Biology:Troponin
Troponin, or the troponin complex, is a complex of three regulatory proteins (troponin C, troponin I, and troponin T) that are integral to muscle contraction[1] in skeletal muscle and cardiac muscle, but not smooth muscle. Measurements of cardiac-specific troponins I and T are extensively used as diagnostic and prognostic indicators in the management of myocarditis, myocardial infarction and acute coronary syndrome.[2] Blood levels of cardiac troponin (cTn) may be used as a diagnostic marker for stroke or other myocardial injury that is ongoing, although the sensitivity of this measurement is low.[3][4][5]
Function
Troponin is attached to the protein tropomyosin and lies within the groove between actin filaments in muscle tissue. In a relaxed muscle, tropomyosin blocks the attachment site for the myosin crossbridge, thus preventing contraction. When the muscle cell is stimulated to contract by an action potential, calcium channels open in the sarcoplasmic membrane and release calcium into the sarcoplasm. Some of this calcium attaches to troponin, which causes it to change shape, exposing binding sites for myosin (active sites) on the actin filaments. Myosin's binding to actin causes crossbridge formation, and contraction of the muscle begins.[6]
Smooth muscle does not have troponin.[7]
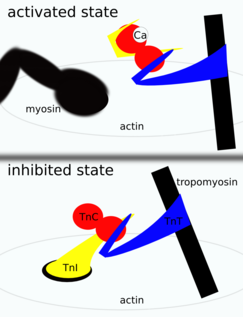
Physiology
Troponin is a component of thin filaments (along with actin and tropomyosin), and is the protein complex to which calcium binds to trigger the production of muscular force. Troponin has three subunits, TnC, TnI, and TnT, each playing a role in force regulation. . Under resting intracellular levels of calcium, tropomyosin covers the active actin sites to which myosin (a molecular motor organized in muscle thick filaments) binds in order to generate force. When calcium becomes bound to specific sites in the N-domain of TnC, a series of protein structural changes occurs,{{Citation needed|date=June 2013} led away from myosin-binding sites on actin, allowing myosin to attach to the thin filament and produce force and shorten the sarcomere.[citation needed] Individual subunits serve different functions:[8]
- Troponin C binds to calcium ions (Ca2+) to produce a conformational change in TnI
- Troponin T binds to tropomyosin, interlocking them to form a troponin-tropomyosin complex with thin filaments
- Troponin I binds to actin in thin myofilaments to hold the actin-tropomyosin complex in place. It inhibits the ATPase activise of actomyosin.
Inside the cardiac troponin complex, the strongest interaction between molecules is the cTnI–TnC binary complex, especially in the presence of Ca2+ (KA = 1.5 × 10−8 M−1).[9] TnC, forming a complex with cTnI, changes the conformation of cTnI molecule and shields part of its surface. According to the latest data cTnI is released in the blood stream of the patient in the form of binary complex with TnC or ternary complex with cTnT and TnC.[10] cTnI-TnC complex formation plays an important positive role in improving the stability of cTnI molecule. cTnI, which is extremely unstable in its free form, demonstrates significantly better stability in complex with TnC or in ternary cTnI-cTnT-TnC complex. It has been demonstrated that stability of cTnI in native complex is significantly better than stability of the purified form of the protein or the stability of cTnI in artificial troponin complexes combined from purified proteins.
Paralogs
Troponin is found in both skeletal muscle and cardiac muscle, but the specific versions of troponin differ between types of muscle. Different combinations of paralogous genes (vaguely called "isoforms", not to be confused with gene isoforms) are used to make the version of troponin seen in each type of muscle.
- Cardiac troponin C (cTnC) TNNC1 is expressed in cardiac and slow skeletal muscle, while skeletal troponin C (sTnC) TNNC2 is expressed in fast skeletal muscle.[11] sTnC has four calcium ion-binding sites, whereas in cTnC there are only three. The actual amount of calcium that binds to troponin has not been definitively established.[12] Because slow skeletal muscle and myocardium share a type, TnC is not used in diagnostics.[13]
- Mammals have three troponin I (TnI) genes: the cardiac (TNNI3, cTnI), the slow skeletal (TNNI1), and the fast skeletal (TNNI2).[14] Because the heart uses its own version, blood levels of cTnI is used as a clinical marker.[13]
- Mammals have three troponin T (TnT) genes: the cardiac (TNNT2, cTnT), the slow skeletal (TNNT1), and the fast skeletal (TNNT3).[14] Again, because the heart uses its own version, cTnT is used as a clinical marker.[13]
In addition to the myocardium, there are reports of CTnI and CTnT expression in the wall muscle coat of the vena cava and pulmonary veins, so they do not purely originate from the heart.[13] The expression of cTnT in skeletal tissue of patients with chronic skeletal muscle injuries has been described.[15]
Use in diagnostics
First cTnI[16] and later cTnT[17] were originally used as markers for cardiac cell death. Both proteins are now widely used to diagnose acute myocardial infarction (AMI), unstable angina, post-surgery myocardium trauma and some other related diseases with cardiac muscle injury. Both markers can be detected in patient's blood 3–6 hours after onset of the chest pain, reaching peak level within 16–30 hours. Elevated concentration of cTnI and cTnT in blood samples can be detected even 5–8 days after onset of the symptoms, making both proteins useful also for the late diagnosis of AMI.[18]
Detection
Cardiac troponin T and I are measured by immunoassay methods.[19][20]
- Due to patent regulations, a single manufacturer (Roche Diagnostics) distributes cTnT.
- A host of diagnostic companies make cTnI immunoassay methods available on many different immunoassay platforms.[20][21]
Cardiac troponin elevation following cardiac cell necrosis starts within 2–3 hours, peaks in approx. 24 hours, and persists for 1–2 weeks.[22]
Lateral-flow tests ("rapid diagnostic kits") have been developed for cardiac troponin I. The more basic kinds are qualitative and detect cTnI > 0.5 ng/L.[23] More advanced types allow a quantitative readout using colorimetry, electrochemical fluorescence, or a magnetic detector.[24]
cTnI/T as biomarkers
Cardiac conditions
Subtypes of troponin (cardiac I and T) are sensitive and specific indicators of heart muscle damage (myocardium). They are measured in the blood to differentiate between unstable angina and myocardial infarction (heart attack) in people with chest pain or acute coronary syndrome. A person who recently had a myocardial infarction has areas of damaged heart muscle and elevated cardiac troponin levels in the blood.[25] This can also occur in people with coronary vasospasm, a type of myocardial infarction involving severe constriction of the cardiac blood vessels. After a myocardial infarction troponins may remain high for up to 2 weeks.[26]
Cardiac troponins are a marker of all heart muscle damage, not just myocardial infarction, which is the most severe form of heart disorder. However, diagnostic criteria for raised troponin indicating myocardial infarction is currently set by the WHO at a threshold of 2 μg/L or higher. Critical levels of other cardiac biomarkers are also relevant, such as creatine kinase.[27] Severe tachycardia (for example due to supraventricular tachycardia) in an individual with normal coronary arteries can also lead to increased troponins for example, it is presumed due to increased oxygen demand and inadequate supply to the heart muscle. Coronary artery stent placement can also cause immediate post-procedure elevated serum cardiac troponin levels. This can be problematic in a clinical setting as troponin values and guidance protocols become subject to more thoughtful interpretation. Essentially, making it difficult for a clinician to use troponin elevation diagnostically in this patient group. After stenting and related PCI procedures, troponin levels do return to standard levels once the stent has 'settled' and is no longer causing localized cardiac muscle inflammation.[28]
In patients with non-severe asymptomatic aortic valve stenosis and no overt coronary artery disease, the increased troponin T (above 14 pg/mL) was found associated with an increased 5-year event rate of ischemic cardiac events (myocardial infarction, percutaneous coronary intervention, or coronary artery bypass surgery).[29]
Non-cardiac conditions
The distinction between cardiac and non-cardiac conditions is somewhat artificial; the conditions listed below are not primary heart diseases, but they exert indirect effects on the heart muscle.[citation needed] Other conditions that directly or indirectly lead to heart muscle damage and death can also increase troponin levels, such as kidney failure.[30][31]
Cardiac troponins are increased in around 40% of patients with critical illnesses such as sepsis. There is an increased risk of mortality and length of stay in the intensive-care unit in these patients.[32] In severe gastrointestinal bleeding, there can also be a mismatch between oxygen demand and supply of the myocardium.[citation needed]
Hypertension
In hypertensive disorders of pregnancy such as preeclampsia, elevated troponin levels indicate some degree of myofibrillary damage.[33][34]
Toxicity
Cardiac troponin T and I can be used to monitor drug and toxin-induced cardiomyocyte toxicity. .[35]
Chemotherapy agents can exert toxic effects on the heart (examples include anthracycline, cyclophosphamide, 5-fluorouracil, and cisplatin). Several toxins and venoms can also lead to heart muscle injury (scorpion venom, snake venom, and venom from jellyfish and centipedes). Carbon monoxide poisoning or cyanide poisoning can also be accompanied by the release of troponins due to hypoxic cardiotoxic effects. Cardiac injury occurs in about one-third of severe CO poisoning cases, and troponin screening is appropriate in these patients.[36][37]
Lung
In both primary pulmonary hypertension, pulmonary embolism, and acute exacerbations of chronic obstructive pulmonary disease (COPD), right ventricular strain results in increased wall tension and may cause ischemia. Of course, patients with COPD exacerbations might also have concurrent myocardial infarction or pulmonary embolism, so care has to be taken to attribute increased troponin levels to COPD.[citation needed]
Kidney
People with end-stage kidney disease can have chronically elevated cardiac troponin T levels, which are linked to a poorer prognosis.[38][39] Troponin I is less likely to be falsely elevated.[38]
Skeletal muscle
Strenuous endurance exercise such as marathons or triathlons can lead to increased cardiac troponin levels in up to one-third of subjects, but it is not linked to adverse health effects in these competitors.[40][41][42]
High cardiac troponin T levels have also been reported in patients with inflammatory muscle diseases such as polymyositis or dermatomyositis.[43][44] Troponins are also increased in rhabdomyolysis.
COVID-19
In 2020, it was found that patients with severe COVID-19 had higher cardiac troponin I levels compared to those with milder disease.[45]
Prognostic use
Elevated cardiac troponin levels are prognostically important in many of the conditions in which they are used for diagnosis.[46]
In a community-based cohort study indicating the importance of silent cardiac damage, cardiac troponin I has been shown to predict mortality and first coronary heart disease event in men free from cardiovascular disease at baseline.[47] In people with stroke, elevated blood troponin levels are not a useful marker to detect the condition.[5]
Evolution
There are five groups of TNNI in vertebrates, the extra two known as TNNI4 and TNNI5 (only found in non-amniotes). They are more similar to TNNI1 than to TNNI2 and TNNI3. Likewise, there are four groups of TNNT in vertebrates, with TNNT4 (only found in cartilaginous fishes, ray-finned fishes and lungfish) most closely resembling TNNT2.[13] In most vertebrates and some non-vertebrate chordates, TNNI and TNNT genes tend to occur in pairs next to each other. It is likely that an ancestor to vertebrates had one such pair, which was duplicated into four during the two rounds of whole-genome duplication in early vertebrate evolution, with TNNT5 arising as an additional tandem dupliaction of TNNT4.[13]
Most vertebrates express TNNI1 and/or TNNI5 in the heart, whereas adult tetrapods (such as mammals) use TNNI3. Embryonic mammals use TNNI1 in the heart. In humans and teterapods in general, TNNI3 differ from the other TnI genes by having an N-termianl extension; a strikingly similar extension is found in the TNNI5 of cartilaginous, non-teleost ray-finned, and sarcopterygian fishes. Ray finned fishes do not have TNNI3 at all. Substituting TNNI3 for TNNI1 may confer increased tolerance to acidosis.[13]
Considering the similarity between TNNI and TNNT, the gene pair may have been the result of an even more ancient tandem duplication event.[48] If this is the case, such a duplication must have happened early in animal evolution (more than 700 million years ago), as genes that encode for all three subunits of troponin have been found in every invertebrate phyla except the Cnidaria (or equivalently, all Bilaterian phyla). The ancestral gene pair most closely resembled TNNI2-TNNT3.[49]
See also
- Calcium-binding protein
- Troponin C
- Troponin I
- Troponin T
References
- ↑ "troponin" at Dorland's Medical Dictionary
- ↑ "Troponin - Understand the Test & Your Results". https://labtestsonline.org/tests/troponin.
- ↑ Strandberg, Love S.; Roos, Andreas; Holzmann, Martin J. (2021-01-01). "Stable high-sensitivity cardiac troponin T levels and the association with frailty and prognosis in patients with chest pain" (in en). American Journal of Medicine Open 1-6. doi:10.1016/j.ajmo.2021.100001. ISSN 2667-0364. PMID 39036625.
- ↑ Thygesen, Kristian; Alpert, Joseph S.; Jaffe, Allan S.; Chaitman, Bernard R.; Bax, Jeroen J.; Morrow, David A.; White, Harvey D. (2018-10-30). "Fourth Universal Definition of Myocardial Infarction (2018)" (in en). Journal of the American College of Cardiology 72 (18): 2231–2264. doi:10.1016/j.jacc.2018.08.1038. ISSN 0735-1097. PMID 30153967.
- ↑ 5.0 5.1 Kerr, Gillian; Ray, Gautamananda; Wu, Olivia; Stott, David J.; Langhorne, Peter (2009). "Elevated troponin after a stroke: a systematic review". Cerebrovascular Diseases 28 (3): 220–226. doi:10.1159/000226773. ISSN 1421-9786. PMID 19571535.
- ↑ Betts, J. Gordon (2013). Anatomy & physiology. Peter Desaix, Eddie Johnson, Jody E. Johnson, Oksana Korol, Dean Kruse, Brandon Poe. Houston, Texas. ISBN 978-1-947172-04-3. OCLC 898069394.
- ↑ Troponins at eMedicine
- ↑ Gomes, A.V; Potter, J.D.; Szczesna-Cordary, D. (2002). "The role of Troponin in muscle contraction". Life (54): 323–333.
- ↑ "Stepwise subunit interaction changes by mono- and bisphosphorylation of cardiac troponin I". Biochemistry 37 (39): 13516–13525. 1998. doi:10.1021/bi980280j. PMID 9753437.
- ↑ "Troponin I is released in bloodstream of patients with acute myocardial infarction not in free form but as complex". Clin. Chem. 43 (8): 1379–1385. 1997. doi:10.1093/clinchem/43.8.1379. PMID 9267317.
- ↑ Marston, S.B.; Redwood, C.S. (2003). "Modulation of thin filament activation by breakdown or isoform switching of thin filament Proteins". Circ. Res. 93 (12): 1170–1178. doi:10.1161/01.RES.0000105088.06696.17. PMID 14670832.
- ↑ Gillis, Todd E.; Marshall, Christian R.; Tibbits, Glen F. (December 2007). "Functional and evolutionary relationships of troponin C" (in en). Physiological Genomics 32 (1): 16–27. doi:10.1152/physiolgenomics.00197.2007. ISSN 1094-8341. PMID 17940202. https://www.physiology.org/doi/10.1152/physiolgenomics.00197.2007.
- ↑ 13.0 13.1 13.2 13.3 13.4 13.5 13.6 Michailovich Chaulin, A (2023). "Diagnostic Role and Methods of Detection of Cardiac Troponins: An Opinion from Historical and Current Points of View.". Current cardiology reviews 19 (1): e100622205865. doi:10.2174/1573403X18666220610164946. PMID 35702796.
- ↑ 14.0 14.1 Joyce, W; Ripley, DM; Gillis, T; Black, AC; Shiels, HA; Hoffmann, FG (4 January 2023). "A Revised Perspective on the Evolution of Troponin I and Troponin T Gene Families in Vertebrates.". Genome biology and evolution 15 (1). doi:10.1093/gbe/evac173. PMID 36518048.
- ↑ "Cardiac troponins". J. Emerg. Med. 23 (1): 57–65. 2002. doi:10.1016/S0736-4679(02)00463-8. PMID 12217473.
- ↑ "Cardiac-specific troponin-I radioimmunoassay in the diagnosis of acute myocardial infarction". Am. Heart J. 113 (6): 1333–1344. 1987. doi:10.1016/0002-8703(87)90645-4. PMID 3591601.
- ↑ "Enzyme linked immunoassay of cardiac troponin T for the detection of acute myocardial infarction in patients". J Mol Cell Cardiol 21 (12): 1349–1353. 1989. doi:10.1016/0022-2828(89)90680-9. PMID 2632816.
- ↑ Hamm CW. (2001). "Acute coronary syndromes. The diagnostic role of troponins". Thromb. Res. 103 (1): 63–69. doi:10.1016/S0049-3848(01)00299-7. PMID 11567671.
- ↑ "Cardiac troponin assays: a view from the clinical chemistry laboratory". Circulation 116 (18): e501–4. October 2007. doi:10.1161/CIRCULATIONAHA.107.722975. PMID 17967982.
- ↑ 20.0 20.1 "Measurement of cardiac troponins". Ann. Clin. Biochem. 38 (Pt 5): 423–49. September 2001. doi:10.1258/0004563011901109. PMID 11587122.
- ↑ Smith, Joe S.; Varga, Anita; Schober, Karsten E. (2020). "Comparison of Two Commercially Available Immunoassays for the Measurement of Bovine Cardiac Troponin I in Cattle with Induced Myocardial Injury". Frontiers in Veterinary Science 7: 531. doi:10.3389/fvets.2020.00531. PMID 33062647.
- ↑ Patil, H.; Vaidya, O.; Bogart, D. (2011). "A Review of Causes and Systemic Approach to Cardiac Troponin Elevation". Clin Cardiol 34 (12): 723–728. doi:10.1002/clc.20983. PMID 22120679.
- ↑ Schilling, Ellie; Sandman, Joy; Sait, Rida; Stich, April; Baryeh, Kwaku; Blankson, Gifty (July 2023). "An Undergraduate Independent Study Project: Using a Lateral Flow Assay to Detect Troponin". Journal of College Science Teaching 52 (6): 76–81. doi:10.1080/0047231X.2023.12315870.
- ↑ Mohammadinejad, Arash; Nooranian, Samin; Kazemi Oskuee, Reza; Mirzaei, Sepideh; Aleyaghoob, Ghazaleh; Zarrabi, Ali; Selda Gunduz, Emine; Nuri Ertas, Yavuz et al. (2 October 2024). "Development of Lateral Flow Assays for Rapid Detection of Troponin I: A Review". Critical Reviews in Analytical Chemistry 54 (7): 1936–1950. doi:10.1080/10408347.2022.2144995.
- ↑ "Cardiac-specific troponin I levels to predict the risk of mortality in patients with acute coronary syndromes". N. Engl. J. Med. 335 (18): 1342–9. October 1996. doi:10.1056/NEJM199610313351802. PMID 8857017.
- ↑ Amsterdam, E. A.; Wenger, N. K.; Brindis, R. G.; Casey, D. E.; Ganiats, T. G.; Holmes, D. R.; Jaffe, A. S.; Jneid, H. et al. (23 September 2014). "2014 AHA/ACC Guideline for the Management of Patients With Non-ST-Elevation Acute Coronary Syndromes: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines". Circulation 130 (25): e344–e426. doi:10.1161/CIR.0000000000000134. PMID 25249585. http://circ.ahajournals.org/content/circulationaha/130/25/e344.full.pdf.
- ↑ "Common and rare variants associating with serum levels of creatine kinase and lactate dehydrogenase". Nature Communications 7. February 2016. doi:10.1038/ncomms10572. PMID 26838040. Bibcode: 2016NatCo...710572K.
- ↑ Ferreira, Roberto Muniz; de Souza e Silva, Nelson Albuquerque; Salis, Lúcia Helena Alvares; Maia, Paula Dias; Horta, Lucas Felipe Bastos (2017-05-01). "Troponin I elevation after elective percutaneous coronary interventions: Prevalence and risk factors". Indian Heart Journal 69 (3): 322–324. doi:10.1016/j.ihj.2016.11.319. ISSN 0019-4832. PMID 28648422.
- ↑ Hadziselimovic, Edina; Greve, Anders M.; Sajadieh, Ahmad; Olsen, Michael H.; Kesäniemi, Y. Antero; Nienaber, Christoph A.; Ray, Simon G.; Rossebø, Anne B. et al. (April 2023). "Association of high-sensitivity troponin T with outcomes in asymptomatic non-severe aortic stenosis: a post-hoc substudy of the SEAS trial". The Lancet eClinicalMedicine 58. doi:10.1016/j.eclinm.2023.101875. ISSN 2589-5370. PMID 36915288.
- ↑ "Raised cardiac troponins". BMJ 328 (7447): 1028–9. May 2004. doi:10.1136/bmj.328.7447.1028. PMID 15117768.
- ↑ "Use and interpretation of cardiac troponins in the ED". Am. J. Emerg. Med. 26 (3): 331–41. March 2008. doi:10.1016/j.ajem.2007.05.031. PMID 18358946.
- ↑ "Elevated cardiac troponin measurements in critically ill patients". Arch. Intern. Med. 166 (22): 2446–54. 2006. doi:10.1001/archinte.166.22.2446. PMID 17159009.
- ↑ "Cardiac troponin I in pre-eclampsia and gestational hypertension". BJOG 107 (11): 1417–20. November 2000. doi:10.1111/j.1471-0528.2000.tb11658.x. PMID 11117772.
- ↑ Morton A (July 2004). "Raised cardiac troponins: troponin is raised in pre-eclampsia". BMJ 329 (7457): 111. doi:10.1136/bmj.329.7457.111-a. PMID 15242925.
- ↑ Gaze DC, Collinson PO; Collinson, PO (December 2005). "Cardiac troponins as biomarkers of drug- and toxin-induced cardiac toxicity and cardioprotection". Expert Opin Drug Metab Toxicol 1 (4): 715–25. doi:10.1517/17425255.1.4.715. PMID 16863435.
- ↑ "Myocardial injury and long-term mortality following moderate to severe carbon monoxide poisoning". JAMA 295 (4): 398–402. January 2006. doi:10.1001/jama.295.4.398. PMID 16434630.
- ↑ "Cardiovascular manifestations of moderate to severe carbon monoxide poisoning". J. Am. Coll. Cardiol. 45 (9): 1513–6. May 2005. doi:10.1016/j.jacc.2005.01.044. PMID 15862427.
- ↑ 38.0 38.1 "Troponin I and T levels in renal failure patients without acute coronary syndrome: a systematic review of the literature". Can. J. Cardiol. 20 (12): 1212–8. October 2004. PMID 15494773.
- ↑ "Prognostic value of troponin T and I among asymptomatic patients with end-stage renal disease: a meta-analysis". Circulation 112 (20): 3088–96. November 2005. doi:10.1161/CIRCULATIONAHA.105.560128. PMID 16286604.
- ↑ "Cardiac troponin T and I, echocardiographic [correction of electrocardiographic] wall motion analyses, and ejection fractions in athletes participating in the Hawaii Ironman Triathlon". Am. J. Cardiol. 83 (7): 1085–9. April 1999. doi:10.1016/S0002-9149(99)00020-X. PMID 10190525.
- ↑ "Plasma levels of cardiac troponin I after prolonged strenuous endurance exercise". Am. J. Cardiol. 87 (3): 369–71, A10. February 2001. doi:10.1016/S0002-9149(00)01382-5. PMID 11165984.
- ↑ "Clinical significance of increased cardiac troponins T and I in participants of ultra-endurance events". Am. J. Cardiol. 94 (5): 696–8. September 2004. doi:10.1016/j.amjcard.2004.05.050. PMID 15342317.
- ↑ "Serum cardiac troponin T in polymyositis/dermatomyositis". Lancet 340 (8821): 726. September 1992. doi:10.1016/0140-6736(92)92262-E. PMID 1355820.
- ↑ "Cardiac troponin and beta-type myosin heavy chain concentrations in patients with polymyositis or dermatomyositis". Clin. Chim. Acta 306 (1–2): 27–33. April 2001. doi:10.1016/S0009-8981(01)00392-8. PMID 11282091.
- ↑ "Cardiac troponin I in patients with coronavirus disease 2019 (COVID-19): Evidence from a meta-analysis". Prog Cardiovasc Dis 63 (3): 390–391. 2020. doi:10.1016/j.pcad.2020.03.001. PMID 32169400.
- ↑ Mannu, GS (August 2014). "The non-cardiac use and significance of cardiac troponins". Scottish Medical Journal 59 (3): 172–8. doi:10.1177/0036933014540090. PMID 24934496.
- ↑ "Troponin I as a predictor of coronary heart disease and mortality in 70-year-old men: a community-based cohort study". Circulation 113 (8): 1071–8. February 2006. doi:10.1161/CIRCULATIONAHA.105.570762. PMID 16490824.
- ↑ Chong, SM; Jin, JP (May 2009). "To investigate protein evolution by detecting suppressed epitope structures.". Journal of molecular evolution 68 (5): 448-60. doi:10.1007/s00239-009-9202-0. PMID 19365646.
- ↑ Cao, T; Thongam, U; Jin, JP (15 May 2019). "Invertebrate troponin: Insights into the evolution and regulation of striated muscle contraction.". Archives of biochemistry and biophysics 666: 40-45. doi:10.1016/j.abb.2019.03.013. PMID 30928296.
External links
- Troponin at the US National Library of Medicine Medical Subject Headings (MeSH)
- Troponins at Lab Tests Online
 |
