Chemistry:Trinitromethane
| |
| |
| Names | |
|---|---|
| IUPAC name
Trinitromethane
| |
| Other names
Nitroform
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| CHN3O6 | |
| Molar mass | 151.04 g/mol |
| Appearance | Pale yellow crystals |
| Density | 1.469 g/cm3 |
| Melting point | 15 °C (59 °F; 288 K) |
| 44g/100ml at 20 °C | |
| Acidity (pKa) | 0.25 (see text) |
| Hazards | |
| Main hazards | Oxidant, Explosive (esp. in contact with metals), Corrosive. |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Related compounds
|
Hexanitroethane Octanitropentane Tetranitromethane |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
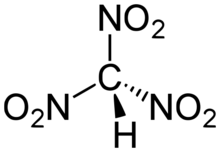
Trinitromethane, also referred to as nitroform, is a nitroalkane and oxidizer with chemical formula HC(NO2)3. It was first obtained in 1857 as the ammonium salt by the Russian chemist Leon Nikolaevich Shishkov (1830–1908).[2][3] In 1900, it was discovered that nitroform can be produced by the reaction of acetylene with anhydrous nitric acid.[4] This method went on to become the industrial process of choice during the 20th century. In the laboratory, nitroform can also be produced by hydrolysis of tetranitromethane under mild basic conditions.[5]
Acidity
Trinitromethane as a neutral molecule is colorless. It is highly acidic, easily forming an intensely yellow anion, (NO2)3C−. The pKa of trinitromethane has been measured at 0.17 ± 0.02 at 20 °C, which is remarkably acidic for a methane derivative.[6] Trinitromethane easily dissolves in water to form an acidic yellow solution.
There is some evidence that the anion, which obeys the 4n+2 Hückel rule, is aromatic.[7]
Nitroform salts
Trinitromethane forms a series of bright yellow ionic salts. Many of these salts tend to be unstable and can be easily detonated by heat or impact.
The potassium salt of nitroform, KC(NO2)3 is a lemon yellow crystalline solid that decomposes slowly at room temperatures and explodes above 95 °C. The ammonium salt is somewhat more stable, and deflagrates or explodes above 200 °C. The hydrazine salt, hydrazinium nitroformate is thermally stable to above 125 °C and is being investigated as an ecologically friendly oxidizer for use in solid fuels for rockets.
References
- ↑ Budavari, Susan, ed. (1996), An Encyclopedia of Chemicals, Drugs, and Biologicals (12th ed.), Merck, ISBN 0911910123, 9859.
- ↑ For biographical information about Leon Nikolaevich Shishkov, see The Free Dictionary
- ↑ See:
- Chichkoff, Léon (1857). "Note sur le nitroforme" (in French). Comptes rendus 45: 144–146. https://babel.hathitrust.org/cgi/pt?id=mdp.39015035405979&view=1up&seq=154.
- Schischkoff, L. (1857). "Ueber das Nitroform" (in German). Annalen der Chemie 103 (3): 364–366. doi:10.1002/jlac.18571030316. https://babel.hathitrust.org/cgi/pt?id=uva.x002457949&view=1up&seq=378.
- Abstracted in: Schischkoff, L. (1857). "On nitroform". The Chemical Gazette 15: 448. https://books.google.com/books?id=xQcAAAAAMAAJ&pg=PA448.
- Abstracted in: Schischkoff, L. (1858). "On nitroform". Philosophical Magazine. 4th series 15: 302. https://books.google.com/books?id=1bZJAQAAMAAJ&pg=PA302.
- ↑ The Italian chemist Adolfo Baschieri discovered that nitroform (Italian: nitroformio) could be produced from acetylene (acetilene) and nitric acid (acido nitrico).
- Baschieri, A. (1900). "Sul comportamento dell' acetilene con alcuni ossidanti" (in Italian). Atti della Reale Accademia dei Lincei. 5th series 9 (part I): 391–393. https://www.biodiversitylibrary.org/item/155899#page/397/mode/1up. See p. 392.
- Abstracted in German in: Baschieri, A. (1900). "Über das Verhalten des Acetylens gegen einige Oxydationsmittel" (in German). Chemisches Zentralblatt. 5th series 4 (part II): 528. https://babel.hathitrust.org/cgi/pt?id=mdp.39015020184084&view=1up&seq=576.
- See also: Nieuwland, Julius A.; Vogt, Richard R. (1945). The Chemistry of Acetylene. New York City, New York, USA: Reinhold Publishing Corp.. p. 158. https://archive.org/details/in.ernet.dli.2015.239313.
- ↑ Gakh, A. A.; Bryan, J. C.; Burnett, M. N.; Bonnesen, P. V. (2000). "Synthesis and structural analysis of some trinitromethanide salts". Journal of Molecular Structure 520 (1–3): 221–228. doi:10.1016/S0022-2860(99)00333-6. Bibcode: 2000JMoSt.520..221G. https://zenodo.org/record/1259627.
- ↑ Novikov, S. S.; Slovetskii, V. I.; Shevelev, S. A.; Fainzilberg, A. A. (1962). "Spectrophotometric Determination of the Dissociation Constants of Aliphatic Nitro Compounds". Russian Chemical Bulletin 11 (4): 552–559. doi:10.1007/BF00904751.
- ↑ Cioslowski, J.; Mixon, S. T.; Fleischmann, E. D. (1991). "Electronic structures of trifluoro-, tricyano-, and trinitromethane and their conjugate bases". Journal of the American Chemical Society 113 (13): 4751–4755. doi:10.1021/ja00013a007.
 |

