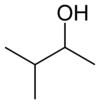
Chemistry:3-Methyl-2-butanol
From HandWiki
| |
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
3-Methylbutan-2-ol[2] | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 1105 |
| |
| |
| Properties | |
| C5H12O | |
| Molar mass | 88.150 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 818 mg cm−3 |
| Boiling point | 109 to 115 °C; 228 to 239 °F; 382 to 388 K |
| 59 g dm−3 | |
| Solubility in ethanol | miscible |
| log P | 1.036 |
| Vapor pressure | 1.20 kPa |
| Thermochemistry | |
Heat capacity (C)
|
245.9 J K−1 mol−1 |
Std enthalpy of
formation (ΔfH⦵298) |
-371.3--368.5 kJ mol−1 |
Std enthalpy of
combustion (ΔcH⦵298) |
-3.3157--3.3145 MJ mol−1 |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | WARNING |
| H226, H332, H335 | |
| P261 | |
| NFPA 704 (fire diamond) | |
| Flash point | 34 °C (93 °F; 307 K) |
| Related compounds | |
Related compounds
|
Amyl alcohol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
3-Methyl-2-butanol (IUPAC name, commonly called sec-isoamyl alcohol) is an organic chemical compound. It is used as a solvent and an intermediate in the manufacture of other chemicals.[3]
References
- ↑ Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, Florida: CRC Press, pp. 3-374, 5-42, 8-102, 15-22, ISBN 0-8493-0594-2
- ↑ "sec-Isoamyl alcohol - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 26 March 2005. https://pubchem.ncbi.nlm.nih.gov/summary/summary.cgi?cid=11732.
- ↑ The compound is one of the eight isomers of pentyl alcohol. McKetta, John J.; Cunningham, William Aaron (1977), Encyclopedia of Chemical Processing and Design, 3, Boca Raton, Florida: CRC Press, pp. 280–281, ISBN 978-0-8247-2480-1, https://books.google.com/books?id=iwSU5G5VzO0C&pg=PA279, retrieved 2010-01-17
 |


