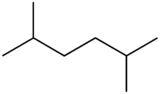
Chemistry:2,5-Dimethylhexane
From HandWiki
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
2,5-Dimethylhexane[1] | |
| Identifiers | |
3D model (JSmol)
|
|
| 1696877 | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 3295 |
| |
| |
| Properties | |
| C8H18 | |
| Molar mass | 114.232 g·mol−1 |
| Appearance | Colourless liquid |
| Odor | Odourless |
| Density | 694 mg mL−1 |
| Melting point | −93 to −89 °C; −136 to −128 °F; 180 to 184 K |
| Boiling point | 108.1 to 109.9 °C; 226.5 to 229.7 °F; 381.2 to 383.0 K |
| Vapor pressure | 7.582 kPa (at 37.7 °C) |
Henry's law
constant (kH) |
3.0 nmol Pa−1 kg−1 |
| -98.15·10−6 cm3/mol | |
Refractive index (nD)
|
1.392 |
| Thermochemistry | |
Heat capacity (C)
|
249.20 J K−1 mol−1 |
Std enthalpy of
formation (ΔfH⦵298) |
−262.0–−259.0 kJ mol−1 |
Std enthalpy of
combustion (ΔcH⦵298) |
−5.4615–−5.4587 MJ mol−1 |
| Hazards | |
| GHS pictograms |    
|
| GHS Signal word | DANGER |
| H225, H304, H315, H336, H410 | |
| P210, P261, P273, P301+310, P331 | |
| Flash point | 26 °C (79 °F; 299 K) |
| Explosive limits | 0.98–?% |
| Related compounds | |
Related alkanes
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
2,5-Dimethylhexane is a branched alkane used in the aviation industry in low revolutions per minute helicopters.[2] As an isomer of octane, the boiling point is very close to that of octane, but can in pure form be slightly lower. 2,5-Dimethylhexane is moderately toxic.
References
- ↑ "2,5-DIMETHYLHEXANE - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 26 March 2005. Identification and Related Records. https://pubchem.ncbi.nlm.nih.gov/summary/summary.cgi?cid=11592&loc=ec_rcs. Retrieved 12 March 2012.
- ↑ Sarathy, S. Mani; Javed, Tamour; Karsenty, Florent; Heufer, Alexander; Wang, Weijing; Park, Sungwoo; Elwardany, Ahmed; Farooq, Aamir et al. (2014-01-16). "A comprehensive combustion chemistry study of 2,5-dimethylhexane" (in English). Combustion and Flame 161 (6): 1444–1459. doi:10.1016/j.combustflame.2013.12.010. ISSN 0010-2180. https://www.osti.gov/biblio/1755830.
 |
