Chemistry:Xylenol orange
From HandWiki
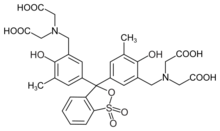
| |
| Names | |
|---|---|
| Preferred IUPAC name
2,2′,2′′,2′′′-{(1,1-Dioxo-2,1λ6-benzoxathiole-3,3(1H)-diyl)bis[(6-hydroxy-5-methyl-3,1-phenylene)methylenenitrilo]}tetraacetic acid | |
| Other names
XO[1]
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII |
|
| |
| |
| Properties | |
| C31H32N2O13S | |
| Molar mass | 672.66 g·mol−1 |
| Melting point | 195 °C (383 °F; 468 K) |
| 200 mg/mL | |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+352, P304+340, P305+351+338, P312, P321, P332+313, P337+313, P362, P403+233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | > 93 °C (199 °F; 366 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Xylenol orange is an organic reagent, most commonly used as a tetrasodium salt as an indicator for metal titrations. When used for metal titrations, it will appear red in the titrand and become yellow once it reaches its endpoint. Historically, commercial preparations of it have been notoriously impure,[2] sometimes consisting of as little as 20% xylenol orange, and containing large amounts of semi-xylenol orange and iminodiacetic acid. Purities as high as 90% are now available.
It is fluorescent, and has excitation maximums of 440 & 570 nm and an emission maximum of 610 nm.[3]
References
- ↑ Li, Zhong-Guang (2019-01-01), Khan, M. Iqbal R.; Reddy, Palakolanu Sudhakar; Ferrante, Antonio et al., eds., "Chapter 5 - Measurement of Signaling Molecules Calcium Ion, Reactive Sulfur Species, Reactive Carbonyl Species, Reactive Nitrogen Species, and Reactive Oxygen Species in Plants", Plant Signaling Molecules (Woodhead Publishing): pp. 83–103, ISBN 978-0-12-816451-8, https://www.sciencedirect.com/science/article/pii/B9780128164518000058, retrieved 2023-11-11
- ↑ Gay, Craig; Collins, James; Gebicki, Janusz M. (1999), "Determination of Iron in Solutions with the Ferric–Xylenol Orange Complex", Analytical Biochemistry 273 (2): 143–148, doi:10.1006/abio.1999.4207, PMID 10469483
- ↑ "Fluorescence - Fluorochrome Data Tables". 2018-09-11. https://micro.magnet.fsu.edu/primer/techniques/fluorescence/fluorotable1.html.
 |

