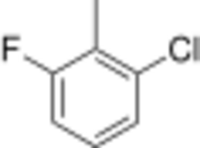
Chemistry:2-Chloro-6-fluorotoluene
From HandWiki
| |
| Names | |
|---|---|
| IUPAC name
1-chloro-3-fluoro-2-methylbenzene
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| C7H6ClF | |
| Molar mass | 144.57 g·mol−1 |
| Boiling point | 154–156 °C (309–313 °F) |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | Warning |
| H226, H302, H312, H315, H332, H335 | |
| P210, P233, P240, P241, P242, P243, P261, P264, P270, P271, P280, P302+352, P303+361+353, P304+340, P305+351+338, P321, P330, P362+364Script error: No such module "Preview warning".Category:GHS errors, P370+378, P403+233, P403+235, P405, P501 | |
| Flash point | 46 °C (115 °F) |
| Related compounds | |
Related compounds
|
2-Chloro-6-fluorobenzaldehyde, toluene |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
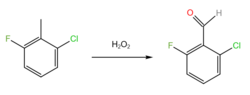
2-Chloro-6-fluorotoluene (CFT) is a halogenated derivative of toluene that is used as an intermediate in numerous organic syntheses.[1][2]
Uses
CFT is used to prepare 2-chloro-6-fluorobenzaldehyde via oxidation with hydrogen peroxide, which forms an aldehyde group.[2]
CFT is also used in the preparation of 4-chloro-1H-indazole.[1]
References
- ↑ 1.0 1.1 Meng, Ge; Yang, Tao; Liu, Yang (2011). "An Improved Preparation of 4-Chloro-1 H -indazole" (in en). Organic Preparations and Procedures International 43 (4): 354–359. doi:10.1080/00304948.2011.594005. ISSN 0030-4948. http://www.tandfonline.com/doi/abs/10.1080/00304948.2011.594005.
- ↑ 2.0 2.1 Bunnett, J. F.; Miles, J. H.; Nahabedian, K. V. (1961). "Kinetics and Mechanism of the Alkali Cleavage of 2,6-Dihalobenzaldehydes 1" (in en). Journal of the American Chemical Society 83 (11): 2512–2516. doi:10.1021/ja01472a022. ISSN 0002-7863. https://pubs.acs.org/doi/abs/10.1021/ja01472a022.
 |