Medicine:Seasonal influenza vaccine brands
| Influenza (Flu) |
|---|
 |
Seasonal influenza vaccine brands include Fluzone/Fluzone Quadrivalent[1] and Vaxigrip/VaxigripTetra[2], Influvac and Optaflu.
CSL Limited
CSL Limited obtain Novartis' flu vaccine unit in 2014, and transfer to CSL Subsidiary, bioCSL (Seqirus).[3] The following are list of bioCSL flu vaccine brands:[4]
- Afluria Quadrivalent,[1] an inactivated quadrivalent influenza vaccine (split viron). Also marketed as Afluria Quad and Afluria Tetra in various different markets.
- Afluria, an inactivated influenza vaccine (split viron). Also marketed as Enzira, Fluvax and Nilgrip in various different markets.
- Agrippal, an influenza vaccine (surface antigen inactivated). Also marketed as Begripal, Fluazur, Sandovac, Agriflu, Chiroflu in various different markets.
- Fluad,[1] an inactivated influenza vaccine, adjuvanted. Also marketed as Chiromas in Spain.
- Fluad Pediatric, a pediatric inactivated influenza vaccine, adjuvanted
- Fluad Quadrivalent, a quadrivalent influenza vaccine, adjuvanted
- Fluad Tetra, a quadrivalent influenza vaccine (surface antigen, inactivated, adjuvanted)
- Flucelvax Quadrivalent,[1] a quadrivalent inactivated influenza vaccine
- Flucelvax Tetra,[5] a quadrivalent inactivated influenza vaccine
GlaxoSmithKline
Fluarix and Flulaval are flu vaccines brand of GlaxoSmithKline.[6]
Mylan
Influvac is a subunit vaccine produced and marketed by Mylan. It contains inactivated purified surface fragments (subunits) from the three different strains of the influenza virus (A/H1N1, A/H3N2, and Influenza B virus) that are selected and distributed by the World Health Organization, on the basis of their latest recommendations. Previously, it was produced and marketed by Abbott Laboratories[7]
In February 2010, Abbott acquired the vaccines subunit from Solvay Pharmaceuticals included in its $6.2 billion purchase[8] and the subunit influenza vaccine — Influvac has been commercially available on the market since the early nineteen-eighties.[7] With the acquisition of Solvay, Abbott retained access to the Eastern European, Middle Eastern & Latin American markets. Approximately $850 million of sales revenue from vaccines was reported by Solvay Pharmaceuticals in 2009.[8]
In February 2015, Mylan Laboratories completed the deal with Abbott to purchase Abbott's generic drugs business in developed markets, which includes Influvac.[9][10]
Novartis
Optaflu is a cell culture derived influenza vaccine manufactured by Novartis. On April 27, 2007 Novartis received a positive opinion supporting European Union approval of Optaflu. It is the first influenza vaccine made in a mammalian cell line, rather than chicken eggs.[11] The plan was to manufacture the vaccine in Holly Springs, North Carolina. The United States government provided $500 million in construction costs and guaranteed vaccine purchases.[12]
Novartis' flu vaccine unit was sold to CSL Limited in 2014, and was placed under CSL subsidiary, bioCSL (Seqirus).[3] bioCSL as marketing authorization holder decided to discontinue the usage of Optaflu brand in 2017 due to commercial reasons[13]
Sanofi Pasteur
Sanofi Pasteur produces the split-virus influenza vaccines Fluzone (especially in the United States (US)[1] and Vaxigrip/VaxigripTetra (especially in Europe).[2] Sanofi also produces a recombinant influenza vaccine branded Flublok.[14]
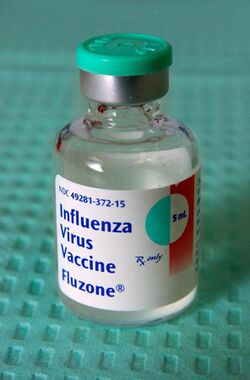
Fluzone
Fluzone is a brand of influenza vaccine distributed by Sanofi Pasteur. It is a split-virus vaccine that is produced by chemical disruption of the influenza virus, making it incapable of causing influenza.[citation needed]
Dosage and storage
Fluzone is typically administered in a single dose by intramuscular injection;[15] an intradermal injection is also available.[16] It is presented as a 0.25 ml syringe for pediatric use, as a 0.5 ml syringe for adults and children, as a 0.5 ml vial for adults and children, and as a 5 ml vial for adults and children.[15] Fluzone must be refrigerated under temperatures from 2 to 8 °C (36 to 46 °F) and is inactivated by freezing. Fluzone was initially approved in 1980 by the FDA.[15]
Adverse effects
The following adverse effects have been reported:[15]
- Mild soreness, local pain and swelling at the local of the injection
- In small children and in people with no previous exposure to a flu vaccine, episodes of fever, malaise, myalgia (muscle pain)
- In people who are sensitive to egg protein, allergic reactions may ensue, such as hives, angioedema, asthma and anaphylaxis
High-dose vaccine
A high-dose vaccine (Fluzone High-Dose) four times the strength of standard flu vaccine was approved by the FDA in 2009.[17][18][19] This vaccine is intended for people 65 and over, who typically have weakened immune response due to normal aging. The vaccine produces a greater immune response than standard vaccine. According to the CDC,[1] "a study published in the New England Journal of Medicine[20] [in August, 2014] indicated that the high-dose vaccine was 24.2% more effective in preventing flu in adults 65 years of age and older relative to a standard-dose vaccine." CDC recommends the high-dose vaccine for people 65 and over but expresses no preference between it and standard vaccine. Further studies were underway (As of 2014).[citation needed]
Vaxigrip/VaxigripTetra
The split-virion influenza vaccines made by Sanofi Pasteur in Europe are called Vaxigrip and VaxigripTetra.[2][21] Vaxigrip provides immune responses to three influenza strains and VaxigripTetra adds another B strain. VaxigripTetra was approved in Europe in 2016 except for infants younger than three years old.[2]
Flublok
Recombinant influenza vaccines are produced using recombinant virus technology. This method does not require an egg-grown vaccine virus and does not use chicken eggs in the production process. Sanofi's recombinant influenza vaccine is called Flublok.[14]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 "Influenza vaccines — United States, 2019–20 influenza season*". U.S. Centers for Disease Control and Prevention (CDC). 22 August 2019. https://www.cdc.gov/flu/professionals/vaccines.htm.
- ↑ 2.0 2.1 2.2 2.3 "Quadrivalent inactivated influenza vaccine (VaxigripTetra)". Expert Review of Vaccines 17 (1): 1–11. 2017-11-27. doi:10.1080/14760584.2018.1407650. PMID 29157068.
- ↑ 3.0 3.1 "Australia's CSL buys Novartis flu vaccine unit for $275 mln". Reuters. 27 October 2014. https://www.reuters.com/article/us-novartis-csl-influenza-idUSKBN0IF0Z720141027.
- ↑ "CSL's product list". Seqirus. https://www.seqirus.com/products.
- ↑ "Flucelvax Tetra". 24 September 2018. https://www.ema.europa.eu/en/medicines/human/EPAR/flucelvax-tetra.
- ↑ "GSK ships 2019-20 seasonal influenza vaccines for US market". 15 July 2019. https://us.gsk.com/en-us/media/press-releases/gsk-ships-2019-20-seasonal-influenza-vaccines-for-us-market/.
- ↑ 7.0 7.1 "Trivalent inactivated subunit influenza vaccine Influvac: 25-Year experience of safety and immunogenicity". Vaccine 27 (18): 2414–7. April 2009. doi:10.1016/j.vaccine.2009.02.008. PMID 19368782.
- ↑ 8.0 8.1 "Abbott Halts Influvac Sale". Drug Discovery & Development. http://www.dddmag.com/news/2010/09/abbott-halts-influvac-sale.
- ↑ Pierson, Ransdell (14 July 2014). "Mylan to buy Abbott generics, cut taxes, in $5.3 billion deal". reuters. https://www.reuters.com/article/us-mylan-abbott-idUSKBN0FJ10F20140714.
- ↑ Tascarella, Patty (27 February 2015). "Mylan inversion deal completed". Pittsburgh Business Times. https://www.bizjournals.com/pittsburgh/news/2015/02/27/mylan-inversion-deal-completed.html.
- ↑ "Optaflu, the Novartis cell culture-derived influenza vaccine, receives positive opinion supporting European Union regulatory approval" (Press release). Novartis. Retrieved 2009-04-29.
- ↑ Pollack, Andrew (April 28, 2009). "Swine Flu Vaccine May Be Months Away, Experts Say". The New York Times. https://www.nytimes.com/2009/04/29/business/economy/29vaccine.html. "But Novartis is building a cell culture flu vaccine factory in Holly Springs, N.C., which might be ready for use in 2010 or 2011. The federal government is providing nearly $500 million in construction costs and guaranteed vaccine purchases."
- ↑ "Optaflu - Expiry of the marketing authorisation in the European Union". European Medicine Agency. 22 June 2017. https://www.ema.europa.eu/en/documents/public-statement/public-statement-optaflu-expiry-marketing-authorisation-european-union_en.pdf.
- ↑ 14.0 14.1 "Recombinant Influenza (Flu) Vaccine". Center for Disease Control and Prevention. https://www.cdc.gov/flu/prevent/qa_flublok-vaccine.htm.
- ↑ 15.0 15.1 15.2 15.3 "Fluzone Prescribing Information" . Sanofi Pasteur. June 2012.
- ↑ "Fluzone intradermal vaccine website". Sanofi Pasteur.
- ↑ "FDA Approves A High Dose Seasonal Influenza Vaccine Specifically Intended for People Ages 65 and Older" (Press release). U.S. Food and Drug Administration (FDA). December 23, 2009. Archived from the original on December 31, 2009.
- ↑ Centers for Disease Control and Prevention (CDC) (April 2010). "Licensure of a high-dose inactivated influenza vaccine for persons aged >or=65 years (Fluzone High-Dose) and guidance for use - United States, 2010". MMWR. Morbidity and Mortality Weekly Report 59 (16): 485–6. PMID 20431524. https://www.cdc.gov/mmwr/PDF/wk/mm5916.pdf.
- ↑ "Fluzone High-Dose Quadrivalent". November 14, 2019. https://www.fda.gov/vaccines-blood-biologics/vaccines/fluzone-high-dose-quadrivalent.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "Efficacy of high-dose versus standard-dose influenza vaccine in older adults". N. Engl. J. Med. 371 (7): 635–45. August 2014. doi:10.1056/NEJMoa1315727. PMID 25119609.
- ↑ "A trivalent, inactivated influenza vaccine (Vaxigrip): summary of almost 50 years of experience and more than 1.8 billion doses distributed in over 120 countries". Expert Rev Vaccines 16 (6): 545–564. June 2017. doi:10.1080/14760584.2017.1324302. PMID 28460594.
External links
- "Types of seasonal influenza vaccine". https://www.ecdc.europa.eu/en/seasonal-influenza/prevention-and-control/vaccines/types-of-seasonal-influenza-vaccine.
- "Different Types of Flu Vaccines". https://www.cdc.gov/flu/prevent/different-flu-vaccines.htm.
- "Upcoming 2019-2020 Influenza Season". https://www.cdc.gov/flu/season/faq-flu-season-2019-2020.htm.
- "Seasonal Flu Shot". https://www.cdc.gov/flu/prevent/flushot.htm.
- "Availability of influenza vaccines by country in EU/EEA in the 2019/20 season". https://www.ecdc.europa.eu/sites/default/files/documents/Seasonal-influenza-vaccines-by-country-Europe-2019-2020.xlsx.