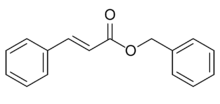
Chemistry:Benzyl cinnamate
From HandWiki
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
Benzyl (2E)-3-phenylprop-2-enoate | |
| Other names
Benzyl cinnamate
Cinnamein Benzyl cinnamoate Benzyl 3-phenylpropenoate 3-Phenyl-2-propenoic acid phenylmethyl ester Cinnamic acid benzyl ester | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C16H14O2 | |
| Molar mass | 238.286 g·mol−1 |
| Appearance | White to pale yellow solid[1] |
| Melting point | 34–37 °C (93–99 °F; 307–310 K)[2] |
| Boiling point | 195–200 °C (383–392 °F; 468–473 K) 5 mmHg[2] |
| Insoluble[1] | |
| Solubility in ethanol | 125 g/L |
| Solubility in glycerin | Insoluble |
| Solubility in propylene glycol | Insoluble |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Benzyl cinnamate is the chemical compound which is the ester derived from cinnamic acid and benzyl alcohol.
Natural occurrence
Balsam is the major producer of benzyl cinnamate.[3] It is used as an ingredient in the medicated cream product Sudocrem.[4]
Uses
It is used as a flavoring agent.[3] It has a low odour smelling faintly sweet, balsamic, floral, fruity, cherry, spicy, and powdery.[5]
It is used pharmaceutically as an antibacterial and antifungal.[6]
References
- ↑ 1.0 1.1 "Specifications for Flavourings". Food and Agriculture Organization. http://www.fao.org/ag/agn/jecfa-flav/details.html?flavId=3497.
- ↑ 2.0 2.1 "Benzyl cinnamate". Sigma-Aldrich. http://www.sigmaaldrich.com/catalog/product/aldrich/234214?lang=en.
- ↑ 3.0 3.1 George A. Burdock (2010), Fenaroli's Handbook of Flavor Ingredients (6th ed.), CRC Press, pp. 147–148
- ↑ "Sudocrem Antiseptic Healing Cream - Summary of Product Characteristics (SmPC) - (emc)". https://www.medicines.org.uk/emc/product/8860/smpc/.
- ↑ "benzyl cinnamate". Scents and Flavors. https://scentsandflavors.com/database/9dbb5052-565d-4cb2-b71d-c0d7e08fae35.
- ↑ Korošec, B.; Sova, M.; Turk, S.; Kraševec, N.; Novak, M.; Lah, L.; Stojan, J.; Podobnik, B. et al. (2014). "Antifungal activity of cinnamic acid derivatives involves inhibition of benzoate 4-hydroxylase (CYP53)" (in en). Journal of Applied Microbiology 116 (4): 955–966. doi:10.1111/jam.12417. ISSN 1365-2672. PMID 24314266.
External links
- Benzyl cinnamate at National Library of Medicine's Toxicology Data Network
 |
