Chemistry:Guanidine nitrate
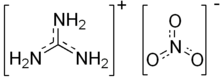
| |
| |
| Names | |
|---|---|
| IUPAC name
Guanidinium nitrate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1467 |
| |
| |
| Properties | |
| CH 5N 3 · HNO 3 | |
| Molar mass | 122.084 g·mol−1 |
| Appearance | White solid |
| Density | 1.44 g/cm3[1] |
| Melting point | 213–215 °C (415–419 °F; 486–488 K)[1] |
| Boiling point | Decomposes over 250 °C (482 °F; 523 K)[2] |
| 50 mg/mL[1] | |
| Hazards[1] | |
| Safety data sheet | Sigma-Aldrich SDS |
| GHS pictograms |  
|
| GHS Signal word | Warning |
| H272, H302, H319, H412 | |
| P210, P220, P221, P264, P270, P273, P280, P301+312+330Script error: No such module "Preview warning".Category:GHS errors, P305+351+338, P337+313, P370+378, P501 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
730 mg/kg Oral, rat, female |
LC50 (median concentration)
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
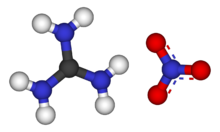
Guanidine nitrate is the chemical compound with the formula CH
5N
3 · HNO
3 (linear formula NH
2C(=NH)NH
2 · HNO
3).[1] It is a colorless, water-soluble salt. It is produced on a large scale and finds use as precursor for nitroguanidine,[3] fuel in pyrotechnics and gas generators. Its correct name is guanidinium nitrate, but the colloquial term guanidine nitrate is widely used.[1][2]
Production and properties
Although it is the salt formed by neutralizing guanidine with nitric acid, guanidine nitrate is produced industrially by the reaction of dicyandiamide (or calcium salt) and ammonium nitrate.[4]
It has been used as a monopropellant in the Jetex engine for model airplanes. It is attractive because it has a high gas output and low flame temperature. It has a relatively high monopropellant specific impulse of 177 seconds (1.7 kN·s/kg).[citation needed]
Guanidine nitrate's explosive decomposition is given by the following equation:[citation needed]
- [C(NH
2)
3]NO
3 (s) → 3 H
2O (g) + 2 N
2 (g) + C (s)
Uses
Guanidine nitrate is used as the gas generator in automobile airbags.[5] It is less toxic than the mixture used in older airbags of sodium azide, potassium nitrate and silica (NaN
3, KNO
3, and SiO
2), and it is less explosive and sensitive to moisture compared to the very cheap ammonium nitrate (NH
4NO
3).[6]
Safety
The compound is a hazardous substance, and is both a fuel and oxidizer. It is also harmful to the eyes, skin, and respiratory tract.[4]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 Sigma-Aldrich Co., Guanidine nitrate. Retrieved on 23 August 2025.
- ↑ 2.0 2.1 2.2 "Guanidine nitrate SDS" (in en). ThermoFisher Scientific. 24 December 2021. p. 3. https://www.fishersci.com/store/msds?partNumber=AC215310010&productDescription=GUANIDINE+NITRATE%2C+98%25+1KG&vendorId=VN00032119&countryCode=US&language=en.
- ↑ Koch, Ernst‐Christian (March 2019). "Insensitive High Explosives: III. Nitroguanidine – Synthesis – Structure – Spectroscopy – Sensitiveness". Propellants, Explosives, Pyrotechnics 44 (3): 267–292. doi:10.1002/prep.201800253.
- ↑ 4.0 4.1 Güthner, Thomas; Mertschenk, Bernd; Schulz, Bernd. "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a12_545.pub2.
- ↑ Tabuchi, Hiroko (2016-08-26). "A Cheaper Airbag, and Takata's Road to a Deadly Crisis" (in en-US). The New York Times. ISSN 0362-4331. https://www.nytimes.com/2016/08/27/business/takata-airbag-recall-crisis.html.
- ↑ Halford, Bethany (November 15, 2022). "What chemicals make airbags inflate, and how have they changed over time?". Chemical & Engineering News 100 (41). https://cen.acs.org/safety/chemicals-make-airbags-inflate-changed/100/i41. Retrieved 4 June 2023. "The chemical reaction used to deploy airbags has evolved, but one iteration resulted in massive recalls".

External links
- Jetex: Propellants
 |

