Chemistry:PyAOP reagent
| |
| Names | |
|---|---|
| IUPAC name
(7-Azabenzotriazol-1-yloxy)tripyrrolidinophosphonium hexafluorophosphate
| |
| Other names
PyAOP
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C17H27F6N7OP2 | |
| Molar mass | 521.389 g·mol−1 |
| Appearance | White crystals |
| Melting point | 163–168 °C (325–334 °F; 436–441 K) |
| Hazards | |
| Main hazards | Irritant |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
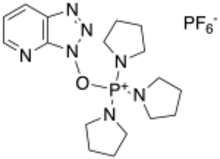
PyAOP ((7-azabenzotriazol-1-yloxy)tripyrrolidinophosphonium hexafluorophosphate) is a reagent used to prepare amides from carboxylic acids and amines in the context of peptide synthesis.[1] It can be prepared from 1-hydroxy-7-azabenzotriazole (HOAt) and a chlorophosphonium reagent under basic conditions.[2] It is a derivative of the HOAt family of amide bond forming reagents. It is preferred over HATU, because it does not engage in side reactions with the N-terminus of the peptide.[3] Compared to the HOBt-containing analog PyBOP, PyAOP is more reactive due to the additional nitrogen in the fused pyridine ring of the HOAt moiety.[4] Thermal hazard analysis by differential scanning calorimetry (DSC) shows PyAOP is potentially explosive.[5]
See also
References
- ↑ Mansour, Tarek S.; Bardhan, Sujata; Wan, Zhao-Kui (2010). "Phosphonium- and Benzotriazolyloxy-Mediated Bond-Forming Reactions and Their Synthetic Applications" (in en). Synlett 2010 (8): 1143–1169. doi:10.1055/s-0029-1219820. ISSN 0936-5214. http://www.thieme-connect.de/DOI/DOI?10.1055/s-0029-1219820.
- ↑ Hoffmann, Frank; Jäger, Lothar; Griehl, Carola (2003-02-01). "Synthesis and Chemical Constitution of Diphenoxyphosphoryl Derivatives and Phosphonium Salts as Coupling Reagents for Peptide Segment Condensation" (in en). Phosphorus, Sulfur, and Silicon and the Related Elements 178 (2): 299–309. doi:10.1080/10426500307942. ISSN 1042-6507. https://www.tandfonline.com/doi/full/10.1080/10426500307942.
- ↑ Albericio, F.; Cases, M.; Alsina, J.; Triolo, S. A.; Carpino, L. A; Kates, S. (1997). "On the use of PyAOP, a phosphonium salt derived from HOAt, in solid-phase peptide synthesis". Tetrahedron Letters 38 (27): 4853–4856. doi:10.1016/S0040-4039(97)01011-3.
- ↑ Albericio, Fernando; Bofill, Josep M.; El-Faham, Ayman; Kates, Steven A. (1998). "Use of Onium Salt-Based Coupling Reagents in Peptide Synthesis1". The Journal of Organic Chemistry (American Chemical Society) 63 (26): 9678–9683. doi:10.1021/jo980807y. ISSN 0022-3263.
- ↑ Sperry, Jeffrey B.; Minteer, Christopher J.; Tao, JingYa; Johnson, Rebecca; Duzguner, Remzi; Hawksworth, Michael; Oke, Samantha; Richardson, Paul F. et al. (2018-09-21). "Thermal Stability Assessment of Peptide Coupling Reagents Commonly Used in Pharmaceutical Manufacturing" (in en). Organic Process Research & Development 22 (9): 1262–1275. doi:10.1021/acs.oprd.8b00193. ISSN 1083-6160. https://pubs.acs.org/doi/10.1021/acs.oprd.8b00193.
 |
