Chemistry:Ethyl maltol
From HandWiki
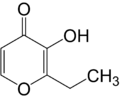
| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Ethyl-3-hydroxy-4H-pyran-4-one | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| C7H8O3 | |
| Molar mass | 140.138 g·mol−1 |
| Appearance | White crystalline powder |
| Melting point | 85 to 95 °C (185 to 203 °F; 358 to 368 K)[1] |
| Boiling point | 161 °C (322 °F; 434 K) |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H302 | |
| P264, P270, P301+312, P330, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Ethyl maltol is an organic compound that is a common flavourant in some confectioneries. It is related to the more common flavorant maltol by replacement of the methyl group by an ethyl group.[2] It is a white solid with a sweet smell that can be described as caramelized sugar or as caramelized fruit. Ethyl maltol is often used in perfumery to create gourmand scents. Its aroma is described as "sweet, caramellic, jammy, strawberry, cotton candy, berry, sugar".[3] Ethyl maltol was first used in Mugler's Angel perfume in 1992.[4]
The conjugate base derived from ethylmaltol, again like maltol, has a high affinity for iron, forming a red coordination complex . In such compounds, the heterocycle is a bidentate ligand.
Original patent:[5]
References
- ↑ Ethyl maltol at Sigma-Aldrich
- ↑ Erich Lück and Gert-Wolfhard von Rymon Lipinski "Foods, 3. Food Additives" in Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim. doi: 10.1002/14356007.a11_561
- ↑ "ethyl maltol". Scents and Flavors. https://scentsandflavors.com/database/9dbb4f66-d081-430c-858a-95a9de6ed907.
- ↑ "Сладкие ароматы в парфюмерии - Лучшие сладкие духи для мужчин и женщин с фото - Гурманские парфюмы.". https://gourmandperfumes.ru/.
- ↑ Charles R Stephens Jr & Robert P Allingham, U.S. Patent 3,446,629 (1969 to Pfizer Inc).
 |
