Chemistry:Lead polonide
From HandWiki
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| PbPo | |
| Molar mass | 416 g·mol−1 |
| Appearance | black crystals |
| Density | 9.64 g·cm−3[1] |
| Melting point | 550–630 °C(decomposes)[2] |
| Related compounds | |
Other anions
|
|
Other cations
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Lead polonide is the polonide of lead, with the chemical formula of PbPo. It occurs naturally, as lead is produced in the alpha decay of polonium.[3]
Preparation
Lead polonide can be formed by reacting polonium vapour and lead under a vacuum.[4]
Properties
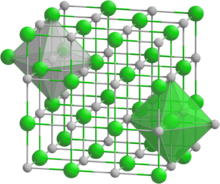
Lead polonide has a sodium chloride structure, which is the same as lead telluride. It has a cubic crystal structure, with the space group Fm3m (No. 225), with lattice constant a = 6.59 Å.[5]
References
- ↑ Moyer, Harvey V. (1956), Chemical Properties of Polonium, pp. 96, doi:10.2172/4367751
- ↑ Miura, Terumitsu; Obara, Toru; Sekimoto, Hiroshi (Nov 2007), "Experimental verification of thermal decomposition of lead polonide", Annals of Nuclear Energy 34 (11): 926–930, doi:10.1016/j.anucene.2007.05.009, Bibcode: 2007AnNuE..34..926M
- ↑ Weigel, F. (1959). "Chemie des Poloniums". Angewandte Chemie 71 (9): 289–316. doi:10.1002/ange.19590710902. Bibcode: 1959AngCh..71..289W.
- ↑ Witteman, W. G.; Giorgi, A. L.; Vier, D. T. (1960). "THE PREPARATION AND IDENTIFICATION OF SOME INTERMETALLIC COMPOUNDS OF POLONIUM1". The Journal of Physical Chemistry 64 (4): 434–440. doi:10.1021/j100833a014. ISSN 0022-3654. https://pubs.acs.org/doi/abs/10.1021/j100833a014.
- ↑ Dalven, Richard (Dec 1973), Recent Studies Of Lead Polonide (PbPo), Lawrence Berkeley National Laboratory, https://escholarship.org/uc/item/4fr8487j
 |
