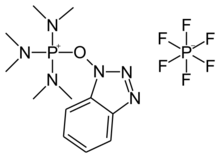
Chemistry:BOP reagent
| |
| Names | |
|---|---|
| IUPAC name
((1H-Benzo[d][1,2,3]triazol-1-yl)oxy)tris(dimethylamino)phosphonium hexafluorophosphate(V)
| |
| Other names
Castro's reagent
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C12H22F6N6OP2 | |
| Molar mass | 442.287 g/mol |
| Appearance | White crystalline powder |
| Melting point | 136 to 140 °C (277 to 284 °F; 409 to 413 K) |
| Partially soluble in cold water reacts (decomposes) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
This article may be too technical for most readers to understand. Please help improve it to make it understandable to non-experts, without removing the technical details. (January 2021) (Learn how and when to remove this template message) |
BOP (benzotriazol-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate) is a reagent commonly used for the synthesis of amides from carboxylic acids and amines in peptide synthesis.[1][2] It can be prepared from 1-hydroxybenzotriazole and a chlorophosphonium reagent under basic conditions.[3] This reagent has advantages in peptide synthesis since it avoids side reactions like the dehydration of asparagine or glutamine residues.[4] BOP has used for the synthesis of esters from the carboxylic acids and alcohols.[5] BOP has also been used in the reduction of carboxylic acids to primary alcohols with sodium borohydride (NaBH4).[6] Its use raises safety concerns since the carcinogenic compound HMPA is produced as a stoichiometric by-product.
See also
- PyBOP, a related phosphonium reagent for amide bond formation
- PyAOP, a related phosphonium reagent for amide bond formation
References
- ↑ "(Benzotriazol-1-yloxy)tris(dimethylamino)phosphonium hexafluorophosphate 226084". https://www.sigmaaldrich.com/catalog/product/aldrich/226084.
- ↑ Mansour, Tarek S.; Bardhan, Sujata; Wan, Zhao-Kui (2010). "Phosphonium- and Benzotriazolyloxy-Mediated Bond-Forming Reactions and Their Synthetic Applications" (in en). Synlett 2010 (8): 1143–1169. doi:10.1055/s-0029-1219820. ISSN 0936-5214. http://www.thieme-connect.de/DOI/DOI?10.1055/s-0029-1219820.
- ↑ Hoffmann, Frank; Jäger, Lothar; Griehl, Carola (2003-02-01). "Synthesis and Chemical Constitution of Diphenoxyphosphoryl Derivatives and Phosphonium Salts as Coupling Reagents for Peptide Segment Condensation" (in en). Phosphorus, Sulfur, and Silicon and the Related Elements 178 (2): 299–309. doi:10.1080/10426500307942. ISSN 1042-6507. https://www.tandfonline.com/doi/full/10.1080/10426500307942.
- ↑ Prasad, KVSRG; Bharathi, K; Haseena, Banu B (2011). "Applications of Peptide Coupling Reagents- An Update". International Journal of Pharmaceutical Sciences Review and Research 8 (1): 108–119. https://globalresearchonline.net/journalcontents/volume8issue1/Article-021.pdf.
- ↑ Kim, Moon H.; Patel, Dinesh V. (1994-08-01). ""BOP" as a reagent for mild and efficient preparation of esters". Tetrahedron Letters 35 (31): 5603–5606. doi:10.1016/S0040-4039(00)77257-1. ISSN 0040-4039. https://www.sciencedirect.com/science/article/pii/S0040403900772571.
- ↑ McGeary, Ross P. (1998). "Facile and chemoselective reduction of carboxylic acids to alcohols using BOP reagent and sodium borohydride" (in en). Tetrahedron Letters 39 (20): 3319–3322. doi:10.1016/S0040-4039(98)00480-8. ISSN 0040-4039.
 |
