Biology:Vaginal flora
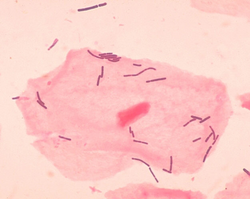
Vaginal flora, vaginal microbiota or vaginal microbiome are the microorganisms that colonize the vagina. They were discovered by the German gynecologist Albert Döderlein in 1892[1] and are part of the overall human flora. The amount and type of bacteria present have significant implications for an individual's overall health.[2] The primary colonizing bacteria of a healthy individual are of the genus Lactobacillus,[3] such as L. crispatus, and the lactic acid they produce is thought to protect against infection by pathogenic species.[4]
Lactobacilli
The primary colonizing bacteria of a healthy individual are of the genus Lactobacillus (90–95%), the most common being L. crispatus, L. iners, L. jensenii, and L. gasseri.[5] Since the first description of lactobacilli by Döderlein, lactobacilli have been generally considered the gatekeepers of the vaginal ecosystem. Lactobacilli have been shown to inhibit in vitro growth of pathogenic microorganisms, e.g. Bacteroides fragilis, Escherichia coli, Gardnerella vaginalis, Mobiluncus spp., Neisseria gonorrhoeae, Peptostreptococcus anaerobius, Prevotella bivia and Staphylococcus aureus. It is generally accepted that this is achieved mainly through the action of lactic acid.[6][7][8][9] Moreover, lactobacilli normally help to prevent long-term colonization of the vagina by adhering to vaginal epithelial cells. This usually reduces pathogens from infecting to the vaginal epithelium.[10]
Next to lactic acid production and competition for adherence, other antagonistic mechanisms include hydrogen peroxide (a broad-spectrum antimicrobial) and bacteriocins (target-specific antimicrobials) production.[11][12]
pH and lactic acid

Low pH is generally accepted to be the main mechanism controlling the composition of the vaginal microflora. Although the lactic acid produced by lactobacilli contributes to the vaginal acidity, it is still not proven to be the primary source of low vaginal pH, but the fact remains that most lactobacilli thrive best at a pH < 3.5 .[13][14][15] Normal vaginal pH is considered to be under 4.5 with a range of 3.8 to 4.4.[5]
Hydrogen peroxide
Production of hydrogen peroxide (H2O2) is a proposed mechanism for bacterial antagonism in vitro,[16][17][18] inhibiting growth of microorganisms via direct interaction or via human myeloperoxidase.[19][20][21] However, in vivo, the role of peroxide is uncertain. Under optimal anaerobic growth conditions, physiological concentrations of lactic acid inactivated the BV-associated pathogens without affecting the vaginal lactobacilli.[12][22] Although the hydrogen peroxide production of lactobacilli has been considered as an important antimicrobial component, contributing to the colonization resistance provided by lactobacilli,[11][23] and although there seems to be a link between H2O2-producing lactobacilli and normal vaginal microflora, recent data do not support this role for H2O2.[12][22]
Experimentally, hydrogen peroxide-producing lactobacilli have been shown to inactivate HIV-1, herpes simplex virus type 2 (HSV-2), Trichomonas vaginalis, G. vaginalis, P. bivia and E. coli. O'Hanlon[12] and Baeten[24] found that 96% of Lactobacillus species from a healthy vaginal ecosystem produced H2O2 (L. jensenii and L. vaginalis produce the highest levels of H2O2),[11][25] whereas only 6% of the lactobacilli recovered from women with BV produced H2O2.[19] In agreement with this, L. iners, most frequently associated with disturbed vaginal microflora,[26][27] is a poor producer of H2O2.[28][29] Vaginal colonization by H2O2-producing lactobacilli has been associated with a decrease in the occurrence of bacterial vaginosis (BV).[30] However, more recently O'Hanlon et al.[22] demonstrated that cervicovaginal fluid and semen have a significant H2O2-blocking activity and they later [12] demonstrated that physiological concentrations of H2O2 below 100 μM fail to inactivate any of the 17 tested BV-associated bacteria, e.g. A. vaginae, G. vaginalis, Mobiluncus spp., P. bivia, P. corporis, Mycoplasma hominis, even in the presence of human myeloperoxidase, known to increase the microbicidal activity of H2O2.[12] Only supraphysiologic concentrations of exogenous H2O2 (0.34% w/v, 100 mM) were sufficient to inactivate BV-associated bacteria at which concentration it more potently inactivated vaginal lactobacilli (L. crispatus, L. gasseri, L. iners and L. jensenii). A concentration of 100 mM H2O2 is approximately 50-fold higher than lactobacilli are capable of producing even under optimal aerobic, low-antioxidant conditions, and approximately 5,000-fold higher than the estimated H2O2 concentration in vivo. Even more remarkable, the addition of only 1% vaginal fluid blocked the microbicidal activity of 1 M H2O2. Possible explanations may be that cervicovaginal fluid and semen contain proteins, glycoproteins, polysaccharides, lipids, and other molecules with the potential to react with and inactivate H2O2. In addition, the vagina is hypoxic most of the time, whereas lactobacilli require oxygen to produce hydrogen peroxide. It is also remarkable that catalase, which provides bacteria protection against toxic H2O2, is absent in lactobacilli,[19][31] and as such they would be unprotected against their own H2O2 production. Consequently, hydrogen peroxide production may not be the major mechanism of defense.
Bacteriocins
Vaginal lactobacilli produce antimicrobial peptides, i.e. bacteriocins such as lactocin 160 and crispasin.[13] with inhibitory activity ranging from narrow (closely related Lactobacillus species) to broad (diverse groups of bacteria, including G. vaginalis and P. bivia),[8] and bacteriocin-like substances, with a broader spectrum of activity than bacteriocins (e.g. a heat-resistant peptide produced by Ligilactobacillus salivarius subsp. salivarius CRL 1328). Several studies have indicated that the activity of bacteriocins is favored by low pH.
The inhibitory substances produced by vaginal Lactobacillus is a primary factor in protecting the vaginal microbiota, with organic acids, bacteriocins, and hydrogen peroxide. These act synergistically against infection by pathogens. Not all Lactobacillus spp. and not all strains within one Lactobacillus species exhibit all 3 mechanisms.[13] Lactobacillus species differ in premenopausal women, i.e. L. crispatus, L. jensenii, L. iners, L. gasseri (and possibly Limosilactobacillus vaginalis), as assessed through cultivation-dependent and cultivation-independent techniques.[26][27][28][32] Vaginal lactobacilli have been shown to display a pronounced vaginotropism, and their pili act as ligands for attachment to receptors of vaginal epithelial cells. The limited number of Lactobacillus spp. found in the human vagina is remarkable, which leads to the possibility that there are host factors that select for specific organisms, that these species have unusual characteristics that allow them to successfully colonize the vagina, or both .[33] However, the vaginotropism, does not only apply to this selected group of lactobacilli that stand for a healthy vagina, but also for the bacterial species associated with BV.[34] The microbiota detected in the human genital and gut econiche do not appear to grow outside their host and probably are likely to rely on the close contact between parents and their children for transmission,[34] e.g. mother to neonate transmission of genital microflora, most probably also with gut microflora homogenously distributed over the baby's body including skin, the oral cavity, nasopharynx, and feces.[35]
Bacterial vaginosis
Bacterial vaginosis (BV) is associated with the presence of Gardnerella vaginalis and Peptostreptococcus anaerobius[36] and a decrease in the number of Lactobacillus species that comprise the healthy vaginal microbiota.[37][38][39][40] BV is characterized by overgrowth of opportunistic bacteria and a decrease in Lactobacillus species. BV can present itself as symptomatic or asymptomatic. Symptoms include vaginal malodor, increased vaginal pH, and vaginal itching.[41] There is no one known cause as to why BV occurs but it is typically associated with abnormal vaginal discharge (Coudray & Madhivanan, 2020). To diagnose BV in a clinical setting, an Amsel's criteria is used. This means that a patient must have an increased homogeneous thin vaginal discharge, pH secretion higher than 4.5, amine odor when secretions are exposed to hydroxide 10% solution, or clue cells visible on a wet mount. Another way to diagnose BV that is less often used in a clinical setting is a Nugent score. To obtain a Nugent score, gram stain techniques are carried out and it characterizes the bacterial morphology on a scale. A score greater than 7 is associated with BV, 4-6 is an intermediate abnormality, and 0-3 is normal.[42]
Typically, only women with symptoms receive treatment. Treatments include antibiotics, specifically metronidazole and clindamycin (Hay, 2010). As rates of recurrence are high,[43] women may also turn to treatments such as probiotics or boric acid suppositories.[44][45][46] Boric acid has been used to treat BV since the 1800s.[47] Yet due to the compound's role in killing pests, the prescription of boric acid by healthcare providers to treat BV may be met with dissatisfaction; science journalist Rachel E. Gross was inspired to write the book Vagina Obscura after receiving 'what was "basically rat poison"' from her gynecologist and accidentally ingesting it midway through her course of treatment, which led her to consider the extent of the medical establishment's neglect of women's health.[48]
Studies have shown a correlation between vaginal microbiota and BV development through 16S RNA sequencing. Community state types (CSTs) are used to identify variation between a healthy vaginal microbiota and vaginal microbiota of someone with BV. Most CSTs in a healthy vaginal microbiome consist of different species of Lactobacillus. Studies found that L.iners is the dominant species in BV and L.crispatus is the dominant species in healthy vaginal microbiomes.[49]
Other microbiota

Healthy, normal vaginal microbiota that is dominated by lactobacilli may differ among some ethnic groups. Non-pathogenic vaginal species are part of the normal microbiota of some women.[26][50] Several studies have demonstrated that a significant proportion (7–33%) of healthy asymptomatic women (especially black and Hispanic women)[51] lack appreciable numbers of Lactobacillus species in the vagina,[33][52] and instead have a vaginal microbiota that consist of other lactic acid-producing bacteria, i.e. species from the genera Atopobium, Leptotrichia, Leuconostoc, Megasphaera, Pediococcus, Streptococcus and Weissella,[32][33][51] All ethnic populations have vaginal microflora communities containing lactic acid producing bacteria.[33][51] This implies that not all communities may be equally resilient, so that if the resilience of a vaginal community is low then transitory changes in the structure of these communities may occur more readily in response to disturbances of various kinds, including menses, sexual intercourse, douching and contraceptive practices. These differences in the structure and composition of microbial communities may underlie well-known differences in the susceptibility of women in these racial groups to BV and various vaginal infections.[51][53][54] Though vaginal microflora may be disturbed, and lactic acid is produced by other species,[55] vaginal pH and acid production establish normal vaginal microbiota. The pH further decreases during pregnancy.[56]
Other vaginal bacterial species

Other bacterial species are frequently found in the vagina, such as the Gram positive cocci: Atopobium vaginae, Peptostreptococcus spp., Staphylococcus spp., Streptococcus spp., and Bacteroides spp., Fusobacterium spp., Gardnerella vaginalis, Mobiluncus, Prevotella spp., and Gram-negative enteric organisms, such as Escherichia coli.[26][27] Mycoplasma and Ureaplasma are frequently found in the vagina. Some of the obligate and facultative anaerobic bacteria are associated with BV.[52] Neisseria gonorrhoeae is found in the vagina and is considered a pathogen that is sexually transmitted.[57]
Pregnancy
The effect of tampon use on vaginal flora is debated, but application of tampons appears not to significantly modify the balance of bacterial presence.[58] Pregnancy alters the microbiota with a reduction in species/genus diversity.[37]
Disease prevention
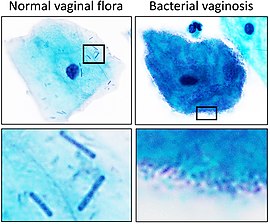
A healthy vaginal microbiome aids in the prevention of bacterial vaginosis, yeast infections and other possible problems by maintaining an acidic pH (< 4.5) that is unfavourable for the growth of common pathogens, such as Gardnerella vaginalis. The lactobacilli present in a healthy vaginal microbiome also occupy the ecological niche that would otherwise be available for exploitation by pathogenic organisms. However, harmful bacteria or an imbalance in bacteria can lead to infection.[59]
There are many factors that contribute to the composition of vaginal microbiota including stress, age, lifestyle, diet, gut microbiota, antibiotics, probiotics, and vaginal infections.
Lifestyle can impact microbiota by means of personal hygiene. Research suggests that vaginal douching is associated with increased risk of BV. It also shows that sexual practices that include multiple sexual partners can increase BV or throw off stable microbiota composition.[60] Smoking and alcohol consumption can also increase likelihood of BV development because it indirectly impacts the metabolite production profile.[61] There is also research supporting the idea that increased stress levels can have an impact on the balance of vaginal lactobacilli by elevating cortisol levels, reducing vaginal glycogen, lowering lactobacilli, elevating vaginal pH, and increasing proinflammatory response.[60]
It is becoming more commonly known how our diets can impact our gut microbiota, but it can have the same impact on vaginal microbiota. Research shows that diets rich in nutrients with low glycemic index and lower fat intake may be associated with a decreased chance of getting BV.[60]
Research
Investigations have found that the presence of lactobacilli in the vagina is associated with a lower incidence of sexually transmitted infections.[62][63]
See also
- Human Microbiome Project
- Lactic acid bacteria
- List of microbiota species of the lower reproductive tract of women
- Skin flora
- Vaginal microbiota in pregnancy
References
- ↑ "Albert und Gustav Döderlein – ein kritischer Blick auf zwei besondere Lebensläufe deutscher Ordinarien" (in German). Zentralblatt für Gynäkologie 128 (2): 56–9. April 2006. doi:10.1055/s-2006-921412. PMID 16673245.
- ↑ "Endometrial microbes and microbiome: Recent insights on the inflammatory and immune "players" of the human endometrium". American Journal of Reproductive Immunology 80 (6). December 2018. doi:10.1111/aji.13065. PMID 30375712.
- ↑ "Vaginal lactobacillus flora of healthy Swedish women". Journal of Clinical Microbiology 40 (8): 2746–9. August 2002. doi:10.1128/JCM.40.8.2746-2749.2002. PMID 12149323.
- ↑ "Bacterial flora of the female genital tract: function and immune regulation". Best Practice & Research. Clinical Obstetrics & Gynaecology 21 (3): 347–54. June 2007. doi:10.1016/j.bpobgyn.2006.12.004. PMID 17215167.
- ↑ 5.0 5.1 Tidbury, Fiona Damaris; Langhart, Anita; Weidlinger, Susanna; Stute, Petra (2020-10-06). "Non-antibiotic treatment of bacterial vaginosis—a systematic review". Archives of Gynecology and Obstetrics (Springer Science and Business Media LLC) 303 (1): 37–45. doi:10.1007/s00404-020-05821-x. ISSN 0932-0067. PMID 33025086.
- ↑ "The role of acidification in the inhibition of Neisseria gonorrhoeae by vaginal lactobacilli during anaerobic growth". Annals of Clinical Microbiology and Antimicrobials 10: 8. February 2011. doi:10.1186/1476-0711-10-8. PMID 21329492.
- ↑ "In vitro inhibitory activity of human vaginal lactobacilli against pathogenic bacteria associated with bacterial vaginosis in Kenyan women". Anaerobe 16 (3): 210–5. June 2010. doi:10.1016/j.anaerobe.2009.11.002. PMID 19925874. http://ir-library.ku.ac.ke/handle/123456789/5929.
- ↑ 8.0 8.1 "Vaginal lactobacilli inhibiting growth of Gardnerella vaginalis, Mobiluncus and other bacterial species cultured from vaginal content of women with bacterial vaginosis". Acta Pathologica et Microbiologica Scandinavica, Section B 94 (6): 399–403. December 1986. doi:10.1111/j.1699-0463.1986.tb03074.x. PMID 3494379.
- ↑ Strus, M.; Malinowska, M.; Heczko, P.B. (2002). "In vitro antagonistic effect of Lactobacillus on organisms associated with bacterial vaginosis". J. Reprod. Med. 47 (1): 41–46. PMID 11838310.
- ↑ "Role played by lactobacilli in controlling the population of vaginal pathogens". Microbes and Infection 2 (5): 543–6. April 2000. doi:10.1016/s1286-4579(00)00313-0. PMID 10865199.
- ↑ 11.0 11.1 11.2 "Biosynthesis and degradation of H2O2 by vaginal lactobacilli". Applied and Environmental Microbiology 76 (2): 400–5. January 2010. doi:10.1128/AEM.01631-09. PMID 19948869. Bibcode: 2010ApEnM..76..400M.
- ↑ 12.0 12.1 12.2 12.3 12.4 12.5 "In vaginal fluid, bacteria associated with bacterial vaginosis can be suppressed with lactic acid but not hydrogen peroxide". BMC Infectious Diseases 11. July 2011. doi:10.1186/1471-2334-11-200. PMID 21771337.
- ↑ 13.0 13.1 13.2 "Defense factors of vaginal lactobacilli". American Journal of Obstetrics and Gynecology 185 (2): 375–9. August 2001. doi:10.1067/mob.2001.115867. PMID 11518895.
- ↑ "Contemporary perspectives on vaginal pH and lactobacilli". American Journal of Obstetrics and Gynecology 204 (2): 120.e1–5. February 2011. doi:10.1016/j.ajog.2010.07.010. PMID 20832044.
- ↑ "Emerging role of lactobacilli in the control and maintenance of the vaginal bacterial microflora". Reviews of Infectious Diseases 12 (5): 856–72. 1990. doi:10.1093/clinids/12.5.856. PMID 2237129.
- ↑ "Hydrogen peroxide formation by lactobacilli and its effect on Staphylococcus aureus". Journal of Dairy Science 51 (10): 1568–72. October 1968. doi:10.3168/jds.s0022-0302(68)87232-7. PMID 5682478.
- ↑ "The inhibitory action of saliva on the diphtheria bacillus: hydrogen peroxide, the inhibitory agent produced by salivary streptococci". The Journal of Infectious Diseases 88 (1): 81–5. 1951. doi:10.1093/infdis/88.1.81. PMID 14803753.
- ↑ "Possible identity of lactobacillin with hydrogen peroxide produced by lactobacilli". Nature 170 (4328): 623–4. October 1952. doi:10.1038/170623a0. PMID 13002389. Bibcode: 1952Natur.170..623W.
- ↑ 19.0 19.1 19.2 "Prevalence of hydrogen peroxide-producing Lactobacillus species in normal women and women with bacterial vaginosis". Journal of Clinical Microbiology 27 (2): 251–6. February 1989. doi:10.1128/JCM.27.2.251-256.1989. PMID 2915019.
- ↑ "The relationship of hydrogen peroxide-producing lactobacilli to bacterial vaginosis and genital microflora in pregnant women". Obstetrics and Gynecology 79 (3): 369–73. March 1992. doi:10.1097/00006250-199203000-00008. PMID 1738516.
- ↑ "Peroxidase-mediated antimicrobial activity of rat uterine fluid". Gynecologic Investigation 1 (1): 21–30. 1970. doi:10.1159/000301903. PMID 4320447.
- ↑ 22.0 22.1 22.2 "Cervicovaginal fluid and semen block the microbicidal activity of hydrogen peroxide produced by vaginal lactobacilli". BMC Infectious Diseases 10. May 2010. doi:10.1186/1471-2334-10-120. PMID 20482854.
- ↑ "Factors associated with acquisition of, or persistent colonization by, vaginal lactobacilli: role of hydrogen peroxide production". The Journal of Infectious Diseases 184 (11): 1431–6. December 2001. doi:10.1086/324445. PMID 11709785.
- ↑ "Prospective study of correlates of vaginal Lactobacillus colonisation among high-risk HIV-1 seronegative women". Sexually Transmitted Infections 85 (5): 348–53. September 2009. doi:10.1136/sti.2008.035451. PMID 19329442.
- ↑ "Identification and H(2)O(2) production of vaginal lactobacilli from pregnant women at high risk of preterm birth and relation with outcome". Journal of Clinical Microbiology 42 (2): 713–7. February 2004. doi:10.1128/jcm.42.2.713-717.2004. PMID 14766841.
- ↑ 26.0 26.1 26.2 26.3 "Comparison between Gram stain and culture for the characterization of vaginal microflora: definition of a distinct grade that resembles grade I microflora and revised categorization of grade I microflora". BMC Microbiology 5. October 2005. doi:10.1186/1471-2180-5-61. PMID 16225680.
- ↑ 27.0 27.1 27.2 "Quantitative determination by real-time PCR of four vaginal Lactobacillus species, Gardnerella vaginalis and Atopobium vaginae indicates an inverse relationship between L. gasseri and L. iners". BMC Microbiology 7. December 2007. doi:10.1186/1471-2180-7-115. PMID 18093311.
- ↑ 28.0 28.1 "The identification of vaginal Lactobacillus species and the demographic and microbiologic characteristics of women colonized by these species". The Journal of Infectious Diseases 180 (6): 1950–6. December 1999. doi:10.1086/315109. PMID 10558952.
- ↑ "Colonization of the rectum by Lactobacillus species and decreased risk of bacterial vaginosis". The Journal of Infectious Diseases 192 (3): 394–8. August 2005. doi:10.1086/430926. PMID 15995952.
- ↑ "Hydrogen peroxide-producing lactobacilli and acquisition of vaginal infections". The Journal of Infectious Diseases 174 (5): 1058–63. November 1996. doi:10.1093/infdis/174.5.1058. PMID 8896509.
- ↑ "Control of the microbial flora of the vagina by H2O2-generating lactobacilli". The Journal of Infectious Diseases 164 (1): 94–100. July 1991. doi:10.1093/infdis/164.1.94. PMID 1647428.
- ↑ 32.0 32.1 "Vaginal microbiome of reproductive-age women". Proceedings of the National Academy of Sciences of the United States of America 108 Suppl 1 (Supplement 1): 4680–7. March 2011. doi:10.1073/pnas.1002611107. PMID 20534435. Bibcode: 2011PNAS..108.4680R.
- ↑ 33.0 33.1 33.2 33.3 "Characterization of vaginal microbial communities in adult healthy women using cultivation-independent methods". Microbiology 150 (Pt 8): 2565–2573. August 2004. doi:10.1099/mic.0.26905-0. PMID 15289553. https://zenodo.org/record/898195.
- ↑ 34.0 34.1 "The genital econiche: focus on microbiota and bacterial vaginosis". Annals of the New York Academy of Sciences 1230 (1): 48–58. August 2011. doi:10.1111/j.1749-6632.2011.06041.x. PMID 21824165. Bibcode: 2011NYASA1230...48D.
- ↑ "Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns". Proceedings of the National Academy of Sciences of the United States of America 107 (26): 11971–5. June 2010. doi:10.1073/pnas.1002601107. PMID 20566857. Bibcode: 2010PNAS..10711971D.
- ↑ "Bacterial Vaginosis (BV): Condition Information". 2013-05-21. https://www.nichd.nih.gov/health/topics/bacterialvag/conditioninfo.
- ↑ 37.0 37.1 "Microbiota and pelvic inflammatory disease". Seminars in Reproductive Medicine 32 (1): 43–9. January 2014. doi:10.1055/s-0033-1361822. PMID 24390920.
- ↑ "Vaginal microbiota and viral sexually transmitted diseases". Annali di Igiene 25 (5): 443–56. September 2013. doi:10.7416/ai.2013.1946. PMID 24048183.
- ↑ "What are the symptoms of bacterial vaginosis?". 2013-05-21. https://www.nichd.nih.gov/health/topics/bacterialvag/conditioninfo/symptoms.
- ↑ "Vaginal microbiome of reproductive-age women". Proceedings of the National Academy of Sciences of the United States of America 108 (Suppl. 1): 4680–7. March 2011. doi:10.1073/pnas.1002611107. PMID 20534435. Bibcode: 2011PNAS..108.4680R.
- ↑ "Natural history of asymptomatic bacterial vaginosis in a high-risk group of women". Sexually Transmitted Diseases 34 (11): 876–7. November 2007. doi:10.1097/OLQ.0b013e318073bd82. PMID 17522586.
- ↑ "Bacterial vaginosis-A brief synopsis of the literature". European Journal of Obstetrics, Gynecology, and Reproductive Biology 245: 143–148. February 2020. doi:10.1016/j.ejogrb.2019.12.035. PMID 31901667.
- ↑ Bradshaw, Catriona S.; Morton, Anna N.; Hocking, Jane; Garland, Suzanne M.; Morris, Margaret B.; Moss, Lorna M.; Horvath, Leonie B.; Kuzevska, Irene et al. (2006-06-01). "High Recurrence Rates of Bacterial Vaginosis over the Course of 12 Months after Oral Metronidazole Therapy and Factors Associated with Recurrence". The Journal of Infectious Diseases 193 (11): 1478–1486. doi:10.1086/503780. ISSN 0022-1899. PMID 16652274.
- ↑ Chen, Rongdan; Li, Rong; Qing, Wei; Zhang, Yingxuan; Zhou, Zuyi; Hou, Yi; Shi, Yiya; Zhou, Hongwei et al. (2022-06-13). "Probiotics are a good choice for the treatment of bacterial vaginosis: a meta-analysis of randomized controlled trial" (in en). Reproductive Health 19 (1): 137. doi:10.1186/s12978-022-01449-z. ISSN 1742-4755. PMID 35698149.
- ↑ Chow, Karen; Wooten, Darcy; Annepally, Sindhuja; Burke, Leah; Edi, Rina; Morris, Sheldon R. (2023-03-18). "Impact of (recurrent) bacterial vaginosis on quality of life and the need for accessible alternative treatments" (in en). BMC Women's Health 23 (1): 112. doi:10.1186/s12905-023-02236-z. ISSN 1472-6874. PMID 36934289.
- ↑ Lærkeholm Müller, Matilde; Damsted Petersen, Christina; Saunte, Ditte Marie L. (2024). "Boric Acid for the Treatment of Vaginitis: New Possibilities Using an Old Anti-Infective Agent: A Systematic Review" (in en). Dermatologic Therapy 2024 (1). doi:10.1155/2024/2807070. ISSN 1529-8019.
- ↑ Mittelstaedt, Rachel; Kretz, Alyssa; Levine, Michael; Handa, Victoria L.; Ghanem, Khalil G.; Sobel, Jack D.; Powell, Anna; Tuddenham, Susan (2021-12-01). "Data on Safety of Intravaginal Boric Acid Use in Pregnant and Nonpregnant Women: A Narrative Review". Sexually Transmitted Diseases 48 (12): e241–e247. doi:10.1097/OLQ.0000000000001562. ISSN 1537-4521. PMID 34561373.
- ↑ Salam, Maya (2022-03-29). "'Vagina Obscura' Demystifies Female Anatomy" (in en-US). The New York Times. ISSN 0362-4331. https://www.nytimes.com/2022/03/29/books/vagina-obscura-rachel-gross.html.
- ↑ "Isolation of Vaginal Lactobacilli and Characterization of Anti-Candida Activity". PLOS ONE 10 (6). 2015. doi:10.1371/journal.pone.0131220. PMID 26098675. Bibcode: 2015PLoSO..1031220P.
- ↑ "Longitudinal study of the dynamics of vaginal microflora during two consecutive menstrual cycles". PLOS ONE 6 (11). 2011. doi:10.1371/journal.pone.0028180. PMID 22140538. Bibcode: 2011PLoSO...628180L.
- ↑ 51.0 51.1 51.2 51.3 "Differences in the composition of vaginal microbial communities found in healthy Caucasian and black women". The ISME Journal 1 (2): 121–33. June 2007. doi:10.1038/ismej.2007.12. PMID 18043622. Bibcode: 2007ISMEJ...1..121Z.
- ↑ 52.0 52.1 "Deep sequencing of the vaginal microbiota of women with HIV". PLOS ONE 5 (8). August 2010. doi:10.1371/journal.pone.0012078. PMID 20711427. Bibcode: 2010PLoSO...512078H.
- ↑ "Births: final data for 2007". National Vital Statistics Reports 58 (24): 1–85. August 2010. PMID 21254725.
- ↑ "Can known risk factors explain racial differences in the occurrence of bacterial vaginosis?". Journal of the National Medical Association 95 (3): 201–12. March 2003. PMID 12749680.
- ↑ "A comparison of lower genital tract glycogen and lactic acid levels in women and macaques: implications for HIV and SIV susceptibility". AIDS Research and Human Retroviruses 28 (1): 76–81. January 2012. doi:10.1089/aid.2011.0071. PMID 21595610.
- ↑ "Association between bacterial vaginosis and preterm delivery of a low-birth-weight infant. The Vaginal Infections and Prematurity Study Group". The New England Journal of Medicine 333 (26): 1737–42. December 1995. doi:10.1056/nejm199512283332604. PMID 7491137.
- ↑ "Vaginal Swab vs Urine for Detection of Chlamydia trachomatis, Neisseria gonorrhoeae, and Trichomonas vaginalis: A Meta-Analysis". Annals of Family Medicine 21 (2): 172–179. 2023. doi:10.1370/afm.2942. PMID 36973065.
- ↑ "A study on the microbial quality of sealed products for feminine hygiene". Journal of Preventive Medicine and Hygiene 59 (3): E226–E229. September 2018. doi:10.15167/2421-4248/JPMH2018.59.3.920. PMID 30397679.
- ↑ Lewis, Felicia M. T.; Bernstein, Kyle T.; Aral, Sevgi O. (April 2017). "Vaginal Microbiome and Its Relationship to Behavior, Sexual Health, and Sexually Transmitted Diseases". Obstetrics and Gynecology 129 (4): 643–654. doi:10.1097/AOG.0000000000001932. ISSN 1873-233X. PMID 28277350.
- ↑ 60.0 60.1 60.2 "Healthy Vaginal Microbiota and Influence of Probiotics Across the Female Life Span". Frontiers in Microbiology 13. 2022. doi:10.3389/fmicb.2022.819958. PMID 35464937.
- ↑ "Cigarette smoking is associated with an altered vaginal tract metabolomic profile". Scientific Reports 8 (1). January 2018. doi:10.1038/s41598-017-14943-3. PMID 29339821. Bibcode: 2018NatSR...8..852N.
- ↑ "Enhanced Trapping of HIV-1 by Human Cervicovaginal Mucus Is Associated with Lactobacillus crispatus-Dominant Microbiota". mBio 6 (5). October 2015. doi:10.1128/mBio.01084-15. PMID 26443453.
- ↑ "The structure of the human vaginal stratum corneum and its role in immune defense". American Journal of Reproductive Immunology 71 (6): 618–23. June 2014. doi:10.1111/aji.12230. PMID 24661416.
External links
 |
