Chemistry:Rimantadine
Rimantadine (INN, sold under the trade name Flumadine[1]) is an orally administered antiviral drug[2] used to treat, and in rare cases prevent, influenzavirus A infection. When taken within one to two days of developing symptoms, rimantadine can shorten the duration and moderate the severity of influenza. Rimantadine can mitigate symptoms, including fever.[3] Both rimantadine and the similar drug amantadine are derivates of adamantane. Rimantadine is found to be more effective than amantadine because when used the patient displays fewer symptoms.[4] Rimantadine was approved by the Food and Drug Administration (FDA) in 1994.
Rimantadine was approved for medical use in 1993.[5] Seasonal H3N2 and 2009 pandemic flu samples tested have shown resistance to rimantadine, and it is no longer recommended to prescribe for treatment of the flu.[6]
Medical uses
Influenza A
Rimantadine inhibits influenza activity by binding to amino acids in the M2 transmembrane channel and blocking proton transport across the M2 channel.[7] Rimantadine is believed to inhibit influenza's viral replication, possibly by preventing the uncoating of the virus's protective shells, which are the envelope and capsid. The M2 channel is known to be responsible for viral replication in the influenza virus. Genetic studies suggest that the virus M2 protein, an ion channel specified by virion M2 gene, plays an important role in the susceptibility of influenza A virus to inhibition by rimantadine. Rimantadine is bound inside the pore to amantadine specific amino acid binding sites with hydrogen binding and van der Waals interactions.[8] The ammonium group (with neighboring water molecules) is positioned towards the C terminus with the amantadane group is positioned towards the N-terminus when bound inside the M2 pore.

Influenza resistance
Resistance to rimantadine can occur as a result of amino acid substitutions at certain locations in the transmembrane region of M2. This prevents binding of the antiviral to the channel.[9]
Rimantadine enantiomers interactions with M2
Rimantadine, when sold as flumadine, is present as a racemic mixture; the R and S states are both present in the drug. Solid state NMR studies have shown that the R enantiomer has a stronger binding affinity to the M2 channel pore than the S-enantiomer of rimantadine.[10] Antiviral assay and electrophysiology studies show that there is no significant difference between the R and S enantiomers in binding affinity to amino acids in the M2 channel.[11] Since the enantiomers have similar binding affinity, they also have similar ability to block the channel pore and work as an effective antiviral.
Parkinson's disease
Rimantadine, like its antiviral cousin amantadine, possesses antiparkinsonian activity and can be used in the treatment of Parkinson's disease.[12][13] However, in general, neither rimantadine nor amantadine is a preferred agent for this therapy and would be reserved for cases of the disease that are less responsive to front-line treatments.
Others
Rimantadine is shown to be effective against other RNA-containing viruses. It can treat arboviruses like Saint Louis encephalitis and Sindbis. Other viruses that can be treated with Rimantadine include respiratory synctial[check spelling] and parainfluenza viruses.[14] Rimantadine has also been shown to treat chronic hepatitis C.[15]
Side effects
Rimantadine can produce gastrointestinal and central nervous system adverse effects. Approximately 6% of patients (compared to 4% of patients taking a placebo) reported side-effects at a dosage of 200 mg/d.[16]
- Nausea
- Upset stomach
- Nervousness
- Tiredness
- Lightheadedness
- Trouble sleeping (insomnia)
- Difficulty concentrating
- Confusion
Rimantadine shows fewer CNS symptoms than its sister drug amantadine.[17]
Interactions
- Taking paracetamol (acetaminophen, Tylenol) or acetylsalicylic acid (aspirin) while taking rimantadine is known to reduce the body's uptake of rimantadine by approximately 12%.[18]
- Taking anticholinergic drugs with amantadine may increase underlying seizure disorders and aggravate congestive heart failure.[19]
Pharmacology
Pharmacodynamics
The related drugs memantine and to a much lesser extent amantadine are known to act as NMDA receptor antagonists.[20][21] The affinity of rimantadine for the NMDA receptor does not seem to have been reported.[22][23][24] Analogues of rimantadine are known to act as NMDA receptor antagonists.[25]
Chemistry
Synthesis
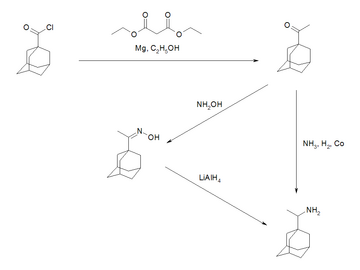
1-carboxyadamatanones are reduced with sodium borohydride to create racemic hydroxy acid. Excess methyllithium is then added to create methyl ketones which when reduced with lithium aluminum hydride gives the amine group.[26]
History
Rimantadine was discovered in 1963[27][28] and patented in 1965 in the US by William W. Prichard in Du Pont & Co., Wilmington, Delaware (patent on new chemical compound U.S. Patent 3,352,912, 1965 and on the first method of synthesis U.S. Patent 3,592,934, 1967).[29][30] Prichard's methods of synthesis of rimantadine from the corresponding ketone oxime were based on its reduction with lithium aluminum hydride.
See also
References
- ↑ "Flumadine (Rimantadine): Side Effects, Uses, Dosage, Interactions, Warnings". https://www.rxlist.com/flumadine-drug.htm.
- ↑ "Neuraminidase inhibitor-rimantadine combinations exert additive and synergistic anti-influenza virus effects in MDCK cells". Antimicrobial Agents and Chemotherapy 48 (12): 4855–63. December 2004. doi:10.1128/AAC.48.12.4855-4863.2004. PMID 15561867.
- ↑ "Rationing of influenza vaccine during a pandemic: ethical analyses". Vaccine 25 (11): 2019–26. March 2007. doi:10.1016/j.vaccine.2006.11.045. PMID 17258359.
- ↑ "Amantadine and rimantadine for influenza A in adults". The Cochrane Database of Systematic Reviews 2006 (2). April 2006. doi:10.1002/14651858.CD001169.pub3. PMID 16625539.
- ↑ (in en) Principles and Practice of Pediatric Infectious Disease. Elsevier Health Sciences. 2012. p. 1502. ISBN 978-1-4377-2702-9. https://books.google.com/books?id=nQ7-o8JAH7kC&pg=PA1502.
- ↑ Antiviral Agents for the Treatment and Chemoprophylaxis of Influenza: Recommendations of the Advisory Committee on Immunization Practices (ACIP)
- ↑ "An effective molecular blocker of ion channel of M2 protein as anti-influenza A drug.". Journal of Biomolecular Structure and Dynamics 39 (7): 2352–2363. April 2020. doi:10.1080/07391102.2020.1747550. ISSN 0739-1102. PMID 32212957.
- ↑ "Affinity of Rimantadine Enantiomers against Influenza A/M2 Protein Revisited". ACS Medicinal Chemistry Letters 8 (2): 145–150. February 2017. doi:10.1021/acsmedchemlett.6b00311. PMID 28217261.
- ↑ "Functional studies indicate amantadine binds to the pore of the influenza A virus M2 proton-selective ion channel". Proceedings of the National Academy of Sciences of the United States of America 105 (31): 10967–72. August 2008. doi:10.1073/pnas.0804958105. PMID 18669647. Bibcode: 2008PNAS..10510967J.
- ↑ "Differential Binding of Rimantadine Enantiomers to Influenza A M2 Proton Channel". Journal of the American Chemical Society 138 (5): 1506–9. February 2016. doi:10.1021/jacs.5b13129. PMID 26804976. Bibcode: 2016JAChS.138.1506W.
- ↑ "Affinity of Rimantadine Enantiomers against Influenza A/M2 Protein Revisited". ACS Medicinal Chemistry Letters 8 (2): 145–150. February 2017. doi:10.1021/acsmedchemlett.6b00311. PMID 28217261.
- ↑ "A pilot study on the motor effects of rimantadine in Parkinson's disease". Clin Neuropharmacol 22 (1): 30–32. 1999. doi:10.1097/00002826-199901000-00006. PMID 10047931.
- ↑ "Rimantadine in Parkinson's disease patients experiencing peripheral adverse effects from amantadine: report of a case series". Mov Disord 20 (7): 873–877. July 2005. doi:10.1002/mds.20471. PMID 15809995.
- ↑ "Study of rimantadine in the USSR: a review of the literature". Reviews of Infectious Diseases 3 (3): 408–21. 1981-05-01. doi:10.1093/clinids/3.3.408. PMID 7025146.
- ↑ "The roles of amantadine, rimantadine, ursodeoxycholic acid, and NSAIDs, alone or in combination with alpha interferons, in the treatment of chronic hepatitis C". Seminars in Liver Disease 19 (Suppl 1): 95–102. 1999. PMID 10349697.
- ↑ "CDC - Influenza (Flu) | Antivirals: Side-Effects | REMOVED!". https://www.cdc.gov/flu/professionals/treatment/side-effects.htm.
- ↑ "Rationing of influenza vaccine during a pandemic: ethical analyses". Vaccine 25 (11): 2019–26. March 2007. doi:10.1016/j.vaccine.2006.11.045. PMID 17258359.
- ↑ "fda.gov". https://www.fda.gov/cder/drug/antivirals/influenza/flumadine.htm.
- ↑ "Rationing of influenza vaccine during a pandemic: ethical analyses". Vaccine 25 (11): 2019–26. March 2007. doi:10.1016/j.vaccine.2006.11.045. PMID 17258359.
- ↑ "Aminoadamantanes as NMDA receptor antagonists and antiparkinsonian agents--preclinical studies". Neurosci Biobehav Rev 21 (4): 455–468. July 1997. doi:10.1016/s0149-7634(96)00037-1. PMID 9195603.
- ↑ "Amantadine: reappraisal of the timeless diamond-target updates and novel therapeutic potentials". J Neural Transm (Vienna) 128 (2): 127–169. February 2021. doi:10.1007/s00702-021-02306-2. PMID 33624170.
- ↑ Liu, Tiqing. "BindingDB BDBM50216627 (alpha-methyl-1-adamantyl)methylamine::1-Adamantan-1-yl-ethylamine::CHEMBL959::RIMANTADINE::US11241393, Rimantadine::rimantadin::rimantidin". https://www.bindingdb.org/rwd/bind/chemsearch/marvin/MolStructure.jsp?monomerid=50216627.
- ↑ "PDSP Database" (in zu). https://pdsp.unc.edu/databases/pdsp.php?receptorDD=&receptor=&speciesDD=&species=&sourcesDD=&source=&hotLigandDD=&hotLigand=&testLigandDD=&testFreeRadio=testFreeRadio&testLigand=Rimantadine&referenceDD=&reference=&KiGreater=&KiLess=&kiAllRadio=all&doQuery=Submit+Query.
- ↑ "rimantadine (NMDA OR NMDAR OR NMDARs OR "N-methyl-D-aspartate")". https://pubmed.ncbi.nlm.nih.gov/?term=rimantadine+%28NMDA+OR+NMDAR+OR+NMDARs+OR+%22N-methyl-D-aspartate%22%29&sort=date&sort_order=asc&size=200.
- ↑ "Synthesis and pharmacological evaluation of several ring-contracted amantadine analogs". Bioorg Med Chem 16 (23): 9925–9936. December 2008. doi:10.1016/j.bmc.2008.10.028. PMID 18954995.
- ↑ "Synthesis and antiviral activity of metabolites of rimantadine". Journal of Medicinal Chemistry 33 (7): 1992–5. July 1990. doi:10.1021/jm00169a029. PMID 2362279.
- ↑ US patent 3352912 to W. W. Prichard
- ↑ United States Patent No. 4551552: Process for preparing rimantadine: Rimantadine and related compounds useful as antivirals were first described by Prichard in U.S. Pat. Nos. 3,352,912 and 3,592,934. Both patents describe the preparation of rimantadine from the corresponding ketone oxime by reduction with lithium aluminum hydride.
- ↑ United States Patent No. 4551552: Process for preparing rimantadine
- ↑ "Study of rimantadine in the USSR: a review of the literature". Reviews of Infectious Diseases 3 (3): 408–21. 1981. doi:10.1093/clinids/3.3.408. PMID 7025146.
External links
- U.S. FDA press release announcing rimantadine's approval
- U.S. Center for Drug Evaluation and Research rimantadine description
- U.S. NIH rimantadine description
- U.S. CDC flu anti-viral treatment information
 |
