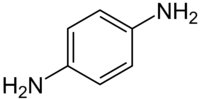
Chemistry:P-Phenylenediamine
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Benzene-1,4-diamine | |||
| Other names
Paraphenylenediamine
1,4-Diaminobenzene 1,4-Phenylenediamine | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 749029 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| EC Number |
| ||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1673 | ||
| |||
| |||
| Properties | |||
| C6H8N2 | |||
| Molar mass | 108.144 g·mol−1 | ||
| Appearance | White crystalline solid, darkens upon exposure to air[1] | ||
| Melting point | 145 to 147 °C (293 to 297 °F; 418 to 420 K)[1] | ||
| Boiling point | 267 °C (513 °F; 540 K)[1] | ||
| 10% at 40°C, 87% at 107 C, 100% at 140 C [2] | |||
| Vapor pressure | <1 mmHg (20°C)[3] | ||
| Acidity (pKa) |
| ||
| -70.28·10−6 cm3/mol | |||
| Hazards | |||
| GHS pictograms |   
| ||
| GHS Signal word | Danger | ||
| H301, H311, H317, H319, H331, H410 | |||
| P261, P264, P270, P271, P272, P273, P280, P301+310, P302+352, P304+340, P305+351+338, P311, P312, P321, P322, P330, P333+313, P337+313, P361, P363, P391, P403+233, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 156 °C; 312 °F; 429 K[3] | ||
| 400 °C (752 °F; 673 K) | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
80 mg/kg (rat, oral) 98 mg/kg (rat, oral) 145 mg/kg (guinea pig, oral)[4] | ||
LDLo (lowest published)
|
250 mg/kg (rabbit, oral) 100 mg/kg (cat, oral)[4] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 0.1 mg/m3 [skin][3] | ||
REL (Recommended)
|
TWA 0.1 mg/m3 [skin][3] | ||
IDLH (Immediate danger)
|
25 mg/m3[3] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
p-Phenylenediamine (PPD) is an organic compound with the formula C6H4(NH2)2. This derivative of aniline is a white solid, but samples can darken due to air oxidation.[1] It is mainly used as a component of engineering polymers and composites like kevlar. It is also an ingredient in hair dyes and is occasionally used as a substitute for henna.
Production
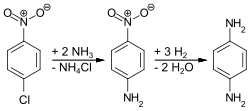
PPD is produced via three routes. Most commonly, 4-nitrochlorobenzene is treated with ammonia and the resulting 4-nitroaniline is then hydrogenated:
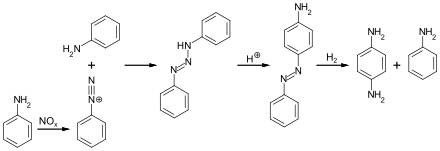
In the DuPont route, aniline is converted to diphenyltriazine, which is then converted by acid-catalysis to 4-aminoazobenzene. Hydrogenation of the latter affords PPD.[6]
Uses
Precursor to polymers
PPD is a precursor to aramid plastics and fibers such as Kevlar and Twaron. These applications exploit PPD's difunctionality, i.e. the presence of two amines which allow the molecules to be strung together. This polymer arises from the reaction of PPD and terephthaloyl chloride. The reaction of PPD with phosgene gives the diisocyanate, a precursor to urethane polymers.[6]

Dyeing
This compound is a common hair dye. Its use is being supplanted by other aniline analogues and derivatives such as 2,5-diamino(hydroxyethylbenzene and 2,5-diaminotoluene). Other popular derivatives include tetraaminopyrimidine and indoanilines and indophenols. Derivatives of diaminopyrazole give both red and violet colours.[7] In these applications, the nearly colourless dye precursor oxidizes to the dye.
Rubber antioxidant
PPD is easily oxidized, and for this reason, derivatives of PPD are used as antiozonants in the production of rubber products (e.g., IPPD). The substituents (naphthyl, isopropyl, etc.) affect the effectiveness of their antioxidant roles as well as their properties as skin irritants.[8]
Other uses
A substituted form of PPD sold under the name CD-4 is also used as a developing agent in the C-41 color photographic film development process, reacting with the silver grains in the film and creating the colored dyes that form the image.
PPD is also used as a Henna surrogate for temporary tattoos. Its usage can lead to severe contact dermatitis.
PPD is also used as a histological stain for lipids such as myelin.
PPD is used by lichenologists in the PD test to aid identification of Lichens.[9] PPD is used extensively as a cross-linking agent in the formation of COFs (covalent organic frameworks), which have a number of applications in dyes and aromatic compounds adsorption.
Safety
The aquatic -1">50 of PPD is 0.028 mg/L.[6] The U.S. Environmental Protection Agency reported that in rats and mice chronically exposed to PPD in their diet, it simply depressed body weights, and no other clinical signs of toxicity were observed in several studies.[10] One review of 31 English-language articles published between January 1992 and February 2005 that investigated the association between personal hair dye use and cancer as identified through the PubMed search engine found "at least one well-designed study with detailed exposure assessment" that observed associations between personal hair dye use and non-Hodgkin's lymphoma, multiple myeloma, acute leukemia, and bladder cancer,[11] but those associations were not consistently observed across studies. A formal meta-analysis was not possible due to the heterogeneity of the exposure assessment across the studies.
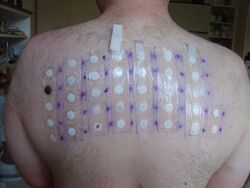
In 2005–06, it was the tenth-most-prevalent allergen in patch tests (5.0%).[12]
The CDC lists PPD as being a contact allergen. Exposure routes are through inhalation, skin absorption, ingestion, and skin and/or eye contact. Symptoms of exposure to PPD include throat irritation (pharynx and larynx), bronchial asthma, and sensitization dermatitis.[13][14] Sensitization is a lifelong issue, which may lead to active sensitization to products, including but not limited to black clothing, various inks, hair dye, dyed fur, dyed leather, and certain photographic products. It was voted Allergen of the Year in 2006 by the American Contact Dermatitis Society.
Poisoning by PPD is rare in Western countries.[15] In contrast, poisoning by PPD has occurred in Eastern countries, such as Pakistan, where people have committed suicide by consuming it.[16]
See also
- Henna
References
- ↑ 1.0 1.1 1.2 1.3 Merck Index, 11th Edition, 7256
- ↑ "P-Phenylenediamine (1,4-Diaminobenzene)". http://chemicalland21.com/specialtychem/perchem/p-phenylenediamine.htm.
- ↑ 3.0 3.1 3.2 3.3 3.4 NIOSH Pocket Guide to Chemical Hazards. "#0495". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0495.html.
- ↑ 4.0 4.1 "p-Phenylene diamine". Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/idlh/106503.html.
- ↑ "p-Phenylenediamine MSDS". Thermo Fisher Scientific. https://www.fishersci.com/store/msds?partNumber=AC130570025&productDescription=P-PHENYLENEDIAMINE+99%2B%25+2.5KG&vendorId=VN00032119&countryCode=US&language=en.
- ↑ 6.0 6.1 6.2 (in en) Ullmann's Encyclopedia of Industrial Chemistry (1 ed.). Wiley. 2003-03-11. doi:10.1002/14356007.a19_405. ISBN 978-3-527-30385-4. https://onlinelibrary.wiley.com/doi/book/10.1002/14356007.
- ↑ Thomas Clausen et al. "Hair Preparations" in Ullmann's Encyclopedia of Industrial Chemistry, 2007, Wiley-VCH, Weinheim. doi:10.1002/14356007.a12_571.pub2
- ↑ Hans-Wilhelm Engels et al., "Rubber, 4. Chemicals and Additives" in Ullmann's Encyclopedia of Industrial Chemistry, 2007, Wiley-VCH, Weinheim. doi:10.1002/14356007.a23_365.pub2
- ↑ "Chemical Tests". The British Lichen Society. http://www.britishlichensociety.org.uk/identification/chemical-tests.
- ↑ p-Phenylenediamine, U.S. Environmental Protection Agency
- ↑ Rollison, DE; Helzlsouer, KJ; Pinney, SM (2006). "Personal hair dye use and cancer: a systematic literature review and evaluation of exposure assessment in studies published since 1992.". Journal of Toxicology and Environmental Health Part B: Critical Reviews 9 (5): 413–39. doi:10.1080/10937400600681455. PMID 17492526. Bibcode: 2006JTEHB...9..413R.
- ↑ Zug, Kathryn A.; Warshaw, Erin M.; Fowler, Joseph F.; Maibach, Howard I.; Belsito, Donald L.; Pratt, Melanie D.; Sasseville, Denis; Storrs, Frances J. et al. (2009). "Patch-test results of the North American Contact Dermatitis Group 2005-2006". Dermatitis: Contact, Atopic, Occupational, Drug 20 (3): 149–160. doi:10.2310/6620.2009.08097. ISSN 2162-5220. PMID 19470301. https://pubmed.ncbi.nlm.nih.gov/19470301/.
- ↑ "The NIOSH Pocket Guide to Chemical Hazards". https://www.cdc.gov/niosh/ipcsneng/neng0805.html.
- ↑ "NIOSH Registry of Toxic Effects of Chemical Substances (RTECS) entry for p-Phenylenediamine (PPD)". https://www.cdc.gov/niosh/rtecs/ss7ad550.html.
- ↑ Ashraf, W.; Dawling, S.; Farrow, L. J. (1994). "Systemic Paraphenylenediamine (PPD) Poisoning: A Case Report and Review". Human & Experimental Toxicology 13 (3): 167–70. doi:10.1177/096032719401300305. PMID 7909678. Bibcode: 1994HETox..13..167A.
- ↑ Omer Sultan, Muhammad; Inam Khan, Muhammad; Ali, Rahmat; Farooque, Umar; Hassan, Syed Adeel; Karimi, Sundas; Cheema, Omer; Pillai, Bharat et al. (2020-05-29). "Paraphenylenediamine (Kala Pathar) Poisoning at the National Poison Control Center in Karachi: A Prospective Study". Cureus 12 (5): e8352. doi:10.7759/cureus.8352. ISSN 2168-8184. PMID 32617225.
 |