Medicine:Myelodysplastic syndrome
| Myelodysplastic syndrome | |
|---|---|
| Other names | Preleukemia, myelodysplasia[1][2] |
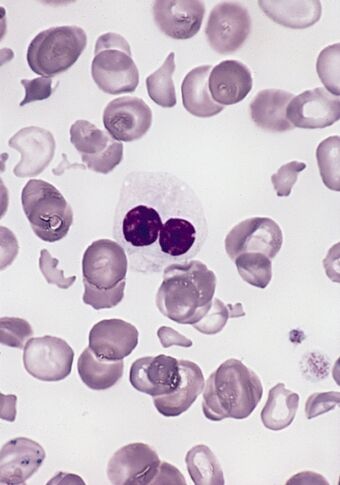 | |
| Blood smear from a person with myelodysplastic syndrome. A hypogranular neutrophil with a pseudo-Pelger-Huet nucleus is shown. There are also abnormally shaped red blood cells, in part related to removal of the spleen. | |
| Specialty | Hematology, oncology |
| Symptoms | None, feeling tired, shortness of breath, easy bleeding, frequent infections[3] |
| Usual onset | ~ 70 years old[4] |
| Risk factors | Previous chemotherapy, radiation therapy, certain chemicals such as tobacco smoke, pesticides, and benzene, exposure to mercury or lead[3] |
| Diagnostic method | Blood test, bone marrow biopsy[3] |
| Treatment | Supportive care, medications, stem cell transplantation[3] |
| Medication | Lenalidomide, antithymocyte globulin, azacitidine[3] |
| Prognosis | Typical survival time 2.5 years[3] |
A myelodysplastic syndrome (MDS) is one of a group of cancers in which immature blood cells in the bone marrow do not mature, and as a result, do not develop into healthy blood cells.[3] Early on, no symptoms typically are seen.[3] Later, symptoms may include fatigue, shortness of breath, bleeding disorders, anemia, or frequent infections.[3] Some types may develop into acute myeloid leukemia.[3]
Risk factors include previous chemotherapy or radiation therapy, exposure to certain chemicals such as tobacco smoke, pesticides, and benzene, and exposure to heavy metals such as mercury or lead.[3] Problems with blood cell formation result in some combination of low red blood cell, platelet, and white blood cell counts.[3] Some types of MDS cause an increase in the production of immature blood cells (called blasts), in the bone marrow or blood.[3] The different types of MDS are identified based on the specific characteristics of the changes in the blood cells and bone marrow.[3]
Treatments may include supportive care, drug therapy, and hematopoietic stem cell transplantation.[3] Supportive care may include blood transfusions, medications to increase the making of red blood cells, and antibiotics.[3] Drug therapy may include the medications lenalidomide, antithymocyte globulin, and azacitidine.[3] Some people can be cured by chemotherapy followed by a stem-cell transplant from a donor.[3]
About seven per 100,000 people are affected by MDS; about four per 100,000 people newly acquire the condition each year.[4] The typical age of onset is 70 years.[4] The prognosis depends on the type of cells affected, the number of blasts in the bone marrow or blood, and the changes present in the chromosomes of the affected cells.[3] The average survival time following diagnosis is 2.5 years.[4] MDS was first recognized in the early 1900s;[5] it came to be called myelodysplastic syndrome in 1976.[5]
Signs and symptoms
Signs and symptoms are nonspecific and generally related to the blood cytopenias:
- Anemia (low RBC count or reduced hemoglobin) – chronic tiredness, shortness of breath, chilled sensation, sometimes chest pain[6]
- Neutropenia (low neutrophil count) – increased susceptibility to infection[7]
- Thrombocytopenia (low platelet count) – increased susceptibility to bleeding and ecchymosis (bruising), as well as subcutaneous hemorrhaging resulting in purpura or petechiae[8][9]
Many individuals are asymptomatic, and blood cytopenia or other problems are identified as a part of a routine blood count:[10]
- Neutropenia, anemia, and thrombocytopenia
- Splenomegaly or rarely hepatomegaly
- Abnormal granules in cells, abnormal nuclear shape and size
- Chromosome abnormality, including chromosomal translocations and abnormal chromosome number
Although some risk exists for developing acute myelogenous leukemia, about 50% of deaths occur as a result of bleeding or infection. However, leukemia that occurs as a result of myelodysplasia is notoriously resistant to treatment. Anemia dominates the early course. Most symptomatic patients complain of the gradual onset of fatigue and weakness, dyspnea, and pallor, but at least half the patients are asymptomatic and their MDS is discovered only incidentally on routine blood counts. Previous chemotherapy or radiation exposure is an important factor in the person's medical history. Fever, weight loss and splenomegaly should point to a myelodysplastic/myeloproliferative neoplasm (MDS/MPN) rather than pure myelodysplastic process.[11]
Cause
Some people have a history of exposure to chemotherapy (especially alkylating agents such as melphalan, cyclophosphamide, busulfan, and chlorambucil) or radiation (therapeutic or accidental), or both (e.g., at the time of stem cell transplantation for another disease). Workers in some industries with heavy exposure to hydrocarbons such as the petroleum industry have a slightly higher risk of contracting the disease than the general population. Xylene and benzene exposures have been associated with myelodysplasia. Vietnam veterans exposed to Agent Orange are at risk of developing MDS. A link may exist between the development of MDS "in atomic-bomb survivors 40 to 60 years after radiation exposure" (in this case, referring to people who were in close proximity to the dropping of the atomic bombs in Hiroshima and Nagasaki during World War II).[12] Children with Down syndrome are susceptible to MDS, and a family history may indicate a hereditary form of sideroblastic anemia or Fanconi anemia.[13]
Pathophysiology
MDS most often develops without an identifiable cause. Risk factors include exposure to an agent known to cause DNA damage, such as radiation, benzene, and certain chemotherapies; other risk factors have been inconsistently reported. Proving a connection between a suspected exposure and the development of MDS can be difficult, but the presence of genetic abnormalities may provide some supportive information. Secondary MDS can occur as a late toxicity of cancer therapy (therapy associated MDS, t-MDS). MDS after exposure to radiation or alkylating agents such as busulfan, nitrosourea, or procarbazine, typically occurs 3–7 years after exposure and frequently demonstrates loss of chromosome 5 or 7. MDS after exposure to DNA topoisomerase II inhibitors occurs after a shorter latency of only 1–3 years and can have a 11q23 translocation. Other pre-existing bone-marrow disorders such as acquired aplastic anemia following immunosuppressive treatment and Fanconi anemia can evolve into MDS.[13]
MDS is thought to arise from mutations in the multipotent bone-marrow stem cell, but the specific defects responsible for these diseases remain poorly understood. Differentiation of blood precursor cells is impaired, and a significant increase in levels of apoptotic cell death occurs in bone-marrow cells. Clonal expansion of the abnormal cells results in the production of cells that have lost the ability to differentiate. If the overall percentage of bone-marrow myeloblasts rises over a particular cutoff (20% for WHO and 30% for FAB), then transformation to acute myelogenous leukemia (AML) is said to have occurred. The progression of MDS to AML is a good example of the multistep theory of carcinogenesis in which a series of mutations occurs in an initially normal cell and transforms it into a cancer cell.[14]
Although recognition of leukemic transformation was historically important (see History), a significant proportion of the morbidity and mortality attributable to MDS results not from transformation to AML, but rather from the cytopenias seen in all MDS patients. While anemia is the most common cytopenia in MDS patients, given the ready availability of blood transfusion, MDS patients rarely experience injury from severe anemia. The two most serious complications in MDS patients resulting from their cytopenias are bleeding (due to lack of platelets) or infection (due to lack of white blood cells). Long-term transfusion of packed red blood cells leads to iron overload.[15]
Genetics
The recognition of epigenetic changes in DNA structure in MDS has explained the success of two (namely the hypomethylating agents 5-azacytidine and decitabine) of three (the third is lenalidomide) commercially available medications approved by the U.S. Food and Drug Administration to treat MDS. Proper DNA methylation is critical in the regulation of proliferation genes, and the loss of DNA methylation control can lead to uncontrolled cell growth and cytopenias. The recently approved DNA methyltransferase inhibitors take advantage of this mechanism by creating a more orderly DNA methylation profile in the hematopoietic stem cell nucleus, thereby restoring normal blood counts and retarding the progression of MDS to acute leukemia.[16]
Some authors have proposed that the loss of mitochondrial function over time leads to the accumulation of DNA mutations in hematopoietic stem cells, and this accounts for the increased incidence of MDS in older patients. Researchers point to the accumulation of mitochondrial iron deposits in the ringed sideroblast as evidence of mitochondrial dysfunction in MDS.[17]
DNA damage
Hematopoietic stem cell aging is thought to be associated with the accrual of multiple genetic and epigenetic aberrations leading to the suggestion that MDS is, in part, related to an inability to adequately cope with DNA damage.[18] An emerging perspective is that the underlying mechanism of MDS could be a defect in one or more pathways that are involved in repairing damaged DNA.[19] In MDS an increased frequency of chromosomal breaks indicates defects in DNA repair processes.[20] Also elevated levels of 8-oxoguanine were found in the DNA of a significant proportion of MDS patients, indicating that the base excision repair pathway that is involved in handling oxidative DNA damages may be defective in these cases.[20]
5q- syndrome
Since at least 1974, the deletion in the long arm of chromosome 5 has been known to be associated with dysplastic abnormalities of hematopoietic stem cells.[21][22] By 2005, lenalidomide, a chemotherapy drug, was recognized to be effective in MDS patients with the 5q- syndrome,[23] and in December 2005, the US FDA approved the drug for this indication. Patients with isolated 5q-, low IPSS risk, and transfusion dependence respond best to lenalidomide. Typically, prognosis for these patients is favorable, with a 63-month median survival. Lenalidomide has dual action, by lowering the malignant clone number in patients with 5q-, and by inducing better differentiation of healthy erythroid cells, as seen in patients without 5q deletion.[citation needed]
Splicing factor mutations
Mutations in splicing factors have been found in 40–80% of cases with myelodysplastic syndrome, particularly in those with ringed sideroblasts.[24]
IDH1 and IDH2 mutations
Mutations in the genes encoding for isocitrate dehydrogenase 1 and 2 (IDH1 and IDH2) occur in 10–20% of patients with myelodysplastic syndrome,[25] and confer a worsened prognosis in low-risk MDS.[26] Because the incidence of IDH1/2 mutations increases as the disease malignancy increases, these findings together suggest that IDH1/2 mutations are important drivers of progression of MDS to a more malignant disease state.[26]
GATA2 deficiency
GATA2 deficiency is a group of disorders caused by a defect, familial, or sporadic inactivating mutations, in one of the two GATA2 genes. These autosomal dominant mutations cause a reduction in the cellular levels of the gene's product, GATA2. The GATA2 protein is a transcription factor critical for the embryonic development, maintenance, and functionality of blood-forming, lymph-forming, and other tissue-forming stem cells. In consequence of these mutations, cellular levels of GATA2 are low and individuals develop over time hematological, immunological, lymphatic, or other presentations. Prominent among these presentations is MDS that often progresses to acute myelocytic leukemia, or less commonly, chronic myelomonocytic leukemia.[27][28]
Transient myeloproliferative disease
Transient myeloproliferative disease is the abnormal proliferation of a clone of noncancerous megakaryoblasts in the liver and bone marrow. The disease is restricted to individuals with Down syndrome or genetic changes similar to those in Down syndrome, develops during pregnancy or shortly after birth, and resolves within 3 months, or in about 10% of cases, progresses to acute megakaryoblastic leukemia.[29][27][30]
Diagnosis
The elimination of other causes of cytopenias, along with a dysplastic bone marrow, is required to diagnose a myelodysplastic syndrome, so differentiating MDS from anemia, thrombocytopenia, and leukopenia is important.[citation needed]
A typical diagnostic investigation includes:
- Full blood count and examination of blood film: The blood film morphology can provide clues about hemolytic anemia, clumping of the platelets leading to spurious thrombocytopenia, or leukemia.
- Blood tests to eliminate other common causes of cytopenias such as lupus, hepatitis, B12, folate, or other vitamin deficiencies, kidney failure or heart failure, HIV, hemolytic anemia, monoclonal gammopathy: Age-appropriate cancer screening should be considered for all anemic patients.
- Bone marrow examination by a hematopathologist: This is required to establish the diagnosis since all hematopathologists consider dysplastic marrow the key feature of myelodysplasia.[31]
- Cytogenetics or chromosomal studies: This is ideally performed on the bone marrow aspirate. Conventional cytogenetics require a fresh specimen since live cells are induced to enter metaphase to allow chromosomes to be seen.
- Interphase fluorescence in situ hybridization testing, usually ordered together with conventional cytogenetic testing, offers rapid detection of several chromosome abnormalities associated with MDS, including del 5q, −7, +8, and del 20q.
- Virtual karyotyping can be done for MDS,[32] which uses computational tools to construct the karyogram from disrupted DNA. Virtual karyotyping does not require cell culture and has a dramatically higher resolution than conventional cytogenetics, but cannot detect balanced translocations.
- Flow cytometry is helpful to identify blasts, abnormal myeloid maturation, and establish the presence of any lymphoproliferative disorder in the marrow.
- Testing for copper deficiency should not be overlooked, as it can morphologically resemble MDS in bone-marrow biopsies.[33]
The features generally used to define an MDS are blood cytopenias, ineffective hematopoiesis, dyserythropoiesis, dysgranulopoiesis, dysmegakaropoiesis, and increased myeloblasts.
Dysplasia can affect all three lineages seen in the bone marrow. The best way to diagnose dysplasia is by morphology and special stains (PAS) used on the bone marrow aspirate and peripheral blood smear. Dysplasia in the myeloid series is defined by:
- Granulocytic series:
- Hypersegmented neutrophils (also seen in vit B12/folate deficiency)
- Hyposegmented neutrophils (pseudo Pelger-Huet)
- Hypogranular neutrophils or pseudo Chediak-Higashi (large azurophilic granules)
- Auer rods – automatically RAEB II (if blast count < 5% in the peripheral blood and < 10% in the bone marrow aspirate); also note Auer rods may be seen in mature neutrophils in AML with translocation t(8;21)
- Dimorphic granules (basophilic and eosinophilic granules) within eosinophils
- Erythroid series:
- Binucleated erythroid precursors and karyorrhexis
- Erythroid nuclear budding
- Erythroid nuclear strings or internuclear bridging (also seen in congenital dyserythropoietic anemias)
- Loss of e-cadherin in normoblasts is a sign of aberrancy.
- PAS (globular in vacuoles or diffuse cytoplasmic staining) within erythroid precursors in the bone marrow aspirate (has no bearing on paraffin-fixed bone-marrow biopsy). Note: one can see PAS vacuolar positivity in L1 and L2 blasts (FAB classification; the L1 and L2 nomenclature is not used in the WHO classification)
- Ringed sideroblasts (10 or more iron granules encircling one-third or more of the nucleus) seen on Perls' Prussian blue iron stain (>15% ringed sideroblasts when counted among red cell precursors for refractory anemia with ring sideroblasts)
- Megakaryocytic series (can be the most subjective):
- Hyposegmented nuclear features in platelet producing megakaryocytes (lack of lobation)
- Hypersegmented (osteoclastic appearing) megakaryocytes
- Ballooning of the platelets (seen with interference contrast microscopy)
Other stains can help in special cases (PAS and naphthol ASD chloroacetate esterase positivity) in eosinophils is a marker of abnormality seen in chronic eosinophilic leukemia and is a sign of aberrancy.
On the bone-marrow biopsy, high-grade dysplasia (RAEB-I and RAEB-II) may show atypical localization of immature precursors, which are islands of immature precursors cells (myeloblasts and promyelocytes) localized to the center of the intertrabecular space rather than adjacent to the trabeculae or surrounding arterioles. This morphology can be difficult to differentiate from treated leukemia and recovering immature normal marrow elements. Also, topographic alteration of the nucleated erythroid cells can be seen in early myelodysplasia (RA and RARS), where normoblasts are seen next to bony trabeculae instead of forming normal interstitially placed erythroid islands.[citation needed]
Differential diagnosis
Myelodysplasia is a diagnosis of exclusion and must be made after proper determination of iron stores, vitamin deficiencies, and nutrient deficiencies are ruled out. Also, congenital diseases such as congenital dyserythropoietic anemia (CDA I through IV) have been recognized, Pearson's syndrome (sideroblastic anemia), Jordans anomaly – vacuolization in all cell lines may be seen in Chanarin-Dorfman syndrome, aminolevulinic acid enzyme deficiency and other more esoteric enzyme deficiencies are known to give a pseudomyelodysplastic picture in one of the cell lines; however, all three cell lines are never morphologically dysplastic in these entities with the exception of chloramphenicol, arsenic toxicity, and other poisons.[citation needed]
All of these conditions are characterized by abnormalities in the production of one or more of the cellular components of blood (red cells, white cells other than lymphocytes, and platelets or their progenitor cells, megakaryocytes).[citation needed]
Classification
World Health Organization
In the late 1990s, a group of pathologists and clinicians working under the World Health Organization (WHO) modified this classification, introducing several new disease categories and eliminating others. In 2008, the WHO developed a new classification scheme that is based more on genetic findings, but morphology of the cells in the peripheral blood, bone marrow aspirate, and bone marrow biopsy are still the screening tests used to decide which classification is best and which cytogenetic aberrations may be related.[citation needed]
The list of dysplastic syndromes under the new WHO system includes:
| Myelodysplastic syndrome | Description |
|---|---|
| Refractory cytopenia with unilineage dysplasia | Refractory anemia, Refractory neutropenia, and Refractory thrombocytopenia. |
| Refractory anemia with ringed sideroblasts (RARS) | Thrombocytosis (RARS-t) (provisional entity) which is in essence a myelodysplastic/myeloproliferative disorder and usually has a JAK2 mutation (janus kinase) – New WHO classification 2008 |
| Refractory cytopenia with multilineage dysplasia (RCMD) | Includes the subset Refractory cytopenia with multilineage dysplasia and ring sideroblasts (RCMD-RS). RCMD includes patients with pathological changes not restricted to red cells (i.e., prominent white cell precursor and platelet precursor (megakaryocyte) dysplasia. |
| Refractory anemia with excess blasts I and II | RAEB was divided into RAEB-I (5–9% blasts) and RAEB-II (10–19%) blasts, which has a poorer prognosis than RAEB-I. Auer rods may be seen in RAEB-II which may be difficult to distinguish from acute myeloid leukemia. |
| 5q- syndrome | Typically seen in older women with normal or high platelet counts and isolated deletions of the long arm of chromosome 5 in bone marrow cells, was added to the classification. |
| Myelodysplasia unclassifiable | Seen in those cases of megakaryocyte dysplasia with fibrosis and others. |
| Refractory cytopenia of childhood (dysplasia in childhood) | – |
Note : not all physicians concur with this reclassification, because the underlying pathology of this disease is not well understood.
Myelodysplastic syndrome unclassified
The WHO has proposed a criterion for diagnosis and classification of MDS that may apply to most cases. However, occasional cases are difficult to classify into defined categories because of one or more unusual features:[citation needed]
- Rare cases with less than 5% blast will present with Auer rods. These cases usually have the features of RAMD.
- Occasionally, cases of MDS present with isolated neutropenia or thrombocytopenia without anemia and with dysplastic changes confined to the single lineage. The terms refractory neutropenia and refractory thrombocytopenia have sometimes been used to describe these cases. A diagnosis of MDS in patients with neutropenia or thrombocytopenia without anemia should be made with caution.
- Patients with RA or RAEB occasionally present with leukocytosis or thrombocytosis instead of the usual cytopenia.
Management
The goals of therapy are to control symptoms, improve quality of life, improve overall survival, and decrease progression to AML.
The IPSS scoring[34] system can help triage patients for more aggressive treatment (i.e. bone marrow transplant) as well as help determine the best timing of this therapy.[35] Supportive care with blood products and hematopoietic growth factors (e.g. erythropoietin) is the mainstay of therapy. The regulatory environment for the use of erythropoietins is evolving, according to a recent US Medicare National coverage determination. However, no comment on the use of hematopoietic growth factors for MDS was made in that document.[36]
Agents have been approved by the U.S. Food and Drug Administration (FDA) for the treatment of MDS:
- 5-azacytidine: 21-month median survival[37][38][39][40]
- Decitabine: Complete response rate reported as high as 43%. A phase I study has shown efficacy in AML when decitabine is combined with valproic acid.[41][42][43][44]
- Lenalidomide: Effective in reducing red blood cell transfusion requirement in patients with the chromosome 5q deletion subtype of MDS[45]
- Decitabine/cedazuridine (Inqovi) is a fixed-dosed combination medication for the treatment of adults with myelodysplastic syndromes (MDS) and chronic myelomonocytic leukemia (CMML).[46]
Chemotherapy with the hypomethylating agents 5-azacytidine and decitabine has been shown to decrease blood transfusion requirements and to retard the progression of MDS to AML. Lenalidomide was approved by the FDA in December 2005 only for use in the 5q- syndrome. In the United States, treatment of MDS with lenalidomide costs about $9,200 per month.[47] The chemotherapy may be supported by other drugs like all-trans retinoic acid (ATRA), however the evidence of benefit is not clear.[48]
HLA-matched allogeneic stem cell transplantation, particularly in younger (i.e. less than 40 years of age) and more severely affected patients, offers the potential for curative therapy. The success of bone marrow transplantation has been found to correlate with severity of MDS as determined by the IPSS score, with patients having a more favorable IPSS score tend to have a more favorable outcome with transplantation.[49]
Iron levels
Iron overload can develop in MDS as a result of the RBC transfusions, which are a major part of the supportive care for anemic MDS patients. Although the specific therapies patients receive may alleviate the RBC transfusion need in some cases, many MDS patients may not respond to these treatments, thus may develop secondary hemochromatosis due to iron overload from repeated RBC transfusions. Patients requiring relatively large numbers of RBC transfusions can experience the adverse effect of chronic iron overload on their liver, heart, and endocrine functions.
For patients requiring many RBC transfusions, serum ferritin levels, number of RBC transfusions received, and associated organ dysfunction (heart, liver, and pancreas) should be monitored to determine iron levels. Monitoring serum ferritin may also be useful, aiming to decrease ferritin levels to < 1000 µg/L.Currently, two iron chelators are available in the US, deferoxamine for intravenous use and deferasirox for oral use. These options now provide potentially useful drugs for treating the iron overload problem. A third chelating agent is available in Europe, deferiprone, for oral use, but is not available in the US.[citation needed]
Clinical trials in MDS patients are ongoing, with iron chelating agents to address the question of whether iron chelation alters the natural history of patients with MDS who are transfusion dependent. Reversal of some of the consequences of iron overload in MDS by iron chelation therapy has been shown. Both the MDS Foundation and the National Comprehensive Cancer Network MDS Guidelines Panel have recommended that chelation therapy be considered to decrease iron overload in selected MDS patients. Evidence also suggests a potential value exists to iron chelation in patients who will undergo a stem-cell transplant. Although deferasirox is generally well tolerated (other than episodes of gastrointestinal distress and kidney dysfunction in some patients), recently a safety warning by the FDA and Novartis was added to deferasirox treatment guidelines. Following postmarketing use of deferasirox, rare cases of acute kidney failure or liver failure occurred, some resulting in death. Due to this, patients should be closely monitored on deferasirox therapy prior to the start of therapy and regularly thereafter.[citation needed]
Prognosis
The outlook in MDS is variable, with about 30% of patients progressing to refractory AML. The median survival time varies from years to months, depending on type. Stem-cell transplantation offers possible cure, with survival rates of 50% at 3 years, although older patients do poorly.[50]
Indicators of a good prognosis: Younger age; normal or moderately reduced neutrophil or platelet counts; low blast counts in the bone marrow (< 20%) and no blasts in the blood; no Auer rods; ringed sideroblasts; normal or mixed karyotypes without complex chromosome abnormalities; and in vitro marrow culture with a nonleukemic growth pattern
Indicators of a poor prognosis: Advanced age; severe neutropenia or thrombocytopenia; high blast count in the bone marrow (20–29%) or blasts in the blood; Auer rods; absence of ringed sideroblasts; abnormal localization or immature granulocyte precursors in bone marrow section; completely or mostly abnormal karyotypes, or complex marrow chromosome abnormalities and in vitro bone marrow culture with a leukemic growth pattern
Karyotype prognostic factors:
- Good: normal, -Y, del(5q), del(20q)
- Intermediate or variable: +8, other single or double anomalies
- Poor: complex (>3 chromosomal aberrations); chromosome 7 anomalies[51]
The IPSS is the most commonly used tool in MDS to predict long-term outcome.[52]
Cytogenetic abnormalities can be detected by conventional cytogenetics, a FISH panel for MDS, or virtual karyotype.
The best prognosis is seen with RA and RARS, where some nontransplant patients live more than a decade (typical is on the order of three to five years, although long-term remission is possible if a bone-marrow transplant is successful). The worst outlook is with RAEB-T, where the mean life expectancy is less than one year. About one-quarter of patients develop overt leukemia. The others die of complications of low blood count or unrelated diseases. The International Prognostic Scoring System is another tool for determining the prognosis of MDS, published in Blood in 1997.[52] This system takes into account the percentage of blasts in the marrow, cytogenetics, and number of cytopenias.
Genetic markers
Although not yet formally incorporated in the generally accepted classification systems, molecular profiling of myelodysplastic syndrome genomes has increased the understanding of prognostic molecular factors for this disease. For example, in low-risk MDS, IDH1 and IDH2 mutations are associated with significantly worsened survival.[26]
Epidemiology
The exact number of people with MDS is not known because it can go undiagnosed and no tracking of the syndrome is mandated. Some estimates are on the order of 10,000 to 20,000 new cases each year in the United States alone. The number of new cases each year is probably increasing as the average age of the population increases, and some authors propose that the number of new cases in those over 70 may be as high as 15 per 100,000 per year.[53]
The typical age at diagnosis of MDS is between 60 and 75 years; a few people are younger than 50, and diagnoses are rare in children. Males are slightly more commonly affected than females.[citation needed]
History
Since the early 20th century, some people with acute myelogenous leukemia were begun to be recognized to have a preceding period of anemia and abnormal blood cell production. These conditions were lumped together with other diseases under the term "refractory anemia". The first description of "preleukemia" as a specific entity was published in 1953 by Block et al.[54] The early identification, characterization and classification of this disorder were problematical, and the syndrome went by many names until the 1976 FAB classification was published and popularized the term MDS.[citation needed]
French-American-British (FAB) classification
In 1974 and 1975, a group of pathologists from France, the US, and Britain produced the first widely used classification of these diseases. This French-American-British classification was published in 1976,[55] and revised in 1982. It was used by pathologists and clinicians for almost 20 years. Cases were classified into five categories:
| ICD-O | Name | Description |
|---|---|---|
| M9980/3 | Refractory anemia (RA) | characterized by less than 5% primitive blood cells (myeloblasts) in the bone marrow and pathological abnormalities primarily seen in red cell precursors |
| M9982/3 | Refractory anemia with ring sideroblasts (RARS) | also characterized by less than 5% myeloblasts in the bone marrow, but distinguished by the presence of 15% or greater of red cell precursors in the marrow being abnormal iron-stuffed cells called "ringed sideroblasts" |
| M9983/3 | Refractory anemia with excess blasts (RAEB) | characterized by 5–19% myeloblasts in the marrow |
| M9984/3 | Refractory anemia with excess blasts in transformation (RAEB-T) | characterized by 5–19% myeloblasts in the marrow (>20% blasts is defined as acute myeloid leukemia) |
| M9945/3 | Chronic myelomonocytic leukemia (CMML), not to be confused with chronic myelogenous leukemia or CML | characterized by less than 20% myeloblasts in the bone marrow and greater than 1*109/L monocytes (a type of white blood cell) circulating in the peripheral blood. |
(A table comparing these is available from the Cleveland Clinic.[56])
People with MDS
- Michael Brecker, musician[57]
- Laurentino Cortizo, the President of Panama[58]
- Roald Dahl, author
- Nora Ephron
- Joe Farrell, musician
- Pat Hingle, actor[59]
- John Kirby, attorney
- Joe Morgan, baseball player
- Paul Motian, musician[60]
- James W. Nance
- Robin Roberts, newscaster
- Carl Sagan, astrophysicist[61]
- Susan Sontag, author[62]
- Fred Willard
See also
- Chloroma – type of tumor made of leukemic cells
- Clonal hematopoiesis
- Myeloproliferative syndrome
References
- ↑ "Myelodysplasia". https://seer.cancer.gov/seertools/hemelymph/53319917e4b0626b1927628f/.
- ↑ "Myelodysplastic Syndromes". https://rarediseases.org/rare-diseases/myelodysplastic-syndromes/.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 3.16 3.17 3.18 "Myelodysplastic Syndromes Treatment (PDQ®) – Patient Version". NCI. 12 August 2015. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq#section/all.
- ↑ 4.0 4.1 4.2 4.3 "Myelodysplastic syndromes: diagnosis, prognosis, and treatment". Deutsches Ärzteblatt International 110 (46): 783–90. November 2013. doi:10.3238/arztebl.2013.0783. PMID 24300826.
- ↑ 5.0 5.1 Hong, Waun Ki; Holland, James F. (2010) (in en). Holland-Frei Cancer Medicine 8 (8 ed.). PMPH-USA. p. 1544. ISBN 9781607950141. https://books.google.com/books?id=R0FbhLsWHBEC&pg=PA1544.
- ↑ "Anemia: Overview". https://www.lecturio.com/concepts/anemia-overview/.
- ↑ "Neutropenia". https://www.lecturio.com/concepts/neutropenia/.
- ↑ Myelodysplastic Syndrome. The Leukemia & Lymphoma Society. White Plains, NY. 2001. p 24. Retrieved 05-12-2008.
- ↑ "Thrombocytopenia". https://www.lecturio.com/concepts/thrombocytopenia/.
- ↑ "Myelodysplastic Syndromes". https://www.lecturio.com/concepts/myelodysplastic-syndromes/.
- ↑ Patnaik, Mrinal M.; Lasho, Terra (2020-12-04). "Evidence-Based Minireview: Myelodysplastic syndrome/myeloproliferative neoplasm overlap syndromes: a focused review". Hematology 2020 (1): 460–464. doi:10.1182/hematology.2020000163. ISSN 1520-4391. PMID 33275673. PMC 7727594. https://doi.org/10.1182/hematology.2020000163.
- ↑ "Risk of myelodysplastic syndromes in people exposed to ionizing radiation: a retrospective cohort study of Nagasaki atomic bomb survivors". Journal of Clinical Oncology 29 (4): 428–34. February 2011. doi:10.1200/JCO.2010.31.3080. PMID 21149671.
- ↑ 13.0 13.1 Dokal, Inderjeet; Vulliamy, Tom (August 2010). "Inherited bone marrow failure syndromes". Haematologica 95 (8): 1236–1240. doi:10.3324/haematol.2010.025619. ISSN 0390-6078. PMID 20675743.
- ↑ Bejar, Rafael (December 2018). "What biologic factors predict for transformation to AML?". Best Practice & Research. Clinical Haematology 31 (4): 341–345. doi:10.1016/j.beha.2018.10.002. ISSN 1532-1924. PMID 30466744. https://pubmed.ncbi.nlm.nih.gov/30466744/.
- ↑ Rasel, Mohammad; Mahboobi, Sohail K. (2022), "Transfusion Iron Overload", StatPearls (Treasure Island (FL): StatPearls Publishing), PMID 32965817, http://www.ncbi.nlm.nih.gov/books/NBK562146/, retrieved 2022-02-03
- ↑ Wolach, O; Stone, R. M. (2015). "How I treat mixed-phenotype acute leukemia". Blood 125 (16): 2477–85. doi:10.1182/blood-2014-10-551465. PMID 25605373.
- ↑ "Mitochondrial ferritin expression in erythroid cells from patients with sideroblastic anemia". Blood 101 (5): 1996–2000. March 2003. doi:10.1182/blood-2002-07-2006. PMID 12406866.
- ↑ Zhou T, Chen P, Gu J, Bishop AJ, Scott LM, Hasty P, Rebel VI. Potential relationship between inadequate response to DNA damage and development of myelodysplastic syndrome. Int J Mol Sci. 2015 Jan 5;16(1):966-89. doi: 10.3390/ijms16010966. PMID: 25569081; PMCID: PMC4307285
- ↑ Zhou T, Hasty P, Walter CA, Bishop AJ, Scott LM, Rebel VI. Myelodysplastic syndrome: an inability to appropriately respond to damaged DNA? Exp Hematol. 2013 Aug;41(8):665-74. doi: 10.1016/j.exphem.2013.04.008. Epub 2013 Apr 30. PMID: 23643835; PMCID: PMC3729593
- ↑ 20.0 20.1 Jankowska AM, Gondek LP, Szpurka H, Nearman ZP, Tiu RV, Maciejewski JP. Base excision repair dysfunction in a subgroup of patients with myelodysplastic syndrome. Leukemia. 2008 Mar;22(3):551-8. doi: 10.1038/sj.leu.2405055. Epub 2007 Dec 6. PMID: 18059482
- ↑ "5q- and disordered haematopoiesis". Clinics in Haematology 15 (4): 1023–35. November 1986. PMID 3552346.
- ↑ "Distinct haematological disorder with deletion of long arm of no. 5 chromosome". Nature 251 (5474): 437–38. October 1974. doi:10.1038/251437a0. PMID 4421285. Bibcode: 1974Natur.251..437V.
- ↑ "Efficacy of lenalidomide in myelodysplastic syndromes". The New England Journal of Medicine 352 (6): 549–57. February 2005. doi:10.1056/NEJMoa041668. PMID 15703420.
- ↑ Rozovski U, Keating M, Estrov Z (2012) The significance of spliceosome mutations in chronic lymphocytic leukemia. Leuk Lymphoma
- ↑ "The driver and passenger effects of isocitrate dehydrogenase 1 and 2 mutations in oncogenesis and survival prolongation". Biochimica et Biophysica Acta (BBA) - Reviews on Cancer 1846 (2): 326–41. December 2014. doi:10.1016/j.bbcan.2014.05.004. PMID 24880135.
- ↑ 26.0 26.1 26.2 "Clinical and biological implications of ancestral and non-ancestral IDH1 and IDH2 mutations in myeloid neoplasms". Leukemia 29 (11): 2134–42. November 2015. doi:10.1038/leu.2015.91. PMID 25836588.
- ↑ 27.0 27.1 "GATA factor mutations in hematologic disease". Blood 129 (15): 2103–10. April 2017. doi:10.1182/blood-2016-09-687889. PMID 28179280.
- ↑ "Heterogeneity of GATA2-related myeloid neoplasms". International Journal of Hematology 106 (2): 175–82. August 2017. doi:10.1007/s12185-017-2285-2. PMID 28643018.
- ↑ "Transient Abnormal Myelopoiesis and AML in Down Syndrome: an Update". Current Hematologic Malignancy Reports 11 (5): 333–41. October 2016. doi:10.1007/s11899-016-0338-x. PMID 27510823.
- ↑ "Acute leukemias in children with Down syndrome". Molecular Genetics and Metabolism 107 (1–2): 25–30. September 2012. doi:10.1016/j.ymgme.2012.07.011. PMID 22867885.
- ↑ "Rudhiram Hematology Clinic - Google Search". https://www.google.com/search?q=Rudhiram+Hematology+Clinic&ludocid=5150959102409787010&lpsid=CIHM0ogKEICAgIDmpez2cA&lsig=AB86z5XrzWBckbIx8_P21jwulFPG.
- ↑ Gondek LP, Tiu R, O'Keefe CL, Sekeres MA, Theil KS, Maciejewski JP. Chromosomal lesions and uniparental disomy detected by SNP arrays in MDS, MDS/MPD, and MDS-derived AML. Blood. 2008 Feb 1;111(3):1534–42.
- ↑ "Copper deficiency causes reversible myelodysplasia". American Journal of Hematology 82 (7): 625–30. July 2007. doi:10.1002/ajh.20864. PMID 17236184.
- ↑ "Marrowforums.org: MDS – Myelodysplastic Syndromes". http://www.marrowforums.org/mds.html.
- ↑ "A decision analysis of allogeneic bone marrow transplantation for the myelodysplastic syndromes: delayed transplantation for low-risk myelodysplasia is associated with improved outcome". Blood 104 (2): 579–85. July 2004. doi:10.1182/blood-2004-01-0338. PMID 15039286.
- ↑ "Centers for Medicare & Medicaid Services". http://www.cms.hhs.gov/mcd/viewdecisionmemo.asp?id=203.
- ↑ "Low-dose 5-aza-2'-deoxycytidine, a DNA hypomethylating agent, for the treatment of high-risk myelodysplastic syndrome: a multicenter phase II study in elderly patients". Journal of Clinical Oncology 18 (5): 956–62. March 2000. doi:10.1200/JCO.2000.18.5.956. PMID 10694544.
- ↑ "Cytogenetic responses in high-risk myelodysplastic syndrome following low-dose treatment with the DNA methylation inhibitor 5-aza-2'-deoxycytidine". British Journal of Haematology 114 (2): 349–57. August 2001. doi:10.1046/j.1365-2141.2001.02933.x. PMID 11529854.
- ↑ "Randomized controlled trial of azacitidine in patients with the myelodysplastic syndrome: a study of the cancer and leukemia group B". Journal of Clinical Oncology 20 (10): 2429–40. May 2002. doi:10.1200/JCO.2002.04.117. PMID 12011120.
- ↑ "Further analysis of trials with azacitidine in patients with myelodysplastic syndrome: studies 8421, 8921, and 9221 by the Cancer and Leukemia Group B". Journal of Clinical Oncology 24 (24): 3895–903. August 2006. doi:10.1200/JCO.2005.05.4346. PMID 16921040.
- ↑ "Update of the decitabine experience in higher risk myelodysplastic syndrome and analysis of prognostic factors associated with outcome". Cancer 109 (2): 265–73. January 2007. doi:10.1002/cncr.22376. PMID 17133405.
- ↑ "Decitabine improves patient outcomes in myelodysplastic syndromes: results of a phase III randomized study". Cancer 106 (8): 1794–803. April 2006. doi:10.1002/cncr.21792. PMID 16532500.
- ↑ "Results of a randomized study of 3 schedules of low-dose decitabine in higher-risk myelodysplastic syndrome and chronic myelomonocytic leukemia". Blood 109 (1): 52–57. January 2007. doi:10.1182/blood-2006-05-021162. PMID 16882708.
- ↑ "Phase I study of decitabine alone or in combination with valproic acid in acute myeloid leukemia". Journal of Clinical Oncology 25 (25): 3884–91. September 2007. doi:10.1200/JCO.2006.09.4169. PMID 17679729.
- ↑ "Lenalidomide in the myelodysplastic syndrome with chromosome 5q deletion". The New England Journal of Medicine 355 (14): 1456–65. October 2006. doi:10.1056/NEJMoa061292. PMID 17021321.
- ↑ "FDA Approves New Therapy for Myelodysplastic Syndromes (MDS) That Can Be Taken at Home". U.S. Food and Drug Administration (FDA) (Press release). 7 July 2020. Retrieved 7 July 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "Lenalidomide (Revlimid) for anemia of myelodysplastic syndrome". The Medical Letter on Drugs and Therapeutics 48 (1232): 31–32. April 2006. PMID 16625140.
- ↑ "Effects of all-trans retinoic acid (ATRA) in addition to chemotherapy for adults with acute myeloid leukaemia (AML) (non-acute promyelocytic leukaemia (non-APL))". The Cochrane Database of Systematic Reviews 2018 (8): CD011960. August 2018. doi:10.1002/14651858.CD011960.pub2. PMID 30080246.
- ↑ "The impact of intensive antileukaemic treatment strategies on prognosis of myelodysplastic syndrome patients aged less than 61 years according to International Prognostic Scoring System risk groups". British Journal of Haematology 123 (1): 81–89. October 2003. doi:10.1046/j.1365-2141.2003.04544.x. PMID 14510946.
- ↑ Kasper, Dennis L et al. (2005). Harrison's Principles of Internal Medicine (16th ed.). New York: McGraw-Hill. p. 625. ISBN 978-0-07-139140-5. https://archive.org/details/harrisonsprincip00kasp.
- ↑ "Incidence, characterization and prognostic significance of chromosomal abnormalities in 640 patients with primary myelodysplastic syndromes. Grupo Cooperativo Español de Citogenética Hematológica". British Journal of Haematology 108 (2): 346–56. February 2000. doi:10.1046/j.1365-2141.2000.01868.x. PMID 10691865.
- ↑ 52.0 52.1 "International scoring system for evaluating prognosis in myelodysplastic syndromes". Blood 89 (6): 2079–88. March 1997. doi:10.1182/blood.V89.6.2079. PMID 9058730.
- ↑ "Epidemiological features of myelodysplastic syndromes: results from regional cancer surveys and hospital-based statistics". International Journal of Hematology 73 (4): 405–10. June 2001. doi:10.1007/BF02994001. PMID 11503953.
- ↑ "Preleukemic acute human leukemia". Journal of the American Medical Association 152 (11): 1018–28. July 1953. doi:10.1001/jama.1953.03690110032010. PMID 13052490.
- ↑ "Proposals for the classification of the acute leukaemias. French-American-British (FAB) co-operative group". British Journal of Haematology 33 (4): 451–58. August 1976. doi:10.1111/j.1365-2141.1976.tb03563.x. PMID 188440.
- ↑ "Table 1: French-American-British (FAB) Classification of MDS". http://www.clevelandclinicmeded.com/diseasemanagement/hematology/myelo/table1.htm.
- ↑ "Saxophonist Brecker dies from MDS". 14 January 2007. https://variety.com/2007/music/markets-festivals/saxophonist-brecker-dies-from-mds-1117957370/.
- ↑ "Panama says President Cortizo still in remission from rare blood disorder" (in en). Reuters. 2023-12-06. https://www.reuters.com/world/americas/panama-says-president-cortizo-still-remission-rare-blood-disorder-2023-12-06/.
- ↑ Staff, JournalNow (4 January 2009). "Veteran actor Pat Hingle dies at 84 in NC home". https://www.journalnow.com/news/local/veteran-actor-pat-hingle-dies-at-in-nc-home/article_439f8c0e-92f1-51df-9b0f-2706a36dca84.html.
- ↑ McClellan, Dennis (November 24, 2011). "Paul Motian dies at 80; jazz drummer and composer". Los Angeles Times. https://www.latimes.com/local/obituaries/la-me-paul-motian-20111124-story.html.
- ↑ "Remembering Carl Sagan". 9 November 2012. http://www.universetoday.com/98390/remembering-carl-sagan-2/.
- ↑ "Illness as More Than Metaphor". 4 December 2005. https://www.nytimes.com/2005/12/04/magazine/illness-as-more-than-metaphor.html.
External links
- Myelodysplastic syndrome at Curlie
- Fenaux, P., et al. (2014). Myelodysplastic syndromes: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up † Myelodysplastic syndromes: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Oncology 25(suppl 3): iii57–iii69.
| Classification | |
|---|---|
| External resources |
 |


