Biology:Rubredoxin
| Rubredoxin | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 rubredoxin domain ii from pseudomonas oleovorans | |||||||||
| Identifiers | |||||||||
| Symbol | Rubredoxin | ||||||||
| Pfam | PF00301 | ||||||||
| Pfam clan | CL0045 | ||||||||
| InterPro | IPR004039 | ||||||||
| PROSITE | PDOC00179 | ||||||||
| SCOP2 | 7rxn / SCOPe / SUPFAM | ||||||||
| |||||||||
Rubredoxins are a class of low-molecular-weight iron-containing proteins found in sulfur-metabolizing bacteria and archaea. Sometimes rubredoxins are classified as iron-sulfur proteins; however, in contrast to iron-sulfur proteins, rubredoxins do not contain inorganic sulfide. Like cytochromes, ferredoxins and Rieske proteins, rubredoxins are thought to participate in electron transfer in biological systems. Recent work in bacteria[1] and algae[2] have led to the hypothesis that some rubredoxins may instead have a role in delivering iron to metalloproteins.
Structure
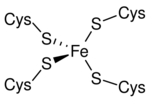
The 3-D structures of a number of rubredoxins have been solved. The fold belongs to the α+β class, with 2 α-helices and 2-3 β-strands. Rubredoxin active site contains an iron ion which is coordinated by the sulfurs of four conserved cysteine residues forming an almost regular tetrahedron. This is sometimes denoted as a [1Fe-0S] or an Fe1S0 system, in analogy to the nomenclature for iron-sulfur proteins. While the vast majority of rubredoxins are soluble, there exists a membrane-bound rubredoxin, referred to as rubredoxin A, in oxygenic photoautotrophs.[3]
Rubredoxins perform one-electron transfer processes. The central iron atom changes between the +2 and +3 oxidation states. In both oxidation states, the metal remains high spin, which helps to minimize structural changes. The reduction potential of a rubredoxin is typically in the range +50 mV to -50 mV.
This iron-sulphur protein is an electron carrier, and it is easy to distinguish its metallic centre changes: the oxidized state is reddish (due to a ligand metal charge transfer), while the reduced state is colourless (because the electron transition has an energy of the infrared level, which is imperceptible to the human eye).
Rubredoxin in some biochemical reactions
- EC 1.14.15.2 camphor 1,2-monooxygenase [(+)-camphor, reduced-rubredoxin:oxygen oxidoreductase (1,2-lactonizing)]
- (+)-bornane-2,5-dione + reduced rubredoxin + O2 = 5-oxo-1,2-campholide + oxidized rubredoxin + H2O
- EC 1.14.15.3 alkane 1-monooxygenase (alkane, reduced-rubredoxin:oxygen 1-oxidoreductase)
- octane + reduced rubredoxin + O2 = 1-octanol + oxidized rubredoxin + H2O
- EC 1.15.1.2 superoxide reductase (rubredoxin:superoxide oxidoreductase)
- reduced rubredoxin + superoxide + 2 H+ = rubredoxin + H2O2
- EC 1.18.1.1 rubredoxin—NAD+ reductase (rubredoxin:NAD+ oxidoreductase)
- reduced rubredoxin + NAD+ = oxidized rubredoxin + NADH + H+
- EC 1.18.1.4 rubredoxin—NAD(P)+ reductase (rubredoxin:NAD(P)+ oxidoreductase)
- reduced rubredoxin + NAD(P)+ = oxidized rubredoxin + NAD(P)H + H+
Electron transfer rate
The electron exchange rate is accurately determined by standard kinetics measurements of visible absorption (490 nm) spectra.[4] The electron transfer rate has three parameters: electronic coupling, reorganization energy and free energy of reaction (ΔG°).
Protein mechanism and effects
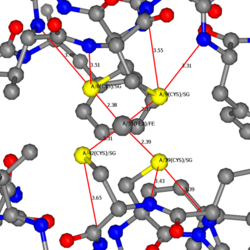

The electron transfer reaction of rubredoxin is carried out by a reversible Fe3+/Fe2+ redox coupling by the reduction of Fe3+ to Fe2+ and a gating mechanism caused by the conformational changes of Leu41.[5]
Upon the reduction of Fe3+ to Fe2+, the four Fe-S bond lengths increase and the amide-NH H-bonding to the S(Cys) become shortened. The reduced Fe2+ structure of rubredoxin results in a small increase in electrostatic stabilization of the amide-NH H-bonding to the S-Cys, leading to a lower reorganizational energy that allows faster electron transfer.[5]
A gating mechanism involving the conformational change of the Leu41's non-polar sidechain further stabilizes the Fe2+ oxidation state. A site-directed mutagenesis of Leu41 to Alanine shows a 50mV shift of the Fe3+/2+redox potential.[6] The substitution of the smaller CH3 shows that the Leu41 side chain stabilizes the Fe2+ oxidation state more than the Fe3+ oxidation state. The X-ray structure in the reduced Fe2+ state shows the Leu41 side chain adopting two different conformations with 40% in an "open conformation" and 60% in a "closed conformation".[5] The Leu41's non-polar side chain controls access to the redox site by adopting either an open or closed conformation. In the reduced Fe2+ state, the Leu41 side-chain faces away from Cys 9 Sγ, exposing the Cys 9 Sγ and increasing the polarity of the Fe3+ /Fe2+ center. [1] The lower Fe2+ cation change of the reduced state leaves a higher negative charge on the Cys 9 Sγ-donor which attracts water strongly. As a result, water is able to penetrate and form H-bonds with the Cys 9 Sγ thiolate that blocks the gate from closing, resulting in an open conformation. In contrast, the oxidized Fe3+ state produces a less negatively charged Cys 9 Sγ-donor that does not attract the water strongly. Without H-bonding of the water to the Cys 9 Sγ, the gate remains closed. Thus, the conformation of Leu41 is determined by the presence of water and the oxidation state of rubredoxin. The proximity of water to the [Fe(S-Cys)4] 2- active site stabilizes the higher net negative charge of the Fe2+ oxidation state.[5] The stabilization of the Fe2+ oxidation state shifts the reduction potential to a more positive E0 value.[5]
See also
References
- ↑ Liu, F; Geng, J; Gumper, RH; Barman, A; Ozarowski, A; Hamelberg, D; Liu, A (June 2015). "An iron reservoir to the catalytic metal: The rubredoxin iron in an extradiol dioxygenase". The Journal of Biological Chemistry 290 (25): 15621–15634. doi:10.1074/jbc.M115.650259. PMID 25918158. PMC 4505474. https://www.jbc.org/article/S0021-9258(20)35060-2/fulltext. Retrieved 6 February 2023.
- ↑ Calderon, RH; de Vitry, C; Wollman, FA; Niyogi, KK (February 2023). "Rubredoxin 1 promotes the proper folding of D1 and is not required for heme b559 assembly in Chlamydomonas photosystem II". The Journal of Biological Chemistry 299 (3). doi:10.1016/j.jbc.2023.102968. PMID 36736898. PMC 9986647. https://www.jbc.org/article/S0021-9258(23)00100-X/fulltext. Retrieved 6 February 2023.
- ↑ "A conserved rubredoxin is necessary for photosystem II accumulation in diverse oxygenic photoautotrophs". The Journal of Biological Chemistry 288 (37): 26688–26696. September 2013. doi:10.1074/jbc.M113.487629. PMID 23900844.
- ↑ "Electron transport to clostridial rubredoxin: kinetics of the reduction by hexaammineruthenium(II), vanadous and chromous ions". Proceedings of the National Academy of Sciences of the United States of America 71 (4): 1118–1122. April 1974. doi:10.1073/pnas.71.4.1118. PMID 4524621. Bibcode: 1974PNAS...71.1118J.
- ↑ 5.0 5.1 5.2 5.3 5.4 "Leucine 41 is a gate for water entry in the reduction of Clostridium pasteurianum rubredoxin". Protein Science 10 (3): 613–621. March 2001. doi:10.1110/gad.34501. PMID 11344329.
- ↑ "The unique hydrogen bonded water in the reduced form of Clostridium pasteurianum rubredoxin and its possible role in electron transfer". Journal of Biological Inorganic Chemistry 9 (4): 423–428. June 2004. doi:10.1007/s00775-004-0542-3. PMID 15067525.
Further reading
- Lippard, Stephen J.; Berg, Jeremy M. (1994). Principles of Bioinorganic Chemistry. University Science Books. ISBN 978-0-935702-72-9.
- Fraústo da Silva, J.J.R.; Williams, R.J.P. (2001). The biological chemistry of the elements: The inorganic chemistry of life (2nd ed.). Oxford University Press. ISBN 978-0-19-850848-9.
External links
- PDB: 1IRO – X-ray structure of rubredoxin from Clostridium pasteurianum
- PDB: 1VCX – Neutron diffraction structure of rubredoxin from Pyrococcus furiosus
- InterPro: IPR001052 – InterPro entry for rubredoxin
- A little iron-sulfur protein
 |