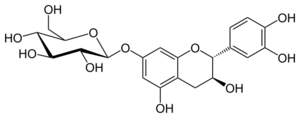
Chemistry:Catechin-7-O-glucoside
| |
| Names | |
|---|---|
| IUPAC name
(2S,4S,5S)-2-[[(2R,3S)-2-(3,4-Dihydroxyphenyl)-3,5-dihydroxy-3,4-dihydro-2H-chromen-7-yl]oxy]-6-(hydroxymethyl)oxane-3,4,5-triol
| |
| Other names
(2R,3S)-Catechin-7-O-β-D-glucopyranoside
Catechin 7-O-β-glucopyranoside (+)-Catechin 7-O-β-glucoside (+)-Catechin 7-O-beta-D-glucopyranoside Catechin 7-glucoside C7G CA-G | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C21H24O11 | |
| Molar mass | 452.412 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Catechin-7-O-glucoside is a flavan-3-ol glycoside formed from catechin.
Natural occurrences
Catechin-7-O-glucoside can be isolated from the hemolymph of the European pine sawfly (Neodiprion sertifer).[1] It also occurs in relatively large quantities in cowpea (Vigna unguiculata) as the dominant flavan-3-ol monomer, and actually accounts for up to 70% of cowpea proanthocyanidins (tannins).[2]
It can also be produced by biotransformation of (+)-catechin by cultured cells of Eucalyptus perriniana.[3]
Presence in natural traditional drugs
Catechin-7-O-glucoside can be found in paeoniae radix, the crude drug made from the roots of Chinese peony (Paeonia lactiflora),[4] in red knotweed (Bistorta macrophylla, also known as Polygonum macrophyllum),[5] in the stem barks of the Nepali hog plum (Choerospondias axillaris),[6] in the Korean plum yew (Cephalotaxus koreana)[7] and in Huanarpo Macho (Jatropha macrantha).[8] (−)-Catechin 7-O-β-D-glucopyranoside is found in the bark of Rhaphiolepis umbellata.[9]
Presence in food
It is found in buckwheat groats,[10] in the red bean (the seed of Vigna umbellata, formerly known as Phaseolus calcaratus),[11] in barley (Hordeum vulgare L.) and malt.[12] (−)-Catechin 7-O'-β-D-glucopyranoside is found in rhubarb.[9]
Health effects
This compound has an antioxidant activity leading to a cytoprotective effect.[11][13]
References
- ↑ Vihakas, Matti; Tähtinen, Petri; Ossipov, Vladimir; Salminen, Juha-Pekka (2012). "Flavonoid Metabolites in the Hemolymph of European Pine Sawfly (Neodiprion sertifer) Larvae". Journal of Chemical Ecology 38 (5): 538–46. doi:10.1007/s10886-012-0113-y. PMID 22527054.
- ↑ Ojwang, Leonnard O.; Yang, Liyi; Dykes, Linda; Awika, Joseph (2013-08-15). "Proanthocyanidin profile of cowpea (Vigna unguiculata) reveals catechin-O-glucoside as the dominant compound". Food Chemistry 139 (1–4): 35–43. doi:10.1016/j.foodchem.2013.01.117. PMID 23561075.
- ↑ Otani, Shuichi; Kondo, Yoko; Asada, Yoshihisa; Furuya, Tsutomu; Hamada, Hatsuyuki; Nakajima, Nobuyoshi; Ishihara, Kohji; Hamada, Hiroki (2004). "Biotransformation of (+)-catechin by plant cultured cells of Eucalyptus perriniana". Plant Biotechnology 21 (5): 407–409. doi:10.5511/plantbiotechnology.21.407.
- ↑ Tanaka, Takashi; Kataoka, Maki; Tsuboi, Nagisa; Kouno, Isao (2000). "New Monoterpene Glycoside Esters and Phenolic Constituents of Paeoniae Radix, and Increase of Water Solubility of Proanthocyanidins in the Presence of Paeoniflorin". Chemical and Pharmaceutical Bulletin 48 (2): 201—207. doi:10.1248/cpb.48.201. PMID 10705504.
- ↑ Wang, S.; Wang, D.; Feng, S. (2004). "Studies on chemical constituents from Polygonum macrophyllum". Journal of Chinese Medicinal Materials 27 (6): 411–413. PMID 15524292.
- ↑ Li, Chang-wei; Cui, Cheng-bin; Cai, Bing; Han, Bing; Li, Ming-ming; Fan, Ming (2009). "Flavanoidal constituents of Choerospondias axillaris and their in vitro antitumor and anti-hypoxia activities". Chinese Journal of Medicinal Chemistry 19 (1): 48–51, 64. http://www.zgyhzz.cn/qikan/epaper/zhaiyao.asp?bsid=14526.)
- ↑ Yoon, Kee-dong; Jeong, Doc-gyun; Hwang, Yun-ha; Ryu, Jei-man; Kim, Jin-woong (2007). "Inhibitors of Osteoclast Differentiation from Cephalotaxus koreana". Journal of Natural Products 70 (12): 2029–2032. doi:10.1021/np070327e. PMID 17994703.
- ↑ Benavides, Angelyne; Montoro, Paola; Bassarello, Carla; Piacente, Sonia; Pizza, Cosimo (2006). "Catechin derivatives in Jatropha macrantha stems: Characterisation and LC/ESI/MS/MS quali–quantitative analysis". Journal of Pharmaceutical and Biomedical Analysis 40 (3): 639–647. doi:10.1016/j.jpba.2005.10.004. PMID 16300918.
- ↑ Jump up to: 9.0 9.1 Nonaka, Gen'ichiro; Ezakia, Emiko; Hayashia, Katsuya; Nishioka, Itsuo (1983). "Flavanol glucosides from rhubarb and Rhaphiolepis umbellata". Phytochemistry 22 (7): 1659–1661. doi:10.1016/0031-9422(83)80105-8.
- ↑ Report on cereals at Phenol-Explorer.eu. Retrieved 18 December 2012.
- ↑ Jump up to: 11.0 11.1 Baek, Jin-a; Son, Young-ok; Fang, Minghao; Lee, Young-jae; Cho, Hyoung-kwon; Whang, Wan-kyunn; Lee, Jeong-Chae (2011). "Catechin-7-O-β-D-glucopyranoside scavenges free radicals and protects human B lymphoma BJAB cells on H2O2-mediated oxidative stress". Food Science and Biotechnology 20: 151–158. doi:10.1007/s10068-011-0021-x., INIST:23809899
- ↑ Friedrich, Wolfgang; Galensa, Rudolf (2002). "Identification of a new flavanol glucoside from barley (Hordeum vulgare L.) and malt". European Food Research and Technology 214 (5): 388. doi:10.1007/s00217-002-0498-x.
- ↑ Kim, Ki-cheon; Kim, Jin-sook; Kang, Kyoung-ah; Kim, Jong-min; Hyun, Jin-won (2010). "Cytoprotective effects of catechin 7-O-β-D-glucopyranoside against mitochondrial dysfunction damaged by streptozotocin in RINm5F cells". Cell Biochemistry and Function 28 (8): 651–660. doi:10.1002/cbf.1703. PMID 21104932.
 |

