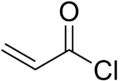
Chemistry:Acryloyl chloride
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Prop-2-enoyl chloride | |
| Other names
2-Propenoyl chloride
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C3H3ClO | |
| Molar mass | 90.51 g·mol−1 |
| Density | 1.119 g/cm3 |
| Boiling point | 75.0 °C (167.0 °F; 348.1 K) |
| Hazards | |
| GHS pictograms |   
|
| GHS Signal word | Danger |
| H225, H302, H314, H330 | |
| P210, P280, P301+330+331, P303+361+353, P305+351+338, P310 | |
| Flash point | −4 °C (25 °F; 269 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Acryloyl chloride, also known as 2-propenoyl chloride or acrylic acid chloride, is the organic compound with the formula CH2=CHCO(Cl). It is a colorless liquid, although aged samples appear yellow. It belongs to the acid chlorides group of compounds.[1]
Preparation
Acryloyl chloride can be efficiently prepared by treating acrylic acid with benzoyl chloride:[2]
- CH
2=CHCO
2H + C
6H
5COCl → CH
2=CHCOCl + C
6H
5CO
2H
Conventional phosphorus-based chlorinating agents, e.g. phosphorus trichloride, are ineffective. Flow conditions allows use of a broadened range of chlorinating agents including oxalyl chloride and thionyl chloride.[1] [3]
Reactions
This compound undergoes the reactions common for acid chlorides. For example, it reacts readily with water, producing acrylic acid. When treated with sodium salts of carboxylic acids, the anhydride is formed. Reactions with alcohols and amines gives esters and amides, respectively. It acylates organozinc compounds.[4]
Acryloyl chloride is most commonly employed for the introduction of acrylic groups into other compounds, e.g. the preparation of acrylate monomers and polymers.[5]
Toxicity
Acryloyl chloride, like volatile acid chlorides, is a skin irritant, with pulmonary edema in more severe exposures.[6] Other signs and symptoms of acute exposure may include headache, dizziness, and weakness. Gastrointestinal effects may include nausea, vomiting, diarrhea, and stomach ulceration.[7]
References
- ↑ 1.0 1.1 PatentStorm LLC (2006). "Process for the manufacture of acryloyl chloride". PatentStorm LLC. http://www.patentstorm.us/patents/5395966-description.html. Retrieved December 21, 2007.
- ↑ Stempel, Guido H. Jr.; Cross, Robert P.; Mariella, Raymond P. (1950). "Preparation of acrylyl chloride". Journal of the American Chemical Society 72 (5): 2299–2300. doi:10.1021/ja01161a527.
- ↑ Movsisyan, Marine; Heugebaert, Thomas S. A.; Dams, Rudy; Stevens, Christian V. (9 August 2016). "Safe, Selective, and High-Yielding Synthesis of Acryloyl Chloride in a Continuous-Flow System". ChemSusChem 9 (15): 1945–1952. doi:10.1002/cssc.201600348. PMID 27325562.
- ↑ Yoshinao Tamaru, Hirofumi Ochiai, Tatsuya Nakamura, Zen-ichi Yoshida (1989). "Ethyl 5-Oxo-6-Methyl-6-Heptenoate from Methacryloyl Chloride and Ethyl 4-Iodobutyrate". Organic Syntheses 67: 98. doi:10.15227/orgsyn.067.0098.
- ↑ Ohara, Takashi; Sato, Takahisa; Shimizu, Noboru; Prescher, Günter; Schwind, Helmut; Weiberg, Otto; Marten, Klaus (2003). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a01_161.
- ↑ Pathan, Asama; Ahmad, Iqrar; Girase, Rukaiyya; Jagatap, Vilas; Patel, Harun (August 23, 2022). "A Laboratory Accident of Acryloyl Chloride, Its Consequences, Treatment, and Safety Measures: An Arduous Lesson to All Researchers". ACS Chemical Health & Safety 29 (5): 405-420. doi:10.1021/acs.chas.2c00036. https://pubs.acs.org/doi/10.1021/acs.chas.2c00036. Retrieved 8 April 2023.
- ↑ "ACRYLYL CHLORIDE | CAMEO Chemicals | NOAA". https://cameochemicals.noaa.gov/chemical/4850.
See also
 |
