Chemistry:Iodate
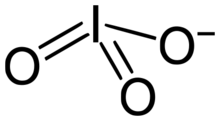 The iodate anion, IO−
3 | |
| Error creating thumbnail: Space-filling model of the iodate anion
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChEBI |
|
| ChemSpider | |
| 1676 | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| IO3− | |
| Molar mass | 174.902 g·mol−1 |
| Related compounds | |
Related compounds
|
Periodate, Fluoroiodate, Bromate, Chlorate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
An iodate is the polyatomic anion with the formula IO−
3. It is the most common form of iodine in nature, as it comprises the major iodine-containing ores.[1] Iodate salts are often colorless. They are the salts of iodic acid.
Structure
Iodate is pyramidal in structure. The O–I–O angles range from 97° to 105°, somewhat smaller than the O–Cl–O angles in chlorate.[2]
Reactions
Redox
Iodate is one of several oxyanions of iodine, and has an oxidation number of +5. It participates in several redox reactions, such as the iodine clock reaction. Iodate shows no tendency to disproportionate to periodate and iodide, in contrast to the situation for chlorate.
Iodate is reduced by sulfite:[1]
- 6HSO−
3 + 2IO−
3 → 2I−
+ 6HSO−
4
Iodate oxidizes iodide:
- 5I−
+ IO−
3 + 3H
2SO
4 → 3I
2 + 3H
2O + 3SO
4^−
2
Similarly, chlorate oxidizes iodide to iodate:
- I−
+ ClO−
3 → Cl−
+ IO−
3
Iodate is also obtained by reducing a periodate with a sulfide. The byproduct of the reaction is a sulfoxide.[3]
Acid-base
Iodate is unusual in that it forms a strong hydrogen bond with its parent acid:[2]
- IO−
3 + HIO
3 → H(IO
3)−
2
The anion H(IO
3)−
2 is referred to as biiodate.
Principal compounds
- Calcium iodate, Ca(IO3)2, is the principal ore of iodine. It is also used as a nutritional supplement for cattle.
- Potassium iodate, KIO3, like potassium iodide, has been issued as a prophylaxis against radioiodine absorption in some countries.[4][5]
- Potassium hydrogen iodate (or potassium biiodate), KH(IO3)2, is a double salt of potassium iodate and iodic acid, as well as an acid itself.
- When some oxygen is replaced by fluorine, fluoroiodates are produced.
Natural occurrence
Minerals containing iodate are found in the caliche deposits of Chile. The most important iodate minerals are lautarite and brüggenite, but also copper-bearing iodates such as salesite are known.[6]
References
- ↑ 1.0 1.1 Lyday, Phyllis A. (2005). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. pp. 382–390. doi:10.1002/14356007.a14_381.
- ↑ 2.0 2.1 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ↑ Qiu, Chao; Sheng Han; Xingguo Cheng; Tianhui Ren (2005). "Distribution of Thioethers in Hydrotreated Transformer Base Oil by Oxidation and ICP-AES Analysis" (abstract). Industrial & Engineering Chemistry Research 44 (11): 4151–4155. doi:10.1021/ie048833b. http://pubs.acs.org/cgi-bin/abstract.cgi/iecred/2005/44/i11/abs/ie048833b.html. Retrieved 2007-05-03. "Thioethers can be oxidized to sulfoxides by periodate, and periodate is reduced to iodate".
- ↑ "Radiological Protection Institute of Ireland | | Media | Press releases | Radioactivity released from Wylfa nuclear power plant is extremely low and of no health significance". http://www.rpii.ie/Site/Media/Press-Releases/Radioactivity-released-from-Wylfa-nuclear-power-pl.aspx.
- ↑ "Decision to Discontinue the Future Distribution of Iodine Tablets". http://www.dohc.ie/press/releases/2008/20080403c.html.
- ↑ "Home". http://www.mindat.org/.
 |
