Chemistry:Formononetin
From HandWiki
| |
| Error creating thumbnail: Unable to save thumbnail to destination | |
| Names | |
|---|---|
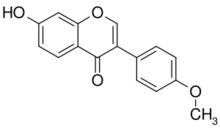
| IUPAC name
7-Hydroxy-4′-methoxyisoflavone
| |
| Preferred IUPAC name
7-Hydroxy-3-(4-methoxyphenyl)-4H-1-benzopyran-4-one | |
| Other names
Biochanin B
Formononetol 4'-O-methyldaidzein | |
| Identifiers | |
3D model (JSmol)
|
|
| 237979 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C16H12O4 | |
| Molar mass | 268.26 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Formononetin is an O-methylated isoflavone.
Natural occurrences
Formononetin is found in a number of plants and herbs such as red clover.[1] Along with other phytoestrogens, it predominantly occurs in leguminous plants and Fabaceae, particularly in beans, such as green beans, lima beans, soy and many others, as the free aglycone or in form of its glucoside ononin.[2]
It can also be found in Maackia amurensis cell cultures.[3]
Pharmacodynamics
Formononetin promotes angiogenesis.[4]
Derivatives
Ononin is the 7-O-β-D-glucopyranoside of formononetin.[5]
References
- ↑ Medjakovic, S.; Jungbauer, A. (2008). "Red Clover Isoflavones Biochanin A and Formononetin are Potent Ligands of the Human Aryl Hydrocarbon Receptor". The Journal of Steroid Biochemistry and Molecular Biology 108 (1–2): 171–177. doi:10.1016/j.jsbmb.2007.10.001. PMID 18060767.
- ↑ "Iowa State University Database on the Isoflavone Content of Foods, Release 1.3". USDA. 2002. http://www.nal.usda.gov/fnic/foodcomp/Data/isoflav/isoflav.html.
- ↑ Fedoreyev, S.A; Pokushalova, T.V; Veselova, M.V; Glebko, L.I; Kulesh, N.I; Muzarok, T.I; Seletskaya, L.D; Bulgakov, V.P et al. (2000). "Isoflavonoid production by callus cultures of Maackia amurensis". Fitoterapia 71 (4): 365–72. doi:10.1016/S0367-326X(00)00129-5. PMID 10925005.
- ↑ Li, Shang; Dang, Yuanye; Zhou, Xuelin; Huang, Bin; Huang, Xiaohui; Zhang, Zherui; Kwan, Yiu Wa; Chan, Shun Wan et al. (2015). "Formononetin promotes angiogenesis through the estrogen receptor alpha-enhanced ROCK pathway". Scientific Reports 5: 16815. doi:10.1038/srep16815. PMID 26568398.
- ↑ You-Ping Zhu (1998-05-28). Chinese Materia Medica: Chemistry, Pharmacology and Applications. p. 622. ISBN 9057022850. https://archive.org/details/chinesemateriame0000zhuy.
