Chemistry:Cysteic acid
From HandWiki
| |
| |
| Names | |
|---|---|
| IUPAC name
(R)-2-Amino-3-sulfopropanoic acid
| |
| Other names
3-Sulfo-l-alanine
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| DrugBank | |
| EC Number |
|
| MeSH | Cysteic+acid |
PubChem CID
|
|
| UNII |
|
| |
| |
| Properties[1] | |
| C3H7NO5S | |
| Molar mass | 169.15 g·mol−1 |
| Appearance | White crystals or powder |
| Melting point | Decomposes around 272 °C |
| Soluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
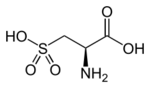
Cysteic acid also known as 3-sulfo-l-alanine is the organic compound with the formula HO3SCH2CH(NH2)CO2H. It is often referred to as cysteate, which near neutral pH takes the form −O3SCH2CH(NH3+)CO2−.
It is an amino acid generated by oxidation of cysteine, whereby a thiol group is fully oxidized to a sulfonic acid/sulfonate group. It is further metabolized via 3-sulfolactate, which converts to pyruvate and sulfite/bisulfite. The enzyme L-cysteate sulfo-lyase catalyzes this conversion. Cysteate is a biosynthetic precursor to taurine in microalgae.[2] By contrast, most taurine in animals is made from cysteine sulfinate.[3]
References
- ↑ Weast, Robert C., ed (1981). CRC Handbook of Chemistry and Physics (62nd ed.). Boca Raton, FL: CRC Press. p. C259. ISBN 0-8493-0462-8..
- ↑ Tevatia, Rahul; Allen, James; Rudrappa, Deepak; White, Derrick; Clemente, Thomas E.; Cerutti, Heriberto; Demirel, Yaşar; Blum, Paul (2015-05-01). "The taurine biosynthetic pathway of microalgae" (in en). Algal Research 9: 21–26. doi:10.1016/j.algal.2015.02.012. ISSN 2211-9264. https://digitalcommons.unl.edu/cgi/viewcontent.cgi?article=1167&context=plantscifacpub.
- ↑ Cook, Alasdair M.; Denger, Karin; Smits, Theo H. M. (2006). "Dissimilation of C3-Sulfonates". Archives of Microbiology 185 (2): 83–90. doi:10.1007/s00203-005-0069-1. PMID 16341843. http://nbn-resolving.de/urn:nbn:de:bsz:352-opus-67394.
 |
