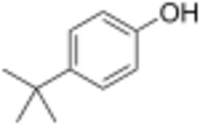
Chemistry:Para tertiary butylphenol
| |
| Names | |
|---|---|
| IUPAC name
4-tert-Butylphenol
| |
| Other names
p-tert-Butylphenol; Butylphen
| |
| Identifiers | |
3D model (JSmol)
|
|
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| Properties | |
| C10H14O | |
| Molar mass | 150.221 g·mol−1 |
| Density | 0.908 g/cm3 (20 °C)[1] |
| Melting point | 100 °C (212 °F; 373 K)[1] |
| Boiling point | 237 °C (459 °F; 510 K)[1] |
| 0.6 g/L (20 °C)[1] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Para tertiary butylphenol or 4-tert-butylphenol is an organic aromatic compound, specifically a substituted phenol. It has the CAS Registry Number 98-54-4 and a formula of C10H14O.[2] The International Union of Pure and Applied Chemistry (IUPAC) name is 4-tert-butylphenol. It has a number of synonyms including para tertiary butyl phenol.
Properties
The material is a white crystalline solid with a distinct phenolic odor. It has a reported melting point between 96 and 101 °C, depending on the source. NIH/HMDB lists it as 99 °C, EPA lists it as 98 °C, OSHA as 98 °C, DIC corporation as 97 °C, and Sigma-Aldrich as 96-101 °C. OSHA records show it as an irritant and has developed suggested test methods for its detection.[3] A European Union risk assessment report into the molecule dated May 2006 suggested it had potential depigmenting properties and was a potential endocrine disruptor. This was done under its EINECS number of 202-679-0.[4] Other papers have been published showing similar findings.[5]
Manufacture
The usual method of manufacture is by reaction of phenol and isobutylene.[6][7] Another method using phenol and tert-butanol in water has also been reported.[8]
Uses
It major uses are in the production of epoxy resins and curing agents and also in polycarbonate resins. It has also found use in the production of phenolic resins. Another use is in the production of para tertiary butylphenol formaldehyde resin. It has also found use as a plasticizer.
Bisphenol A is difunctional and used to produce epoxy resin and polycarbonate. Para tertiary butylphenol is monofunctional and so in polymer science terms, bisphenol A is a polymer chain extender but para tertiary butyl phenol is a chain stopper or sometimes called endcapper. It is thus use to control molecular weight by limiting chain growth.
Para tertiary butyl phenol has an OH group and so it may be reacted with epichlorohydrin and caustic soda to produce the glycidyl ether which is used in epoxy resin chemistry. This molecule has the CAS Registry number of 3101-60-8.[9][10]
References
- ↑ 1.0 1.1 1.2 1.3 Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ↑ PubChem. "4-Tert-Butylphenol" (in en). https://pubchem.ncbi.nlm.nih.gov/compound/7393.
- ↑ "Sampling and Analytical Methods: p-tert-Butylphenol, PV2085". https://www.osha.gov/dts/sltc/methods/partial/t-pv2085-01-9203-ch/t-pv2085-01-9203-ch.html.
- ↑ European Union Risk Assessment report P-TERTIARY-BUTYLPHENOL May 2006 carried out in Norway
- ↑ Meier, Sonnich; Andersen, Thorny Cesilie; Lind-Larsen, Kristin; Svardal, Asbjørn; Holmsen, Holm (April 2007). "Effects of alkylphenols on glycerophospholipids and cholesterol in liver and brain from female Atlantic cod (Gadus morhua)". Comparative Biochemistry and Physiology. Toxicology & Pharmacology 145 (3): 420–430. doi:10.1016/j.cbpc.2007.01.012. ISSN 1532-0456. PMID 17344102. https://pubmed.ncbi.nlm.nih.gov/17344102.
- ↑ Japanese patent JP2003040822A in English published September 18 2008
- ↑ "4-Tertiary Butylphenol | Business & Products" (in en). https://www.dic-global.com/en/products/alkylphenol/ptbp/.
- ↑ Organic reactions in water : principles, strategies and applications. U. Marcus Lindström. Oxford: Blackwell Pub. 2007. ISBN 978-0-470-98881-7. OCLC 184983451. https://www.worldcat.org/oclc/184983451.
- ↑ "CAS Common Chemistry". https://commonchemistry.cas.org/detail?cas_rn=3101-60-8.
- ↑ "4-tert-butylphenyl glycidyl ether" (in en). https://www.wikidata.org/wiki/Q27276612.
External links

