Engineering:Bioprinting drug delivery
Bioprinting drug delivery is a method of using the three-dimensional printing of biomaterials through an additive manufacturing technique to develop drug delivery vehicles that are biocompatible tissue-specific hydrogels or implantable devices. 3D bioprinting uses printed cells and biological molecules to manufacture tissues, organs, or biological materials in a scaffold-free manner that mimics living human tissue to provide localized and tissue-specific drug delivery, allowing for targeted disease treatments with scalable and complex geometry.
This technique was first developed in the 1950s as patients with incurable diseases demanded higher organ transplantation needs than available donors and organ transplantations showed limitations with immune responses and organ rejection.[1] Techniques that have been studied include bioprinting hydrogels with various Bio-ink (cell-laden microgel) materials and bioprinting implantable devices that mimic specific tissues or biological functions. A few applications of these printed vehicles include promoting wound healing by delivering antibiotics, anti-inflammatory treatments, or drugs that promote cell differentiation and cell proliferation, providing anticancer treatments directly to tumors, and promoting or inhibiting angiogenesis and vascularization to treat cancer, arterial diseases, heart diseases, and arthritis. In addition, implants can be printed in unique shapes and forms to deliver drugs directly into targeted tissues. A new approach includes adding a fourth dimension to 3D bioprinting, which allows the printed biomaterials to conform, either folding or unfolding, to release drugs in a more controlled manner. Whether 3D or 4D, the application of bioprinting to the development of drug delivery systems allows for biocompatible, biodegradable, universal, and personalized drug vehicles.
Methods of 3D bioprinting
Layer-by-layer printing of biochemicals and living cells requires precise placement and viable materials. The basic technology of a bioprinter starts with data taken from computer-assisted design (CAD) or a similar program, uses motion control systems to control the X/Y/Z axis direction drive mechanisms along with a material control system for the Bio-ink printhead, and deposits material into a 3D construct.[2] Bioprinting can be done by material jetting, material extrusion, or vat polymerization.[2][3]
Material jetting
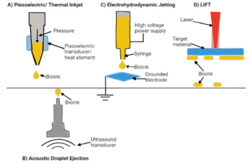
Material jetting, sometimes referred to as fused deposition modeling (FDM), is a method that involves depositing cells into a 3D bed using piezoelectric/thermal ink-jetting, acoustic wave jetting, electrohydrodynamic jetting, or laser-induced forward transfer (LIFT).[3] Piezoelectric/thermal ink-jetting is a process that uses the same non-contact process as a desktop inkjet printers by generating pressure pulsed into a nozzle to expel droplets. Acoustic wave jetting uses acoustic radiation force to produce droplets, electrohydrodynamic jetting uses electric voltage to form droplets, and LIFT is a nozzle-free jetting technique where a laser generates a high-pressure bubble that propels droplets.[3] These methods provide precise placement of the Bio-ink and enable scaffold-free bioprinting through the direct deposition of the Bio-ink.
Material extrusion
Another method of bioprinting is material extrusion. This is a mechanical-based method that uses motors to drive a piston downwards. The extrusion of the material is based on the rate of the motor’s displacement, where the difference between the supplied air and the ambient pressure drives the flow of the material through an angular turn of a rotary screw.[3]
Vat polymerization printing
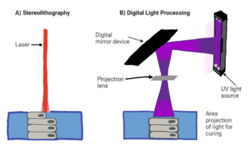
The last method of 3D bioprinting is vat polymerization printing (VPP). This method uses a cell-hydrogel suspension to form 3D structures, similar to the method of other additive manufacturing devices. The constructs are formed layer-by-layer through laser curing in stereolithography (SLA) or UV digital light processing (DLP) into the vat of a photopolymer using a micromirror device.[3]
3D bio-printed drug-loaded hydrogels
Hydrogels are three-dimensional polymeric networks that can maintain their structure while absorbing large amounts of water or biological fluids. These have been widely studied as bio-inks in 3D bioprinting due to their similarities to the human extracellular matrix (ECM) and their ability to encapsulate drugs. They are mainly printed using material jetting processes and material extrusion, where droplets of the hydrogel are dispensed.[3]
Alginate-based hydrogels
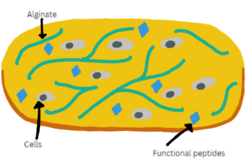
Hydrogels can be made of many different synthetic polymers or natural polysaccharides. A common polysaccharide used in biomedical hydrogel applications is alginate, a naturally occurring polyanionic copolymer.[1] The 3D structure and high water absorption of alginate provides a tissue environment that closely mimics human soft tissue. In addition, it is an ideal candidate for biomedical applications due to its natural biodegradability and biocompatibility. To construct alginate hydrogels, a layer-by-layer technique is used by assembling a series of negatively and positively charged polyelectrolytes. Alginate is used as the matrix in bio-ink that can be extruded from the bioprinter’s syringe with increasing shear, resulting in a tough hydrogel with low viscosity. Depending on the surrounding medium, alginate has the potential to form two different types of gels. When the pH is low, it shrinks and produces a viscous acidic gel, holding onto encapsulated drugs. Once the pH is raised, such as the medium inside an intestinal tract, the alginate turns into a viscose gel which causes drug dissolution and release. This process allows for a controlled and sustained release of drugs to specific tissues.
Researcher Faramarzi et al. used alginate-based hydrogels combined with the growth factor Platelet-Rich Plasma to develop a bio-ink with personalized biological factors.[4] Faramarzi, along with his fellow researchers, extracted this plasma from the blood of specific patients, then mixed it with the alginate solution. To turn this solution into a bio-ink, they coated it with calcium chloride agarose gel. The result was a hydrogel disk that had a decreased risk of immune responses from the host since it was specific to each patient. The disk also showed potential for promoting mesenchymal stem cells and endothelial cells in tissue healing. The researchers concluded that the Platelet-Rich Plasma and alginate hydrogel bio-ink could be used by any bioprinter to produce personalized drug delivery therapies.
Thus, due to its biocompatibility and its personalization, alginate provides a sufficient bio-ink candidate for drug-loaded hydrogel production. The hydrogel provides a foundation to be loaded with drugs while the alginate encapsulates the drugs and serves as a viable medium. The hydrogel can be loaded with whatever type of drug is necessary for the patient’s condition, and wherever the target tissue is located. This method of personalizing drug delivery reduces the immune responses of patients. The low toxicity and controllable factors of alginate make it a suitable candidate for hydrogel incorporation, resulting in a hydrogel that leverages the delivery of drugs, protects drugs with encapsulation, and allows for tunable drug release and degradability. Alginate hydrogels have been seen to deliver bortezomib, an anticancer drug, tetracycline hydrochloride and silver sulfadiazine, which are hydrophobic antibiotics, and simvastatin, which promotes local stem cell differentiation.[5]
Peptide-based hydrogels
Another bio-ink that has been studied for 3D bioprinting applications is low molecular weight self-assembling peptide-based materials. Peptide-based hydrogels are candidates for bio-inks since they show a resemblance to the ECM. In addition, their mechanical strength and stiffness of up to 40 kPa allow for strong and rigid hydrogels.[6]
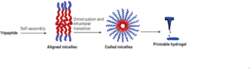
Xin Yang et al. experimented with using self-assembling peptide hydrogels in bioprinting applications. They used the “helical coiling of micelles induced by disulfide crosslinking in a lyotropic peptide liquid crystal” to produce a printable hydrogel.[7] They found that the tripeptide self-assembled into aligned micelles at high pH values, which produced a viscous solution, which could be transformed into a self-supporting hydrogel when the cross-linking of the sulfhydryl group of the side chain peptides increased the storage modulus of the solution.
These self-assembling peptides provide organization and strength that would be beneficial as a bio-ink for 3D bioprinting applications. In addition, their resemblance to the native cellular microenvironment and tunable mechanical strength allow them to support the proliferation of human stem cells. Using self-assembling peptides to print hydrogels would provide drug delivery vehicles that represent the ECM and provide the potentiality to differentiate primary cells into organotypic structures and deliver antimicrobial, anti-inflammatory, anticancer, and wound healing drugs.[6][7] Specifically, hydrogels made of these self-assembling peptides have been studied to encapsulate chemotherapy drugs that can disassemble and release the loaded drug under the stimulation of tumor environments, providing an alternative to the current limitations of typical chemotherapy which inevitably damages healthy cells while killing cancer cells[8]
Nanocellulose-based bio-inks
Another bio-ink that has been successful in producing drug delivery systems via bioprinting is cellulosic nanomaterials. Cellulose-based bio-inks are easily accessible, inexpensive, biodegradable, biocompatible, and highly stiff. It is a polysaccharide that is obtained from the biosynthesis of plants and bacteria. It is extracted from raw materials with mechanical shearing actions and biological treatments, such as hydrolysis, resulting in highly structured nanofibrils. Cellulose materials are defined by their high viscosity and shear-thinning behavior.[9]
Nanocellulose-based hydrogels
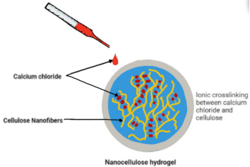
Researcher Adam Rees et al. studied the use of nanocellulose bio-inks as wound dressings.[10] They used extrusion by 3D bioprinting to develop porous structures with ionic calcium chloride cross-linking. These hydrogels were seen to support bacterial growth and incorporate and release antimicrobial drugs due to their porosity. These porous structures provide strong, moist environments that would be ideal for delivering drugs to tissues that require wound healing aid.
Nanocellulose-based implants
Cellulose nanofibrils have also been used as a bio-ink for non-hydrogel applications. Studies conducted by Kempin et al. evaluated the use of bioprinting cellulose as drug-loaded implants.[11] He used FDM to evaluate the drug release behavior of cellulose. Fluorescent dye quinine was used as a visual to see the distribution of drugs in the implants. He loaded the quinine filaments into bio-printed cellulose implants and then incubated the implants to observe their drug-release behaviors. His studies showed that around 5% of the quinine was released from the cellulose implant in 100 days. The findings showed that cellulose nanofibrils as a bio-ink for printing drug-loaded implants might provide implants with customizable shapes and controlled release of loaded drugs or antibiotics via FDM.
Protein-based hydrogels
Common protein-based bio-inks include collagen, keratin, gelatin, and silk. These proteins are advantageous since they are in the ECM and display good cytocompatibility, biocompatibility, and biodegradability. They are all derived from different natural sources, are isolated with different methods, and have different advantages and disadvantages. Collagen is typically printed using extrusion or SLA and provides good structural responses and adhesion for cells.[12][13][14] Silk is printed using digital light processing, and provides strength and robustness. However, a disadvantage of silk is its potential to conform in response to high shear forces.[12][13][14][15] Gelatin is printed using extrusion and provides good cellular affinity, however, studies have shown that limitations occur with the use of gelatin due to its covalent crosslinking-based stabilization which requires chemical reactions that are not cytocompatible.[12][13][14][15][16] Overall, protein-based bio-inks are abundant, inexpensive, biocompatible, and biodegradable, and have been commonly used for 3D bioprinting. Advantages of protein-based bio-inks over synthetic bio-inks include their similarity to human host tissue and their ability to match their degradation rate with the regeneration of host tissue.
| Protein | Source(s) | Printing Method | Advantages | Disadvantages |
|---|---|---|---|---|
| Silk | Silkworm | Digital light processing | High strength and robustness. High solubility and printing fidelity. | Conforms at high shear forces, so not suitable for printing at low concentrations. |
| Keratin | Spider, sheep wool, and human hair | Extrusion | High strength and fidelity. Good cell adherence and viability. | Can be difficult to achieve and have varying properties in humid air. |
| Gelatin | Gelatin | Extrusion | Good cellular affinity and proliferation. | Requires non-cytocompatible chemical reactions and has a risk of degradation at high temperatures. |
| Collagen | Porcine and fish | Extrusion or SLA | Good structural responses and cell adhesion. High porosity and tensile strength. | Possible lack of biocompatibility and immunogenicity concerns. |
Current applications of bio-printed protein-based hydrogels
Since proteins have been seen as effective bio-ink candidates for drug delivery hydrogels, several applications exist for using proteins as bio-inks for 3D printing. Bertassoni et al. experimented to bioprint cell-laden methacrylated gelatin (GelMA) hydrogels at concentrations ranging from 7 to 15% with varying cell densities.[17] Their experiment presents a strategy for “direct-write bioprinting of cell-laden photolabile ECM-derived hydrogels”.[17] They found a direct correlation between printability and hydrogel mechanical properties. A commercially available bioprinter was used to dispense the GelMA hydrogel fibers using digital light processing since GelMA is a photosensitive polymer. They found that the hydrogels provide cell viability for at least eight days, showing that cell-laden hydrogel tissue constructs can be bioprinting in multiple different ways to deliver different compounds[17]
Other research conducted on these hydrogels includes gelatin-sulfonated skin composite tissue that has been manufactured to deliver cells to open wounds by seeding matrices.[18][19] Doing this helps wounds to heal faster and more efficiently. Gelatin hydrogels have also successfully delivered fluorescein, a hydrophobic molecule, and microRNA to promote osteogenic cell differentiation.[16] Silk has successfully delivered aspirin, an anti-inflammatory drug that aids in wound dressing, and gentamicin, an antibiotic that also aids in wound dressing.[16]
3D bioprinted drug-loaded non-hydrogel implants
Another application of bioprinting in the field of drug delivery is non-hydrogel drug delivery systems. These systems allow localized delivery of personalized drugs. The implants are printed in the same additive manufacturing manner as hydrogels, using various flexible bio-inks, and then loaded with drugs and inserted into specific areas of the body. An example of this is seen in a study that was conducted to use SLA 3D printing to develop an implant that delivers drug systems to the ear.[20] Researchers printed several implants out of a flexible resin that contained 0.5% levofloxacin, then conducted several mechanical and in vitro tests to evaluate the ability of the implants to release the levofloxacin. The results showed no interaction between the resin and the drug, the resistance of the implant without compromise, and high antimicrobial activity. Using 3D printing to be able to print implants with complex shapes, antibiotics were able to be delivered efficiently directly to the inner ear to perform antimicrobial actions on inner ear infections.
Researchers have also studied 3D printing a bladder device for intravesical drug delivery.[21] This method takes a slightly different approach than the above-mentioned procedure of loading an implant with a specific drug. Instead, intravesical instillation provides an alternative to oral medication and offers high drug concentrations to specific sites. Studies have been conducted that show the use of 3D printing to develop indwelling bladder devices with an elastic polymer bio-ink that delivers lidocaine hydrochloride directly to the site of action. This method offers treatments for severe bladder diseases.
3D printing pursuable vascular constructs have also been explored. Researchers used 3D bioprinting to deposit perfusable vascular structures with a specifically designed cell-responsive bio-ink that consisted of GelMA, sodium alginate, and poly(ethylene glycol)-tetra-acrylate (PEGTA).[22] Results showed that this blend of bio-ink supported the spreading and proliferation of encapsulated endothelial and stem cells, leading to the formation of perfusable vessels. Large-scale production of these may lead to the application of vascularized tissue constructs in organ transplantation and repairs. Using this method of bioprinting vascular structures may lead to cancer, arterial disease, heart disease, and arthritis treatments by regulating vascularization and angiogenesis.
Incorporating 4D bioprinting
Bioprinting has many applications and routes that can be explored in future studies. One newer study that would allow for more versatile drug delivery is adding a fourth dimension to the 3D printed devices. The current 3D devices are static and inanimate. Adding a fourth dimension allows printed objects to change their shapes and functions when external factors are applied, broadening the range of biomedical applications as cellular self-organization becomes a possibility.[23] This technique allows for more advanced control of drug release. While 4D bioprinting has not yet been fully developed, studies have shown results in the use of responsive materials and bio-inks. Responsive materials contain inherent properties that allow them to reshape in response to stimuli, such as transforming via self-folding, assembling, and disassembling. bio-inks have been seen to undergo maturation with cellular coating, self-organization, and matrix deposition.
Current applications of 4D bioprinting
A few examples of this can be seen in a study that resulted in a self-folding hydrogel in vitro model for ductal carcinoma.[24] The goal of this study was to create a self-folding approach to curved hydrogel microstructures to mimic the geometry of ducts and acini within mammary glands. The researchers used microstructures composed of poly(ethylene glycol)-diacrylate (PEGDA) and copolymerized the PEGDA to synthesize microstructures with increased cell adherence. Curved and tubular structures were fabricated via bioprinting, and the proliferation of cells on the outer surface, along with encapsulation of cells on the inner surface, was observed.
4D printing with thermally actuating hydrogels has also been studied and has been reported to be relatively fast and reversible with skeletal muscle-like linear actuation in 3D bio-printed tough hydrogel materials that control the flow of water.[25] Other examples of 4D bioprinting that have been studied include the usage of water absorption[26] and thermal shape memory[27] to demonstrate the shape change of bioprinted materials. Recently, a 4D-printed capsule system that could release drugs on-demand at specific locations with a core-shell hydrogel was developed by Gupta et al.,[28] and a thermo-responsive poly(propylene fumarate) (PPF)-based system that released drugs in a controlled manner for treating the gastrointestinal tract[29] were developed. These products, along with several others that are currently being studied and produced, show direct and localized delivery of drugs in a controlled and flexible manner.
References
- ↑ 1.0 1.1 Abasalizadeh, Moghaddam (2020). "Alginate-based hydrogels as drug delivery vehicles in cancer treatment and their applications in wound dressing and 3D bioprinting.". Journal of Biological Engineering 14: 8. doi:10.1186/s13036-020-0227-7. PMID 32190110.
- ↑ 2.0 2.1 Li, J (2016). "Recent advances in bioprinting techniques: approaches, applications, and prospects". Journal of Translational Medicine 14: 271. doi:10.1186/s12967-016-1028-0. PMID 27645770.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 Lee, J.M.; Sing, S.L.; Zhou, M; Yeong, W.Y. (2018). "3D bioprinting processes: A perspective on classification and terminology". International Journal of Bioprinting 4 (2): 151. doi:10.18063/ijb.v4i2.151. PMID 33102923.
- ↑ Faramarzi, N (2018). "Patient-specific bioinks for 3D bioprinting of tissue engineering scaffolds". Adv Healthc Mater 7 (11): e1701347. doi:10.1002/adhm.201701347. PMID 29663706.
- ↑ Vigata, M; Hutmacher, D; Bock, N (2020). "Hydrogels as drug delivery systems: A review of current characterization and evaluation techniques.". Pharmaceutics 12 (12): 1188. doi:10.3390/pharmaceutics12121188. PMID 33297493.
- ↑ 6.0 6.1 Das, A.K.; Gavel, P.K. (2020). Soft Matter. 16. pp. 10065–10095. doi:10.1039/D0SM01136C. PMID 33073836. https://pubs.rsc.org/en/content/articlelanding/2020/sm/d0sm01136c. Retrieved 8 April 2023.
- ↑ 7.0 7.1 Yang, X; Wang, Y; Qi, W; Xing, R; Yang, X; Xing, Q; Su, R; He, S (2019). "J. Mater. Chem. B". Journal of Materials Chemistry B 7 (18): 2981–2988. doi:10.1039/C8TB03121E. https://pubs.rsc.org/en/content/articlelanding/2019/tb/c8tb03121e. Retrieved 10 April 2023.
- ↑ Gao, Y; Zhang, C; Chang, J; Yang, C; Liu, J; Fan, S; Ren, C (2019). "Biomater. Sci.". Biomaterials Science 7 (4): 1477–1485. doi:10.1039/C8BM01422A. PMID 30672520. https://pubs.rsc.org/en/content/articlelanding/2019/bm/c8bm01422a. Retrieved 8 April 2023.
- ↑ Piras, C; Fernández-Prieto, S; Borggraeve, W.M.D (2017). "Nanocellulosic materials as bio-inks for 3D bioprinting". Biomaterials Science 5 (10): 1988–1992. doi:10.1039/C7BM00510E. PMID 28829453. https://pubs.rsc.org/en/content/articlelanding/2017/bm/c7bm00510e/unauth#!divCitation. Retrieved 9 April 2023.
- ↑ Rees, A; Powell, L.C.; Chinga-Carrasco, G; Gethin, D.T.; Syverud, K; Hill, K.E.; Thomas, D.W. (2015). "BioMed Res. Int". BioMed Research International 2015: e925757. doi:10.1155/2015/925757. PMID 26090461.
- ↑ Kempin, W; Franz, C; Koster, L.C.; Schneider, F; Bogdahn, M; Weitschies, W; Seidlitz, A (2017). "Eur. J. Pharm Biopharm". European Journal of Pharmaceutics and Biopharmaceutics 115: 84–93. doi:10.1016/j.ejpb.2017.02.014. PMID 28232106. https://pubmed.ncbi.nlm.nih.gov/28232106/. Retrieved 9 April 2023.
- ↑ 12.0 12.1 12.2 12.3 Hong, H; Seo, Y.B.; Kim, D.Y.; Lee, J.S.; Lee, Y.J; Lee, H; Ajiteru, O; Sultan, M.T. et al. (2020). "igital light processing 3D printed silk fibroin hydrogel for cartilage tissue engineering". Biomaterials 232: 119679. doi:10.1016/j.biomaterials.2019.119679. PMID 31865191. https://pubmed.ncbi.nlm.nih.gov/31865191/. Retrieved 9 April 2023.
- ↑ 13.0 13.1 13.2 13.3 Cao, Y; Lee, B.H.; Irvine, S.A.; Wong, Y.S.; Bianco, H; Venkatraman, S (2020). "Inclusion of Cross-Linked Elastin in Gelatin/PEG Hydrogels Favourably Influences Fibroblast Phenotype.". Polymers 12 (3): 670. doi:10.3390/polym12030670. PMID 32192137.
- ↑ 14.0 14.1 14.2 14.3 Lee, H.J.; Kim, Y.B.; Ahn, S.H.; Lee, J.S.; Jang, C.H.; Yoon, H; Chun, W; Kim, G.H. (2015). "New Approach for Fabricating Collagen/ECM-Based Bioinks Using Preosteoblasts and Human Adipose Stem Cells". Advanced Healthcare Materials 4 (9): 1359–1368. doi:10.1002/adhm.201500193. PMID 25874573. https://onlinelibrary.wiley.com/doi/10.1002/adhm.201500193. Retrieved 9 April 2023.
- ↑ 15.0 15.1 15.2 Hong, N (2018). "3D bioprinting and its in vivo applications.". J Biomed Mater Res B 106 (1): 444–459. doi:10.1002/jbm.b.33826. PMID 28106947.
- ↑ 16.0 16.1 16.2 16.3 Mirzaei, M; Okoro, O.V.; Nie, L; Petri, D.F.S.; Shavandi, A (2021). "Protein-based 3D bio fabrication of Biomaterials". Bioengineering 8 (4): 48. doi:10.3390/bioengineering8040048. PMID 33923425.
- ↑ 17.0 17.1 17.2 Bertassoni, L.E.; Cardoso, J.C.; Manoharon, V; Cristino, A.L.; Bhise, N.S.; Araujo, W; Zorlutuna, P; Vrana, N.E. et al. (2014). "Direct-write bioprinting of cell-laden methacrylated gelatin hydrogels". Biofabrication 6 (2): 024105. doi:10.1088/1758-5082/6/2/024105. PMID 24695367. PMC 4040163. Bibcode: 2014BioFa...6b4105B. https://dx.doi.org/10.1088/1758-5082/6/2/024105. Retrieved 10 April 2023.
- ↑ Du, H; Taylor, H.S. (2010). "Stem Cells and reproduction". Current Opinion in Obstetrics & Gynecology 22 (3): 235–241. doi:10.1097/GCO.0b013e328338c152. PMID 20305558.
- ↑ Albanna, M; Binder, K.W.; Murphy, S.V.; Kim, J; Qasem, S.A.; Zhao, W; Tan, J; El-Amin, I.B. et al. (2019). "In Situ Bioprinting of Autologous Skin Cells Accelerates Wound Healing of Extensive Excisional Full-Thickness Wounds". Scientific Reports 9 (1): 1856. doi:10.1038/s41598-018-38366-w. PMID 30755653. Bibcode: 2019NatSR...9.1856A.
- ↑ Triacca, A; Pitzanti, G; Matthew, E; Conti, B; Dorati, R; Lamprou, D.A. (2022). "Stereolithography 3D printed implants: A preliminary investigation as potential local drug delivery systems to the ear". International Journal of Pharmaceutics 616: 121529. doi:10.1016/j.ijpharm.2022.121529. PMID 35114311.
- ↑ Goyanes, A; Xu, X; Trenfield, S.J.; Diaz-Gomez, L; Alvarez-Lorenzo, C; Gaisford, S; Basit, A.W. (2021). "Stereolithography (SLA) 3D printing of a bladder device for intravesical drug delivery". Materials Science & Engineering. C, Materials for Biological Applications 120: 111773. doi:10.1016/j.msec.2020.111773. PMID 33545904.
- ↑ Jia, W; Gungor-Ozkerim, P.S.; Zhang, Y.S.; Yue, K; Zhu, K; Liu, W; Pi, Q; Byambaa, B et al. (2016). "Direct 3D bioprinting of perfusable vascular constructs using a blend bio-ink". Biomaterials 106: 58–68. doi:10.1016/j.biomaterials.2016.07.038. PMID 27552316.
- ↑ Gao, B; Yang, Q; Zhao, X; Jin, G; Ma, Y; Xu, F (2016). "4D bioprinting for biomedical applications". Trends in Biotechnology 34 (9): 746–756. doi:10.1016/j.tibtech.2016.03.004. PMID 27056447. https://www.sciencedirect.com/science/article/pii/S0167779916000664. Retrieved 10 April 2023.
- ↑ Kwag, Hye Rin; Serbo, Janna; Korangath, Preethi; Sukumar, Saraswati; Romer, Lewis; Gracias, David (2016). "A Self-Folding Hydrogel In Vitro Model for Ductal Carcinoma". Tissue Engineering. Part C, Methods 22 (4): 398–407. doi:10.1089/ten.TEC.2015.0442. PMID 26831041.
- ↑ Bakarich, SE; Gorkin, R; Panhuis, M; Spinks, GM (2015). "4D printing with mechanically robust, thermally actuating hydrogels". Macromol Rapid Commun 36 (12): 1211–1217. doi:10.1002/marc.201500079. PMID 25864515.
- ↑ Tibbits, S (2014). "4D Printing: Multi-Material Shape Change". Archit. Design 84: 116–121. doi:10.1002/ad.1710. https://onlinelibrary.wiley.com/doi/abs/10.1002/ad.1710. Retrieved 10 April 2023.
- ↑ Ge, Q; Qi, H.J.; Dunn, M.L. (2013). "ctive materials by four-dimension printing". Applied Physics Letters 103 (13): 131901. doi:10.1063/1.4819837. Bibcode: 2013ApPhL.103m1901G. https://aip.scitation.org/doi/10.1063/1.4819837.
- ↑ Gupta, M. K.; Meng, F.; Johnson, B. N.; Kong, Y. L.; Tian, L.; Yeh, Y. W.; Masters, N.; Singamaneni, S. et al. (June 4, 2015). "Nano Lett". Nano Letters 15 (8): 5321–5329. doi:10.1021/acs.nanolett.5b01688. PMID 26042472. PMC 4536147. https://doi.org/10.1021/acs.nanolett.5b01688. Retrieved 10 April 2023.
- ↑ Malachowski, K; Breger, J; Kwag, H.R.; Wang, M.O.; Fisher, J.P.; Selaru, F.M.; Gracais, D.H. (2014). "Stimuli-Responsive Theragrippers for Chemomechanical Controlled Release". Angew. Chem. Int. Ed. 53 (31): 8045–8049. doi:10.1002/anie.201311047. PMID 24634136.
 |