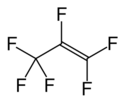
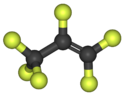
Chemistry:Hexafluoropropylene
From HandWiki
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1,1,2,3,3,3-Hexafluoroprop-1-ene | |||
| Other names
Perfluoropropene,
Perfluoropropylene, freon R 1216, halocarbon R 1216, fluorocarbon 1216 | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1858 | ||
| |||
| |||
| Properties | |||
| C3F6 | |||
| Molar mass | 150.023 g·mol−1 | ||
| Appearance | Colorless, odorless gas | ||
| Density | 1.332 g/ml, liquid at 20 °C | ||
| Melting point | −153 °C (−243 °F; 120 K) | ||
| Boiling point | −28 °C (−18 °F; 245 K) | ||
| Insoluble | |||
| Hazards | |||
| Main hazards | Suffocation | ||
| GHS pictograms |   
| ||
| GHS Signal word | Warning | ||
| H280, H332, H335, H351, H371, H373 | |||
| P201, P202, P260, P261, P264, P270, P271, P281, P304+312, P304+340, P308+313, P309+311, P312, P314, P403+233, P405, P410+403, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | Non flammable gas | ||
| Related compounds | |||
Related alkenes;
organofluorides |
propylene; Hexafluoroacetone, Hexafluoro-2-propanol | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Tracking categories (test):
Hexafluoropropylene is the fluoroalkene with the formula CF3CF=CF2. It is the perfluorocarbon counterpart to the hydrocarbon propylene. It is mainly used to produce copolymers with tetrafluoroethylene. Hexafluoropropylene is used as a chemical intermediate.[1]
Preparation
Hexafluoropropylene can be produced by pyrolysis of tetrafluoroethylene:[1][2]
- 3 CF2=CF2 → 2 CF3CF=CF2
It can also be prepared from chlorodifluoromethane, or produced from various chlorofluorocarbons.[3]
References
- ↑ 1.0 1.1 Siegemund, Günter; Schwertfeger, Werner; Feiring, Andrew; Smart, Bruce; Behr, Fred; Vogel, Herward; McKusick, Blaine; Kirschtitle=Fluorine Compounds, Organic, Peer (2016). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a11_349.pub2.
- ↑ Lehmler, HJ (March 2005). "Synthesis of environmentally relevant fluorinated surfactants—a review". Chemosphere 58 (11): 1471–96. doi:10.1016/j.chemosphere.2004.11.078. PMID 15694468. Bibcode: 2005Chmsp..58.1471L.
- ↑ James L. Webster, Elrey L. McCann, Douglas W. Bruhnke, Jan J. Lerou, "Multistep synthesis of hexafluoropropylene", States5043491A United States patent patent (expires 5-20-2020) 5043491A, published 1991-08-27, issued 1991-08-27, assigned to E. I. Du Pont de Nemours and Company