Chemistry:Diamine
A diamine is an amine with two amino groups. Diamines are used as monomers to prepare polyamides, polyimides, and polyureas. The term diamine refers mostly to primary diamines, as those are the most reactive.[1][2]
Practical considerations
In terms of quantities produced, 1,6-diaminohexane (a precursor to Nylon 6-6) is most important, followed by ethylenediamine.[3] Vicinal diamines (1,2-diamines) are a structural motif in many biological compounds and are used as ligands in coordination chemistry.[4] Many diamines are used as hardeners in the curing of epoxide resins. Putrescine and cadaverine are simple diamines that occur widely in nature.
Aliphatic diamines
Linear
- 2 carbon backbone: ethylenediamine (1,2-diaminoethane). Related derivatives include the N-alkylated compounds, 1,1-dimethylethylenediamine, 1,2-dimethylethylenediamine, ethambutol, tetrakis(dimethylamino)ethylene, TMEDA. Many 1,2-diamine derivatives are of practical interest such as penicillin.[5][6]

- 3 carbon backbone: 1,3-diaminopropane (propane-1,3-diamine)
- 4 carbon backbone: putrescine (butane-1,4-diamine)
- 5 carbon backbone: cadaverine (pentane-1,5-diamine)

- 6 carbon backbone: hexamethylenediamine (hexane-1,6-diamine, HMD). HMD and other long chain diamines can be prepared by hydrocyanation to give dinitrile, which can be hydrogenated.[7]
Branched
Derivatives of ethylenediamine are prominent:
- 1,2-diaminopropane, which is chiral.
- 2,3-Butanediamine, two diastereomers, one of which is C2-symmetric.
- Diphenylethylenediamine, two diastereomers, one of which is C2-symmetric.
- trimethylhexamethylenediamine, several isomers
- 1,2-Diaminocyclohexane, two diastereomers, one of which is C2-symmetric.
Cyclic
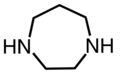
- piperazine ((CH
2CH
2NH)
2) - 1,4-Diazacycloheptane
Xylylenediamines
Xylylenediamines are classified as alkylamines since the amine is not directly attached to an aromatic ring.
- o-xylylenediamine or OXD
- m-xylylenediamine or MXD
- p-xylylenediamine or PXD
Aromatic diamines
Three phenylenediamines are known:[8]
- o-phenylenediamine or OPD
- m-phenylenediamine or MPD
- p-phenylenediamine or PPD. 2,5-diaminotoluene is related to PPD but contains a methyl group on the ring.
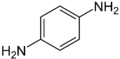
Various N-methylated derivatives of the phenylenediamines are known:
- dimethyl-4-phenylenediamine, a reagent.
- N,N'-di-2-butyl-1,4-phenylenediamine, an antioxidant.
Examples with two aromatic rings include derivatives of biphenyl and naphthalene:
- 4,4'-diaminobiphenyl
- 1,8-diaminonaphthalene

Geminal diamines

Geminal diamines (1,1-diamines) are an uncommon class of diamines mainly of academic interest. Of the few that exist, most are di-tertiary amines. Bis(dimethylamino)methane ([(CH3)2N]2CH2) is an isolable example.
Geminal diamines with N-H bonds are particularly rare. They are invoked as intermediates in transimination reactions and the reduction of amidines. In aqueous conditions they preferentially eliminate the less basic amine to leave an iminium ion.[9] Some stable geminal diamines have been isolated.[10] The parent gem-diamine is methylenediamine (diaminomethane), which again is mainly of theoretical interest.
References
- ↑ Lucet, Denis; Le Gall, Thierry; Mioskowski, Charles (1998). "The Chemistry of Vicinal Diamines". Angewandte Chemie International Edition 37 (19): 2580–2627. doi:10.1002/(SICI)1521-3773(19981016)37:19<2580::AID-ANIE2580>3.0.CO;2-L. PMID 29711625.
- ↑ "Nucleophilicity Trends of Amines" (in en-US). 2018-05-07. https://www.masterorganicchemistry.com/2018/05/07/nucleophilicity-of-amines/.
- ↑ Peter Roose; Karsten Eller; Erhard Henkes; Roland Rossbacher; Hartmut Höke (2005). "Amines, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. pp. 1–55. doi:10.1002/14356007.a02_001.pub2. ISBN 3-527-30673-0.
- ↑ Lucet, Denis; Le Gall, Thierry; Mioskowski, Charles (1998). "The Chemistry of Vicinal Diamines". Angewandte Chemie International Edition 37 (19): 2580–2627. doi:10.1002/(SICI)1521-3773(19981016)37:19<2580::AID-ANIE2580>3.0.CO;2-L. PMID 29711625.
- ↑ Saibabu Kotti, S. R. S.; Timmons, Cody; Li, Guigen (2006). "Vicinal Diamino Functionalities as Privileged Structural Elements in Biologically Active Compounds and Exploitation of their Synthetic Chemistry". Chemical Biology & Drug Design 67 (2): 101–114. doi:10.1111/j.1747-0285.2006.00347.x. PMID 16492158.
- ↑ Lucet, Denis; Le Gall, Thierry; Mioskowski, Charles (1998). "The Chemistry of Vicinal Diamines". Angewandte Chemie International Edition 37 (19): 2580–2627. doi:10.1002/(SICI)1521-3773(19981016)37:19<2580::AID-ANIE2580>3.0.CO;2-L. PMID 29711625.
- ↑ Dahlmann, Marc; Grub, Joachim; Löser, Eckhard (2011). "Butadiene". Ullmann's Encyclopedia of Industrial Chemistry. pp. 1–24. doi:10.1002/14356007.a04_431.pub2. ISBN 978-3-527-30673-2.
- ↑ Robert A. Smiley "Phenylene- and Toluenediamines" in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a19_405
- ↑ Moad, Graeme; Benkovic, S. J. (1 August 1978). "On the mechanism of decomposition of geminal diamines" (in en). Journal of the American Chemical Society 100 (17): 5495–5499. doi:10.1021/ja00485a038. ISSN 0002-7863. Bibcode: 1978JAChS.100.5495M. https://pubs.acs.org/doi/abs/10.1021/ja00485a038.
- ↑ Aydın, Fatma; Arslan, N Burcu (5 September 2021). "Synthesis, spectral properties, crystal structure and theoretical calculations of a new geminal diamine: 2,2,2-Trichloro-N,N׳-bis(2-nitrophenyl)-ethane-1,1-diamine" (in en). Journal of Molecular Structure 1232. doi:10.1016/j.molstruc.2021.129976. Bibcode: 2021JMoSt123229976A. https://linkinghub.elsevier.com/retrieve/pii/S0022286021001071.
External links
- Diamines at the US National Library of Medicine Medical Subject Headings (MeSH)
- Synthesis of diamines
 |
