Chemistry:Methyl phenylacetate
From HandWiki
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
Methyl phenylacetate | |
| Other names
Methyl 2-phenylacetate
Methyl benzene acetate | |
| Identifiers | |
3D model (JSmol)
|
|
| 878795 | |
| ChemSpider | |
| EC Number |
|
| MeSH | C024906 |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C9H10O2 | |
| Molar mass | 150.1745 g mol−1 |
| Appearance | Colorless liquid |
| Density | 1.055±0.060 g/cm3 |
| Melting point | 50 °C (122 °F; 323 K) |
| Boiling point | 218 °C (424 °F; 491 K) |
| 2070 mg/L | |
| Vapor pressure | 17.3 Pa |
| −92.73×10−6 cm3/mol | |
Refractive index (nD)
|
1.505±0.020 at 20 °C |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 90.6 °C (195.1 °F; 363.8 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
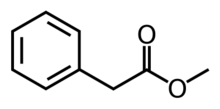
Methyl phenylacetate is an organic compound that is the methyl ester of phenylacetic acid, with the structural formula C
6H
5CH
2CO
2CH
3. It is a colorless liquid that is only slightly soluble in water, but soluble in most organic solvents.
Methyl phenylacetate has a strong odor similar to honey. This compound also occurs in brandy, capsicum, coffee, honey, pepper, and some wine. It is used in the flavor industry and in perfumes to impart honey scents.[1]
Methyl phenyldiazoacetate, precursor to cyclopropanation agents, is prepared by treating methyl phenylacetate with p-acetamidobenzenesulfonyl azide in the presence of base.[2]
References
- ↑ "Methyl Phenyl Acetate". The Good Scents Company. February 22, 2007. http://www.thegoodscentscompany.com/data/rw1008431.html.
- ↑ Davies, Huw M. L.; Hu, Wen-hao; Xing, Dong (2015-04-01). "Methyl Phenyldiazoacetate". Encyclopedia of Reagents for Organic Synthesis. John Wiley & Sons Ltd. pp. 1–10. doi:10.1002/047084289X.rn00444.pub2. ISBN 9780470842898.
 |

