba extract EGb 761 in the symptomatic treatment of mild-to-moderate dementia: a profile of its use". Drugs Ther Perspect 34 (8): 358–366. 2018. doi:10.1007/s40267-018-0537-8. PMID 30546253.
- ↑ "The effectiveness of atypical antipsychotics for the treatment of aggression and psychosis in Alzheimer's disease". The Cochrane Database of Systematic Reviews (1): CD003476. January 2006. doi:10.1002/14651858.CD003476.pub2. PMID 16437455.
- ↑ "The dementia antipsychotic withdrawal trial (DART-AD): long-term follow-up of a randomised placebo-controlled trial". The Lancet Neurology 8 (2): 151–57. February 2009. doi:10.1016/S1474-4422(08)70295-3. PMID 19138567.
- ↑ "Withdrawal versus continuation of chronic antipsychotic drugs for behavioural and psychological symptoms in older people with dementia". The Cochrane Database of Systematic Reviews 3 (3): CD007726. March 2013. doi:10.1002/14651858.CD007726.pub2. PMID 23543555. https://espace.library.uq.edu.au/view/UQ:296637/UQ296637_OA.pdf.
- ↑ 193.0 193.1 193.2 193.3 193.4 193.5 193.6 "American Psychiatric Association practice guideline for the treatment of patients with Alzheimer's disease and other dementias. Second edition". The American Journal of Psychiatry 164 (12 Suppl): 5–56. December 2007. PMID 18340692.
- ↑ "Cognitive rehabilitation combined with drug treatment in Alzheimer's disease patients: a pilot study". Clinical Rehabilitation 19 (8): 861–69. December 2005. doi:10.1191/0269215505cr911oa. PMID 16323385.
- ↑ "Practice parameter: management of dementia (an evidence-based review). Report of the Quality Standards Subcommittee of the American Academy of Neurology". Neurology 56 (9): 1154–66. May 2001. doi:10.1212/WNL.56.9.1154. PMID 11342679.
- ↑ "Non-pharmacological interventions for wandering of people with dementia in the domestic setting". The Cochrane Database of Systematic Reviews (1): CD005994. January 2007. doi:10.1002/14651858.CD005994.pub2. PMID 17253573.
- ↑ "Effectiveness and acceptability of non-pharmacological interventions to reduce wandering in dementia: a systematic review". International Journal of Geriatric Psychiatry 22 (1): 9–22. January 2007. doi:10.1002/gps.1643. PMID 17096455.
- ↑ "Systematic review of systematic reviews of non-pharmacological interventions to treat behavioural disturbances in older patients with dementia. The SENATOR-OnTop series". BMJ Open 7 (3): e012759. March 2017. doi:10.1136/bmjopen-2016-012759. PMID 28302633.
- ↑ "Snoezelen for dementia". The Cochrane Database of Systematic Reviews (4): CD003152. 2002. doi:10.1002/14651858.CD003152. PMID 12519587.
- ↑ "Reminiscence therapy for dementia". The Cochrane Database of Systematic Reviews 2018 (3): CD001120. March 2018. doi:10.1002/14651858.CD001120.pub3. PMID 29493789. PMC 6494367. http://discovery.ucl.ac.uk/1345239/.
- ↑ "Effectiveness of simulated presence therapy for individuals with dementia: a systematic review and meta-analysis". Aging & Mental Health 12 (6): 779–85. November 2008. doi:10.1080/13607860802380631. PMID 19023729.
- ↑ "Validation therapy for dementia". The Cochrane Database of Systematic Reviews (3): CD001394. 2003. doi:10.1002/14651858.CD001394. PMID 12917907.
- ↑ "Snoezelen for dementia". The Cochrane Database of Systematic Reviews (4): CD003152. 2002. doi:10.1002/14651858.CD003152. PMID 12519587. (up to date as of 2009)
- ↑ "Efficacy of an evidence-based cognitive stimulation therapy programme for people with dementia: randomised controlled trial". The British Journal of Psychiatry 183 (3): 248–54. September 2003. doi:10.1192/bjp.183.3.248. PMID 12948999.
- ↑ "A randomized, controlled trial of a home environmental intervention: effect on efficacy and upset in caregivers and on daily function of persons with dementia". The Gerontologist 41 (1): 4–14. February 2001. doi:10.1093/geront/41.1.4. PMID 11220813.
- ↑ "Maintenance of effects of the home environmental skill-building program for family caregivers and individuals with Alzheimer's disease and related disorders". The Journals of Gerontology. Series A, Biological Sciences and Medical Sciences 60 (3): 368–74. March 2005. doi:10.1093/gerona/60.3.368. PMID 15860476.
- ↑ "Treating Behavioral and Psychiatric Symptoms". Alzheimer's Association. 2006. http://www.alz.org/Health/Treating/agitation.asp.
- ↑ "Visual contrast enhances food and liquid intake in advanced Alzheimer's disease". Clinical Nutrition 23 (4): 533–38. August 2004. doi:10.1016/j.clnu.2003.09.015. PMID 15297089.
- ↑ Nutrition Essentials for Nursing Practice. Hagerstown, Maryland: Lippincott Williams & Wilkins. 2007. p. 360. ISBN 978-0-7817-6651-7. https://books.google.com/books?id=01zo6yf0IUEC&pg=PA360. Retrieved 19 August 2008.
- ↑ "Analysis of patients' rights: dementia and PEG insertion". British Journal of Nursing 15 (1): 18–20. 2006. doi:10.12968/bjon.2006.15.1.20303. PMID 16415742.
- ↑ "Tube feeding patients with dementia". Nutrition in Clinical Practice 21 (2): 142–46. April 2006. doi:10.1177/0115426506021002142. PMID 16556924.
- ↑ "Predictors of mortality in patients with Alzheimer's disease living in nursing homes". Journal of Neurology, Neurosurgery, and Psychiatry 67 (1): 59–65. July 1999. doi:10.1136/jnnp.67.1.59. PMID 10369823.
- ↑ Medical issues:
- "Palliative care for persons with dementia". Home Healthcare Nurse 21 (1): 53–60; quiz 61. January 2003. doi:10.1097/00004045-200301000-00012. PMID 12544465.
- "Alzheimer's disease: psychopathology, medical management and dental implications". Journal of the American Dental Association 137 (9): 1240–51. September 2006. doi:10.14219/jada.archive.2006.0381. PMID 16946428.
- "Practical guidelines for the diagnosis and management of weight loss in Alzheimer's disease: a consensus from appropriateness ratings of a large expert panel". The Journal of Nutrition, Health & Aging 11 (1): 33–37. 2007. PMID 17315078.
- "Training caregivers to change the sleep hygiene practices of patients with dementia: the NITE-AD project". Journal of the American Geriatrics Society 51 (10): 1455–60. October 2003. doi:10.1046/j.1532-5415.2003.51466.x. PMID 14511168.
- "Higher respiratory infection rates on an Alzheimer's special care unit and successful intervention". Journal of the American Geriatrics Society 43 (12): 1341–44. December 1995. doi:10.1111/j.1532-5415.1995.tb06611.x. PMID 7490383.
- ↑ "Palliative Excellence in Alzheimer Care Efforts (PEACE): a program description". Journal of Palliative Medicine 6 (2): 315–20. April 2003. doi:10.1089/109662103764978641. PMID 12854952.
- ↑ 215.0 215.1 "Life expectancy in Alzheimer's disease (AD)". Archives of Gerontology and Geriatrics 49 Suppl 1: 237–43. 2009. doi:10.1016/j.archger.2009.09.035. PMID 19836639.
- ↑ "United States Life Tables, 2017". National Vital Statistics Reports, CDC. https://www.cdc.gov/nchs/data/nvsr/nvsr68/nvsr68_07-508.pdf.
- ↑ 217.0 217.1 "Long-term survival and predictors of mortality in Alzheimer's disease and multi-infarct dementia". Acta Neurologica Scandinavica 91 (3): 159–64. March 1995. doi:10.1111/j.1600-0404.1995.tb00426.x. PMID 7793228.
- ↑ "Predictors of mortality in patients diagnosed with probable Alzheimer's disease". Neurology 47 (2): 433–39. August 1996. doi:10.1212/wnl.47.2.433. PMID 8757016.
- ↑ "Survival after initial diagnosis of Alzheimer disease". Annals of Internal Medicine 140 (7): 501–09. April 2004. doi:10.7326/0003-4819-140-7-200404060-00008. PMID 15068977.
- ↑ "Predictors of survival with Alzheimer's disease: a community-based study". Psychological Medicine 25 (1): 171–77. January 1995. doi:10.1017/S0033291700028191. PMID 7792352.
- ↑ "Functional transitions and active life expectancy associated with Alzheimer disease". Archives of Neurology 60 (2): 253–59. February 2003. doi:10.1001/archneur.60.2.253. PMID 12580712.
- ↑ 222.0 222.1 "Incidence and subtypes of dementia in three elderly populations of central Spain". Journal of the Neurological Sciences 264 (1–2): 63–72. January 2008. doi:10.1016/j.jns.2007.07.021. PMID 17727890.
- ↑ 223.0 223.1 223.2 "Incidence of dementia, Alzheimer's disease, and vascular dementia in Italy. The ILSA Study". Journal of the American Geriatrics Society 50 (1): 41–48. January 2002. doi:10.1046/j.1532-5415.2002.50006.x. PMID 12028245.
- ↑ "Gender differences in the incidence of AD and vascular dementia: The EURODEM Studies. EURODEM Incidence Research Group". Neurology 53 (9): 1992–97. December 1999. doi:10.1212/wnl.53.9.1992. PMID 10599770.
- ↑ Tejada-Vera B. (2013). Mortality from Alzheimer's Disease in the United States: Data for 2000 and 2010. Hyattsville, MD: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics.
- ↑ 2000 U.S. estimates:
- "Alzheimer disease in the US population: prevalence estimates using the 2000 census". Archives of Neurology 60 (8): 1119–22. August 2003. doi:10.1001/archneur.60.8.1119. PMID 12925369.
- "Profiles of General Demographic Characteristics, 2000 Census of Population and Housing, United States". U.S. Census Bureau. 2001. https://www.census.gov/prod/cen2000/dp1/2kh00.pdf.
- ↑ 227.0 227.1 "Global prevalence of dementia: a Delphi consensus study". Lancet 366 (9503): 2112–17. December 2005. doi:10.1016/S0140-6736(05)67889-0. PMID 16360788.
- ↑ World Health Organization (2006). Neurological Disorders: Public Health Challenges. Switzerland: World Health Organization. pp. 204–07. ISBN 978-92-4-156336-9. https://www.who.int/mental_health/neurology/neurodiso/en/index.html.
- ↑
2006 prevalence estimate:
- "Forecasting the global burden of Alzheimer's disease". Alzheimer's & Dementia 3 (3): 186–91. July 2007. doi:10.1016/j.jalz.2007.04.381. PMID 19595937. http://works.bepress.com/cgi/viewcontent.cgi?article=1022&context=rbrookmeyer. Retrieved 18 June 2008.
- "Highlights". World Population Prospects: The 2006 Revision. Working Paper No. ESA/P/WP.202 (Population Division, Department of Economic and Social Affairs, United Nations). 2007. http://un.org/esa/population/publications/wpp2006/WPP2006_Highlights_rev.pdf. Retrieved 27 August 2008.
- ↑ Auguste D.:
- Alzheimer, Alois (1907). "Über eine eigenartige Erkrankung der Hirnrinde" (in de). Allgemeine Zeitschrift für Psychiatrie und Psychisch-Gerichtlich Medizin 64 (1–2): 146–48.
- "About a peculiar disease of the cerebral cortex. By Alois Alzheimer, 1907 (Translated by L. Jarvik and H. Greenson)". Alzheimer Disease and Associated Disorders 1 (1): 3–8. 1987. PMID 3331112.
- Ulrike, Maurer; Konrad, Maurer (2003). Alzheimer: The Life of a Physician and the Career of a Disease. New York: Columbia University Press. p. 270. ISBN 978-0-231-11896-5. https://archive.org/details/alzheimerlifeofp00maur/page/270.
- ↑ "Alzheimer's Disease: A Conceptual History". Int. J. Geriatr. Psychiatry 5 (6): 355–65. 1990. doi:10.1002/gps.930050603.
- ↑ Kraepelin Emil (2007). Clinical Psychiatry: A Textbook For Students And Physicians (Reprint). Kessinger Publishing. p. 568. ISBN 978-1-4325-0833-3.
- ↑ Alzheimer's Disease: Senile Dementia and Related Disorders. New York: Raven Press. 1978. p. 595. ISBN 978-0-89004-225-0.
- ↑ "History of dementia and dementia in history: an overview". Journal of the Neurological Sciences 158 (2): 125–33. June 1998. doi:10.1016/S0022-510X(98)00128-2. PMID 9702682.
- ↑ "Origin of the distinction between Alzheimer's disease and senile dementia: how history can clarify nosology". Neurology 36 (11): 1497–9. November 1986. doi:10.1212/wnl.36.11.1497. PMID 3531918.
- ↑ 236.0 236.1 236.2 "[The economical impact of dementia]" (in fr). Presse Médicale 34 (1): 35–41. January 2005. doi:10.1016/s0755-4982(05)83882-5. PMID 15685097.
- ↑ 237.0 237.1 237.2 237.3 "Economic considerations in Alzheimer's disease". Pharmacotherapy 18 (2 Pt 2): 68–73; discussion 79–82. 1998. doi:10.1002/j.1875-9114.1998.tb03880.x. PMID 9543467. https://accpjournals.onlinelibrary.wiley.com/doi/10.1002/j.1875-9114.1998.tb03880.x.
- ↑ "Economic impact of dementia in developing countries: an evaluation of costs of Alzheimer-type dementia in Argentina". International Psychogeriatrics 19 (4): 705–18. August 2007. doi:10.1017/S1041610206003784. PMID 16870037.
- ↑ "The economic costs of dementia in Korea, 2002". International Journal of Geriatric Psychiatry 21 (8): 722–28. August 2006. doi:10.1002/gps.1552. PMID 16858741.
- ↑ "An estimate of the worldwide prevalence and direct costs of dementia in 2003". Dementia and Geriatric Cognitive Disorders 21 (3): 175–81. 2006. doi:10.1159/000090733. PMID 16401889.
- ↑ 241.0 241.1 241.2 "Informal costs of dementia care: estimates from the National Longitudinal Caregiver Study". The Journals of Gerontology. Series B, Psychological Sciences and Social Sciences 56 (4): S219–28. July 2001. doi:10.1093/geronb/56.4.S219. PMID 11445614.
- ↑ "Determinants of costs of care for patients with Alzheimer's disease". International Journal of Geriatric Psychiatry 21 (5): 449–59. May 2006. doi:10.1002/gps.1489. PMID 16676288.
- ↑ 243.0 243.1 "The MetLife study of Alzheimer's disease: The caregiving experience". MetLife Mature Market Institute. August 2006. http://www.metlife.com/assets/cao/mmi/publications/studies/mmi-alzheimers-disease-caregiving-experience-study.pdf.
- ↑ "EUROCARE: a cross-national study of co-resident spouse carers for people with Alzheimer's disease: I – Factors associated with carer burden". International Journal of Geriatric Psychiatry 14 (8): 651–61. August 1999. doi:10.1002/(SICI)1099-1166(199908)14:8<651::AID-GPS992>3.0.CO;2-B. PMID 10489656.
- ↑ "EUROCARE: a cross-national study of co-resident spouse carers for people with Alzheimer's disease: II – A qualitative analysis of the experience of caregiving". International Journal of Geriatric Psychiatry 14 (8): 662–67. August 1999. doi:10.1002/(SICI)1099-1166(199908)14:8<662::AID-GPS993>3.0.CO;2-4. PMID 10489657.
- ↑ 246.0 246.1 "Economic considerations in the management of Alzheimer's disease". Clinical Interventions in Aging 1 (2): 143–54. 2006. doi:10.2147/ciia.2006.1.2.143. PMID 18044111.
- ↑ "Early community-based service utilization and its effects on institutionalization in dementia caregiving". The Gerontologist 45 (2): 177–85. April 2005. doi:10.1093/geront/45.2.177. PMID 15799982.
- ↑ "The dementias". Lancet 360 (9347): 1759–66. November 2002. doi:10.1016/S0140-6736(02)11667-9. PMID 12480441.
- ↑ "Psychosocial effects on carers of living with persons with dementia". The Australian and New Zealand Journal of Psychiatry 24 (3): 351–61. September 1990. doi:10.3109/00048679009077702. PMID 2241719.
- ↑ "Determinants of carer stress in Alzheimer's disease". International Journal of Geriatric Psychiatry 13 (4): 248–56. April 1998. doi:10.1002/(SICI)1099-1166(199804)13:4<248::AID-GPS770>3.0.CO;2-0. PMID 9646153.
- ↑ "A systematic review of the effectiveness of psychosocial interventions for carers of people with dementia". Aging & Mental Health 5 (2): 107–19. May 2001. doi:10.1080/13607860120038302. PMID 11511058.
- ↑ Bayley, John (2000). Iris: A Memoir of Iris Murdoch. London: Abacus. ISBN 978-0-349-11215-2. OCLC 41960006.
- ↑ Sparks, Nicholas (1996). The notebook. Thorndike, Maine: Thorndike Press. p. 268. ISBN 978-0-7862-0821-0. https://archive.org/details/notebook00spar_1/page/268.
- ↑ "Thanmathra". Webindia123.com. http://www.webindia123.com/movie/regional/thanmatra/index.htm.
- ↑ Ogiwara, Hiroshi (2004) (in ja). Ashita no Kioku. Tōkyō: Kōbunsha. ISBN 978-4-334-92446-1. OCLC 57352130.
- ↑ Munro, Alice (2001). Hateship, Friendship, Courtship, Loveship, Marriage: Stories. New York: A.A. Knopf. ISBN 978-0-375-41300-1. OCLC 46929223.
- ↑ "Malcolm and Barbara: A love story". Dfgdocs. http://www.dfgdocs.com/Directory/Titles/700.aspx.
- ↑ "Malcolm and Barbara: A love story". BBC Cambridgeshire. https://www.bbc.co.uk/cambridgeshire/content/articles/2007/08/06/pointon_audio_feature.shtml.
- ↑ "Alzheimer's film-maker to face ITV lawyers". London: Guardian Media. 7 August 2007. https://www.theguardian.com/media/2007/aug/07/broadcasting.itv.
- ↑ "The Caretaker: Persistent Repetition of Phrases". 26 August 2009. https://www.factmag.com/2009/08/26/the-caretaker-persistent-repetition-of-phrases/.
- ↑ Powell, Mike (14 June 2011). "The Caretaker: An Empty Bliss Beyond This World Album Review". https://pitchfork.com/reviews/albums/15518-an-empty-bliss-beyond-this-world/.
- ↑ Ezra, Marcus (23 October 2020). "Why Are TikTok Teens Listening to an Album About Dementia?". https://www.nytimes.com/2020/10/23/style/tiktok-caretaker-challenge-dementia.html.
- ↑ Gerrard, Nicci (19 July 2015). "Words fail us: dementia and the arts". https://www.theguardian.com/culture/2015/jul/19/dementia-and-the-arts-fiction-films-drama-poetry-painting.
- ↑ Grady, Denise (24 October 2006). "Self-Portraits Chronicle a Descent Into Alzheimer's". https://www.nytimes.com/2006/10/24/health/24alzh.html.
- ↑ "Alzheimer's disease drug-development pipeline: few candidates, frequent failures". Alzheimer's Research & Therapy 6 (4): 37. July 2014. doi:10.1186/alzrt269. PMID 25024750.
- ↑ Gutis, Phillip S. (22 March 2019). "An Alzheimer's Drug Trial Gave Me Hope, and Then It Ended". The New York Times. https://www.nytimes.com/2019/03/22/well/mind/alzheimers-drug-trial-study-biogen-dementia-treatment-cure.html.
- ↑ Feuerstein, Adam (14 February 2017). "Merck Alzheimer's Drug Study Halted Early for Futility". New York City: TheStreet, Inc.. https://www.thestreet.com/story/14002423/1/merck-alzheimer-s-drug-study-halted-early-for-futility.html. Merck Alzheimer's Drug Study Halted Early for Futility Independent study monitors concluded that there was "virtually no chance of finding a positive clinical effect."
- ↑ "After A Big Failure, Scientists And Patients Hunt For A New Type Of Alzheimer's Drug". NPR.org. https://www.npr.org/sections/health-shots/2019/05/03/718754791/after-a-big-failure-scientists-and-patients-hunt-for-a-new-type-of-alzheimers-dr.
- ↑ Gallagher, James (2 May 2019). "Dementia is 'greatest health challenge'". https://www.bbc.com/news/health-48094398.
- ↑ "The senescence accelerated mouse (SAMP8) as a model for oxidative stress and Alzheimer's disease". Biochimica et Biophysica Acta (BBA) – Molecular Basis of Disease 1822 (5): 650–6. May 2012. doi:10.1016/j.bbadis.2011.11.015. PMID 22142563.
- ↑ "Effect of meditation on cognitive functions in context of aging and neurodegenerative diseases". Frontiers in Behavioral Neuroscience 8: 17. 2014. doi:10.3389/fnbeh.2014.00017. PMID 24478663.
- ↑ "Potential benefits of mindfulness-based interventions in mild cognitive impairment and Alzheimer's disease: an interdisciplinary perspective". Behavioural Brain Research 276 (276): 199–212. January 2015. doi:10.1016/j.bbr.2014.05.058. PMID 24893317.
- ↑ "Ketogenic Diet in Alzheimer's Disease". Int J Mol Sci 20 (16): 3892. August 2019. doi:10.3390/ijms20163892. PMID 31405021.
- ↑ 274.0 274.1 "[Herpes simplex virus type 1 as risk factor associated to Alzheimer disease]" (in es). Revista Médica de Chile 139 (6): 779–86. June 2011. doi:10.4067/S0034-98872011000600013. PMID 22051760.
- ↑ "Herpes simplex virus type 1 DNA is located within Alzheimer's disease amyloid plaques". The Journal of Pathology 217 (1): 131–38. January 2009. doi:10.1002/path.2449. PMID 18973185.
- ↑ "Herpes simplex virus type 1 and Alzheimer's disease: increasing evidence for a major role of the virus". Frontiers in Aging Neuroscience 6: 202. 2014. doi:10.3389/fnagi.2014.00202. PMID 25157230.
- ↑ Lopatko Lindman, K., Hemmingsson, E.S., Weidung, B., Brännström, J., Josefsson, M., Olsson, J., Elgh, F., Nordström, P. and Lövheim, H. (2021). "Herpesvirus infections, antiviral treatment, and the risk of dementia—a registry‐based cohort study in Sweden". Alzheimer's & Dementia: Translational Research & Clinical Interventions 7 (1): e12119. doi:10.1002/trc2.12119. PMID 33614892.
- ↑ "Microbes and Alzheimer's Disease". Journal of Alzheimer's Disease 51 (4): 979–84. 2016. doi:10.3233/JAD-160152. PMID 26967229.
- ↑ "Alzheimer's disease and disseminated mycoses". European Journal of Clinical Microbiology & Infectious Diseases 33 (7): 1125–32. July 2014. doi:10.1007/s10096-013-2045-z. PMID 24452965.
- ↑ 280.0 280.1 "Different Brain Regions are Infected with Fungi in Alzheimer's Disease". Scientific Reports 5: 15015. October 2015. doi:10.1038/srep15015. PMID 26468932. Bibcode: 2015NatSR...515015P.
- ↑ "Fungus, the bogeyman". The Economist. 2015-10-22. https://www.economist.com/news/science-and-technology/21676754-curious-result-hints-possibility-dementia-caused-fungal.
- ↑ "Amyloid-β peptide protects against microbial infection in mouse and worm models of Alzheimer's disease". Science Translational Medicine 8 (340): 340ra72. May 2016. doi:10.1126/scitranslmed.aaf1059. PMID 27225182.
- ↑ Kolata, Gina (2016-05-25). "Could Alzheimer's Stem From Infections? It Makes Sense, Experts Say". The New York Times. https://www.nytimes.com/2016/05/26/health/alzheimers-disease-infection.html.
- ↑ "Alzheimer's culprit may fight other diseases". Science News. 2016-06-16. https://www.sciencenews.org/article/alzheimer%E2%80%99s-culprit-may-fight-other-diseases.
- ↑ "Preclinical Alzheimer's disease: diagnosis and prediction of progression". The Lancet. Neurology 4 (9): 576–79. September 2005. doi:10.1016/s1474-4422(05)70168-x. PMID 16109364.
- ↑ 286.0 286.1 "Exploring Biomarkers for Alzheimer's Disease". Journal of Clinical and Diagnostic Research 10 (7): KE01–06. July 2016. doi:10.7860/JCDR/2016/18828.8166. PMID 27630867.
- ↑ "Blood-brain barrier breakdown in the aging human hippocampus". Neuron 85 (2): 296–302. January 2015. doi:10.1016/j.neuron.2014.12.032. PMID 25611508.
- ↑ "Blood-brain barrier breakdown is an early biomarker of human cognitive dysfunction". Nature Medicine 25 (2): 270–276. February 2019. doi:10.1038/s41591-018-0297-y. PMID 30643288.
- ↑ "APOE4 leads to blood-brain barrier dysfunction predicting cognitive decline". Nature 581 (7806): 71–76. May 2020. doi:10.1038/s41586-020-2247-3. PMID 32376954. Bibcode: 2020Natur.581...71M.
Further reading
| Library resources about Alzheimer's Disease |
- "Greater cognitive deterioration in women than men with Alzheimer's disease: a meta analysis". Journal of Clinical and Experimental Neuropsychology 34 (9): 989–98. 2012. doi:10.1080/13803395.2012.712676. PMID 22913619.
- "Advancements in nanotherapeutics for Alzheimer's disease: current perspectives". The Journal of Pharmacy and Pharmacology 71 (9): 1370–1383. September 2019. doi:10.1111/jphp.13132. PMID 31304982.
External links
| Classification | |
|---|---|
| External resources |
|
 |



