Chemistry:Amantadine
Amantadine, sold under the brand name Gocovri among others, is a medication used to treat dyskinesia associated with parkinsonism and influenza caused by type A influenzavirus, though its use for the latter is no longer recommended because of widespread drug resistance.[1][2] It is also used for a variety of other conditions. The drug is taken by mouth.
Amantadine has a mild side-effect profile. Common neurological side effects include drowsiness, lightheadedness, dizziness, and confusion.[3] Because of its effects on the central nervous system (CNS), it should be combined cautiously with additional CNS stimulants or anticholinergic drugs. Given that it is cleared by the kidneys, amantadine is contraindicated in persons with end-stage kidney disease.[4] Due to its anticholinergic effects, it should be taken with caution by those with enlarged prostates or glaucoma.[5]
The pharmacology of amantadine is complex.[6][7] It acts as a sigma σ1 receptor agonist, nicotinic acetylcholine receptor negative allosteric modulator, dopaminergic agent, and weak NMDA receptor antagonist, among other actions.[6][7] The precise mechanism of action of its therapeutic effects in the treatment of CNS disorders is unclear.[6][7] The antiviral mechanism of action is inhibition of the influenza virus A M2 proton channel, which prevents endosomal escape (i.e., the release of viral genetic material into the host cytoplasm).[8][9] Amantadine is an adamantane derivative and is related to memantine and rimantadine.[10]
Amantadine was first used for the treatment of influenza A.[6] After its antiviral properties were initially reported in 1963, amantadine received approval for prophylaxis against the influenza virus A in 1966.[6][11] In 1968, its antiparkinsonian effects were serendipitously discovered.[6] In 1973, the Food and Drug Administration (FDA) approved amantadine for use in the treatment of Parkinson's disease.[6] In 2020, the extended-release formulation was approved for use in the treatment of levodopa-induced dyskinesia.[6][12]
Medical uses
Amantadine was initially developed to prevent replication of the influenza A virus.[13] Its main clinical use today is treatment of Parkinson's disease.[13] Other uses include treatment of drug-induced extrapyramidal side effects, motor fluctuations during levodopa therapy in Parkinson's disease, traumatic brain injury, and autistic spectrum disorders.[13]
Parkinson's disease
Amantadine is used to treat Parkinson's disease-related dyskinesia and drug-induced parkinsonism syndromes.[14] Amantadine may be used alone or in combination with another anti-Parkinson's or anticholinergic drug.[15] The specific symptoms targeted by amantadine therapy are dyskinesia and rigidity.[14] The extended release amantadine formulation is commonly used to treat dyskinesias in people receiving levodopa therapy for Parkinson's disease.[14] A 2003 Cochrane review had concluded evidence was insufficient to prove the safety or efficacy of amantadine to treat dyskinesia.[16]
In 2008, the World Health Organization (WHO) reported amantadine is not effective as a stand-alone parkinsonian therapy, but recommended it could be used in combination therapy with levodopa.[17]
Influenza A
Amantadine is not recommended for treatment or prophylaxis of influenza A in the United States.[1] Amantadine has no effect preventing or treating influenza B infections.[1] The US Centers for Disease Control and Prevention (CDC) found 100% of seasonal H3N2 and 2009 pandemic flu samples were resistant to adamantanes (amantadine and rimantadine) during the 2008–2009 flu season.[15][18]
The U.S. CDC guidelines recommend only neuraminidase inhibitors for influenza treatment and prophylaxis. The CDC recommends against amantadine and rimantadine to treat influenza A infections.[1]
Similarly, the 2011 WHO virology report showed all tested H1N1 influenza A viruses were resistant to amantadine.[2] WHO guidelines recommend against use of M2 inhibitors for influenza A. The continued high rate of resistance observed in laboratory testing of influenza A has reduced the priority of M2 resistance testing.
A 2014 Cochrane review did not find evidence for efficacy or safety of amantadine used for the prevention or treatment of influenza A.[19]
Extrapyramidal symptoms
An extended-release formulation of amantadine is used to treat levodopa-induced dyskinesia in patients with Parkinson's disease.[20] The WHO recommends the use of amantadine as a combination therapy to reduce levodopa side effects.[17]
Off-label uses
Fatigue in multiple sclerosis
A 2007 Cochrane literature review concluded that no overall evidence supports the use of amantadine in treating fatigue in patients with multiple sclerosis (MS).[21] A follow-up 2012 Cochrane review stated that some amantadine-induced improvement in fatigue may occur in some people with MS.[22] Despite multiple control trials that have also demonstrated improvements in subjective and objective ratings of fatigue, no final conclusion has been drawn regarding its effectiveness.[23]
Consensus guidelines from the German Multiple Sclerosis Society (GMSS) in 2006 state that amantadine produces moderate improvement in subjective fatigue, problem solving, memory, and concentration. Thus, in 2006, GMSS guidelines recommended the use of amantadine in MS-related fatigue.[24]
In the UK, NICE recommends considering amantadine for MS fatigue.[25]
Disorders of consciousness
Disorders of consciousness (DoC) include coma, vegetative state (VS), and minimally conscious state (MCS). Amantadine has been shown to increase the rate of emergence from a MCS, defined by consistent demonstration of interactive communication and functional objective use. In traumatic brain injury patients in the intensive care unit, amantadine has also been shown in various randomized control trials to increase the rate of functional recovery and arousal, particularly in the time period immediately following an injury.[26] Also, significantly improved consciousness has been reported in patients treated for nontraumatic cases of DoC, such as in the case of a subarachnoid hemorrhage, cerebral hemorrhage, and hypoxic encephalopathy.[27] In 2018, the American Academy of Neurology updated treatment guidelines on the use of amantadine for patients with prolonged DoC, recommending the use of amantadine (100–200 mg b.i.d.) for adults with DoC 4 to 16 weeks after injury to support early functional recovery and reduce disability.[28]
Brain injury recovery
In various studies, amantadine and memantine have been shown to accelerate the rate of recovery from a brain injury.[29][30] The time-limited window following a brain injury is characterized by neuroplasticity, or the capacity of neurons in the brain to adapt and compensate after injury. Thus, physiatrists often start patients on amantadine as soon as impairments are recognized. Some case reports also show improved functional recovery with amantadine treatment occurring years after the initial brain injury.[26] Evidence is insufficient to determine if the functional gains are a result of effects through the dopamine or norepinephrine pathways. Some patients may benefit from direct dopamine stimulation with amantadine, while others may benefit more from other stimulants that act more on the norepinephrine pathway, such as methylphenidate.[26] If treatment with amantadine improves long-term outcomes or simply accelerates recovery is unclear.[29] Nonetheless, amantadine-induced acceleration of recovery reduces the burden of disability, lessens health-care costs, and minimizes psychosocial stressors in patients.[citation needed]
Contraindications
Amantadine is contraindicated in persons with end-stage kidney disease,[20] as the drug is renally cleared.[31][5][32]
Amantadine may have anticholinergic side effects. Thus, patients with an enlarged prostate or glaucoma should use with caution.[3]
Live attenuated vaccines are contraindicated while taking amantadine.[20] Amantadine might inhibit viral replication and reduce the efficacy of administered vaccines. The U.S. Food and Drug Administration recommends avoiding amantadine for two weeks prior to vaccine administration and 48 hours afterward.[5]
Side effects
Amantadine is generally well tolerated and has a mild side effect profile.[33]
Neurological
Side effects include drowsiness (especially while driving), lightheadedness, falls, and dizziness.[20] Patients on amantadine should avoid combination with other CNS-depressing agents, such as alcohol. Excessive alcohol usage may increase the potential for CNS effects such as dizziness, confusion, and light-headedness.[3]
Rare severe adverse effects include neuroleptic malignant syndrome, depression, convulsions, psychosis, and suicidal ideation.[3] It has also been associated with disinhibited actions (gambling, sexual activity, spending, other addictions) and diminished control over compulsions.[20]
Amantadine may cause anxiety, feeling overexcited, hallucinations, and nightmares.[34]
Cardiovascular
Amantadine may cause orthostatic hypotension, syncope, and peripheral edema.[20]
Gastrointestinal
Amantadine has also been associated with dry mouth and constipation.[20]
Skin
Rare cases of skin rashes, such as Stevens–Johnson syndrome and livedo reticularis have also been reported in patients treated with amantadine.[35][36]
Kidney
Amantadine inhibits the kidney's active-transport removal and transfer of creatinine from blood to urine, which normally occurs in the proximal tubules of the nephrons. The active-transport removal mechanism accounts for about 15% of creatinine clearance, so amantadine may increase serum creatinine concentrations 15% above normal levels and give the false impression of mild kidney disease in patients whose kidneys are actually undamaged (because kidney function is often assessed by measuring the concentration of creatinine in blood.) Also, if the patient does have kidney disease, amantadine may cause it to appear as much as 15% worse than it actually is.[37]
Pregnancy and lactation
Amantadine is USFDA category C for pregnancy. Teratogenic effects have been observed in humans (case reports) and animal reproduction studies. Amantadine may also be present in breast milk and negatively alter breast milk production or excretion. The decision to breastfeed during amantadine therapy should consider the risk of infant exposure, the benefits of breastfeeding, and the benefits of the drug to the mother.[3]
Interactions
Amantadine may affect the CNS because of its dopaminergic and anticholinergic properties. The mechanisms of action are not fully known. Because of the CNS effects, caution is required when prescribing additional CNS stimulants or anticholinergic drugs.[5] Thus, concurrent use of alcohol with amantadine is not recommended because of enhanced CNS depressant effects.[38] In addition, antidopaminergic drugs such as metoclopramide and typical antipsychotics should be avoided.[39][40] These interactions are likely related to opposing dopaminergic mechanisms of action, which inhibits amantadine's anti-Parkinson effects.
Pharmacology
Mechanism of action
Central nervous system disorders
The mechanism of action of the antiparkinsonian effects of amantadine is poorly understood.[41] The effects of amantadine in Parkinson's disease were originally assumed to be anticholinergic or dopaminergic, but the situation soon proved more complicated than this.[6][7] The pharmacodynamics of amantadine are complex, and it interacts with many different biological targets at a variety of concentrations and hence potencies.[6][7]
The drug is a weak antagonist of the NMDA-type glutamate receptor, increases dopamine release, and blocks dopamine reuptake.[6][7][42][43][44] It is a negative allosteric modulator of the nicotinic acetylcholine receptors, specifically the α4β2 and α7 nicotinic acetylcholine receptors.[6]
In 1993, amantadine was found to bind to the sigma σ1 receptor with relatively high affinity (Ki = 20.25 μM).[6][45] In 2004, it was discovered that amantadine and memantine bind to and act as agonists of the sigma σ1 receptor (Ki = 7.44 μM and 2.60 μM, respectively) and that activation of the σ1 receptor is potentially involved in the dopaminergic effects of amantadine at therapeutically relevant concentrations.[46] σ1 receptor activation is one of amantadine's more potent actions.[6][46] σ1 receptor agonists enhance tyrosine hydroxylase activity, modulate NMDA-stimulated dopamine release, increase dopamine release in the striatum in vivo, and decrease dopamine reuptake.[6] As such, σ1 receptor activation may be involved in the antiparkinsonian and other central nervous system effects of amantadine.[6][46]
Binding of amantadine to the NMDA receptor was first reported in 1989, and antagonism of the receptor was first reported in 1991.[6] Despite some reports, the NMDA receptor antagonism of amantadine is probably not its primary mechanism of action.[6][7] It occurs at relatively high concentrations and many of the effects of amantadine are different from those of NMDA receptor antagonists.[6] Some of its effects, such as enhancement of dopamine release in the striatum, are even reversed by NMDA receptor antagonists,[6] but NMDA receptor antagonism could still contribute to the effects of amantadine.[6]
Although some publications have reported that amantadine inhibits monoamine oxidase, the drug probably does not actually inhibit this enzyme.[6][7][47]
Amantadine shows amphetamine-like psychostimulant effects (e.g., stimulation of locomotor activity) in animals at sufficiently high doses.[7][48] It has been found to inhibit the reuptake of serotonin, norepinephrine, and dopamine and to induce the release of serotonin, norepinephrine, and dopamine.[7][6][49][50] The concentrations needed for these effects, though, are very high and may not be therapeutically relevant.[7][6] It is about 1/25th to 1/50th as potent as amphetamines.[7] Amantadine has been found to increase dopamine levels in the striatum.[7][6] It does not act as a monoaminergic activity enhancer.[48][51][52]
Amantadine is a phosphodiesterase inhibitor, for example of PDE1.[6]
Amantadine has been found to increase aromatic amino acid decarboxylase expression.[6] This enzyme is responsible for the synthesis of dopamine from L-DOPA.[6] An imaging study in humans found that amantadine increased AADC activity in the striatum by up to 27%.[6]
Various additional actions of amantadine have been described.[6]
Influenza
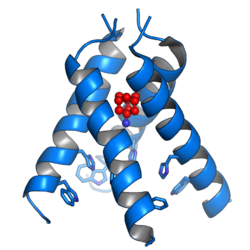
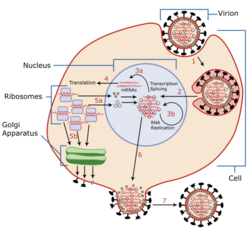
The mechanisms for amantadine's antiviral and antiparkinsonian effects are unrelated.[31][5] Amantadine targets the influenza A M2 ion channel protein. The M2 protein's function is to allow the intracellular virus to replicate (M2 also functions as a proton channel for hydrogen ions to cross into the vesicle), and exocytose newly formed viral proteins to the extracellular space (viral shedding). By blocking the M2 channel, the virus is unable to replicate because of impaired replication, protein synthesis, and exocytosis.[54]
Amantadine and rimantadine function in a mechanistically identical fashion, entering the barrel of the tetrameric M2 channel and blocking pore function—i.e., proton translocation.[15]
Resistance to the drug class is a consequence of mutations to the pore-lining amino acid residues of the channel, preventing both amantadine and rimantadine from binding and inhibiting the channel in their usual way.[55]
Pharmacokinetics
Amantadine is well-absorbed orally. The onset of action is usually within 48 hours when used for parkinsonian syndromes, including dyskinesia. As plasma concentrations of amantadine increase, the risk for toxicity increases.[56][57]
Half-life elimination averages eight days in patients with end-stage kidney disease. Amantadine is only minimally removed by hemodialysis.[57][58]
Amantadine is metabolized to a small extent (5–15%) by acetylation. It is mainly excreted (90%) unchanged in urine by kidney excretion.[56]
Chemistry
Amantadine is the organic compound 1-adamantylamine or 1-aminoadamantane, which consists of an adamantane backbone with an amino group substituted at one of the four tertiary carbons.[59] Rimantadine is a closely related adamantane derivative with similar biological properties;[60] both target the M2 proton channel of influenza A virus.[15]
Amantadine (1-aminoadamantane) is structurally related to other adamantanes including adapromine (1-(adamantan-1-yl)propan-1-amine), bromantane (N-(4-bromophenyl)adamantan-2-amine), memantine (1-amino-3,5-dimethyladamantane), and rimantadine (1-(1-aminoethyl)adamantane), among others.[citation needed]
History
Influenza A
Antiviral properties were first reported in 1963 at the University of Illinois Hospital in Chicago. In this amantadine trial study, volunteer college students were exposed to a viral challenge. The group who received amantadine (100 milligrams 18 hours before viral challenge) had less Asia influenza infections than the placebo group.[11] Amantadine received approval for the treatment of influenza virus A[61][62][63][64] in adults in 1976.[11] It was first used in West Germany in 1966. Amantadine was approved by the U.S. Food and Drug Administration in October 1968, as a prophylactic agent against Asian (H2N2) influenza and received approval for prophylactic use for influenza A in 1976.[11][4][65]
During the 1980 influenza A epidemic, the first amantadine-resistance influenza viruses were reported. The frequency of amantadine resistance among influenza A (H3N2) viruses from 1991 and 1995 was as low as 0.8%. In 2004, the resistance frequency increased to 12.3%. A year later, resistance increase significantly to 96%, 72%, and 14.5% in China, South Korea, and the United States, respectively. By 2006, 90.6% of H3N2 strains and 15.6% of H1N1 were amantadine resistant. A majority of the amantadine-resistant H3N2 isolates (98.2%) was found to contain an S31N mutation in the M2 transmembrane domain that confers resistance to amantadine.[66] Currently, adamantane resistance is high among circulating influenza A viruses. Thus, they are no longer recommended for treatment of influenza A.[67]
Parkinson's disease
An incidental finding in 1969 prompted investigations about amantadine's effectiveness for treating symptoms of Parkinson's disease.[11] A woman with Parkinson's disease was prescribed amantadine to treat her influenza infection and reported her cogwheel rigidity and tremors improved. She also reported that her symptoms worsened after she finished the course of amantadine.[11] The published case report was not initially corroborated by any other instances by the medical literature or manufacturer data. A team of researchers looked at a group of 10 patients with Parkinson's disease and gave them amantadine. Seven of them showed improvement, which was convincing evidence for the need of a clinical trial, which included 163 patients with Parkinson's disease; 66% experienced subjective or objective reduction of symptoms with a maximum daily dose of 200 mg.[11][68] Additional studies followed patients for greater lengths of time and in different combinations of neurological drugs.[69] It was found to be a safe drug that could be used over long periods of time with few side effects as monotherapy or in combination with L-dopa or anticholinergic drugs.[11] By April 1973, the U.S. FDA approved amantadine for use in the treatment of Parkinson's disease.[5][11]
In 2017, the U.S. FDA approved the use of amantadine in an extended-release formulation for the treatment of dyskinesia, an adverse effect of levodopa in people with Parkinson's disease.[70][71]
Society and culture
Names
Brand names of amantadine include Gocovri, Symadine, and Symmetrel.[72][31][73]
Recreational use
Recreational use of amantadine at supratherapeutic doses has been reported.[74][75] It is a weak NMDA receptor antagonist and is reported to produce dissociative and phencyclidine-like effects in animals and humans at sufficiently high doses.[74][76][77][75] However, the very long duration of action of amantadine (>40 hours) has likely limited its misuse potential.[74] In any case, amantadine has been encountered as a novel designer drug.[75] Recreational use of the related drug memantine has similarly been reported.[74]
Veterinary misuse
In 2005, Chinese poultry farmers were reported to have used amantadine to protect birds against avian influenza.[78] In Western countries and according to international livestock regulations, amantadine is approved only for use in humans. Chickens in China have received an estimated 2.6 billion doses of amantadine.[78] Avian flu (H5N1) strains in China and southeast Asia are now resistant to amantadine, although strains circulating elsewhere still seem to be sensitive. If amantadine-resistant strains of the virus spread, the drugs of choice in an avian flu outbreak will probably be restricted to neuraminidase inhibitors oseltamivir and zanamivir, which block the action of viral neuraminidase enzyme on the surface of influenza virus particles.[66] Increasing incidence of oseltamivir resistance in circulating influenza strains (e.g., H1N1) exists, highlighting the need for new anti-influenza therapies.[79]
In September 2015, the U.S. FDA announced the recall of Dingo Chip Twists "Chicken in the Middle" dog treats because the product has the potential to be contaminated with amantadine.[80]
Research
Depression
Interest in and study of amantadine in the treatment of depression has arisen.[7][81][13][6][82] A 2017 systematic review of off-label augmentation for treatment of unipolar depression found two open-label studies of amantadine for augmenting imipramine and found that it was effective.[83] However, the quality of evidence was very low and no conclusions could be drawn about its effectiveness.[83] A 2022 systematic review of randomized controlled trials of glutamatergic agents for treatment-resistant depression identified one clinical trial of amantadine for this use.[82] Amantadine was found to be effective in treating depressive symptoms in the trial.[82] The mechanism of action of amantadine in the treatment of depression is unclear, but various mechanisms have been postulated.[13][81] These include dopaminergic actions like indirect enhancement of dopamine release, dopamine reuptake inhibition, and D2 receptor interactions, noradrenergic actions, glutamatergic actions such as NMDA receptor antagonism, and immunomodulation, among many others.[13][81][7]
Attention deficit hyperactivity disorder
Amantadine has been studied in the treatment of attention-deficit hyperactivity disorder (ADHD).[84] A 2010 randomized clinical trial showed similar improvements in ADHD symptoms in children treated with amantadine as in those treated with methylphenidate, with less frequent side effects.[85] A 2021 retrospective study showed that amantadine may serve as an effective adjunct to stimulants for ADHD-related symptoms and appears to be a safer alternative to second- or third-generation antipsychotics.[86]
COVID-19
Amantadine has been studied in the treatment of COVID-19.[87][88]
References
- ↑ 1.0 1.1 1.2 1.3 "Influenza Antiviral Medications: Summary for Clinicians". 17 April 2019. https://www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm.
- ↑ 2.0 2.1 "Summary of influenza antiviral susceptibility surveillance findings". September 2010 – March 2011. https://www.who.int/influenza/gisrs_laboratory/updates/antiviral_susceptibility/en/.
- ↑ 3.0 3.1 3.2 3.3 3.4 "Amantadine". StatPearls. Treasure Island (FL): StatPearls Publishing. 2020. https://www.ncbi.nlm.nih.gov/books/NBK499953/. Retrieved 2 November 2020.
- ↑ 4.0 4.1 "Gocovri- amantadine capsule, coated pellets". 26 December 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=82ff865f-78f3-4c94-b6a9-727a54944b28.
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 "Symmetrel (Amantadine Hydrochloride, USP) fact sheet". https://www.accessdata.fda.gov/drugsatfda_docs/label/2009/016023s041,018101s016lbl.pdf.
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 6.10 6.11 6.12 6.13 6.14 6.15 6.16 6.17 6.18 6.19 6.20 6.21 6.22 6.23 6.24 6.25 6.26 6.27 6.28 6.29 6.30 "Amantadine: reappraisal of the timeless diamond-target updates and novel therapeutic potentials". J Neural Transm (Vienna) 128 (2): 127–169. February 2021. doi:10.1007/s00702-021-02306-2. PMID 33624170.
- ↑ 7.00 7.01 7.02 7.03 7.04 7.05 7.06 7.07 7.08 7.09 7.10 7.11 7.12 7.13 7.14 "Possible use of amantadine in depression". Pharmacopsychiatry 32 (2): 47–55. March 1999. doi:10.1055/s-2007-979191. PMID 10333162.
- ↑ "Influenza Viruses". Infectious Diseases. Elsevier. 2017. pp. 1465–1471.e1. doi:10.1016/b978-0-7020-6285-8.00172-6. ISBN 978-0-7020-6285-8.
- ↑ "Inhibitors of the influenza A virus M2 proton channel discovered using a high-throughput yeast growth restoration assay". PLOS ONE 8 (2). 1 February 2013. doi:10.1371/journal.pone.0055271. PMID 23383318. Bibcode: 2013PLoSO...855271B.
- ↑ "Nascent pharmacological advancement in adamantane derivatives". Arch Pharm (Weinheim) 357 (3). March 2024. doi:10.1002/ardp.202300595. PMID 38128028.
- ↑ 11.0 11.1 11.2 11.3 11.4 11.5 11.6 11.7 11.8 "Amantadine: the journey from fighting flu to treating Parkinson disease". Neurology 78 (14): 1096–9. April 2012. doi:10.1212/WNL.0b013e31824e8f0d. PMID 22474298.
- ↑ "Amantadine delayed release/extended release capsules significantly reduce OFF time in Parkinson's disease". npj Parkinson's Disease 8 (1). March 2022. doi:10.1038/s41531-022-00291-1. PMID 35304480.
- ↑ 13.0 13.1 13.2 13.3 13.4 13.5 "Potential antidepressant effect of amantadine: a review of preclinical studies and clinical trials". Braz J Psychiatry 40 (4): 449–458. 2018. doi:10.1590/1516-4446-2017-2393. PMID 29898194.
- ↑ 14.0 14.1 14.2 "Amantadine in the treatment of Parkinson's disease and other movement disorders". The Lancet. Neurology 20 (12): 1048–1056. December 2021. doi:10.1016/s1474-4422(21)00249-0. PMID 34678171.
- ↑ 15.0 15.1 15.2 15.3 Principles of pharmacology: the pathophysiologic basis of drug therapy (4th ed.). Philadelphia: Wolters Kluwer. 2017. pp. 142, 199, 205t, 224t, 608, 698–700. ISBN 9781451191004. OCLC 914593652.
- ↑ "Amantadine for dyskinesia in Parkinson's disease". The Cochrane Database of Systematic Reviews 2010 (2). 22 April 2003. doi:10.1002/14651858.CD003467. PMID 12804468.
- ↑ 17.0 17.1 WHO Model Formulary 2008. World Health Organization (WHO). 2009. p. 242. ISBN 978-9241547659.
- ↑ "Seasonal Influenza (Flu) – Weekly Report: Influenza Summary Update". 5 September 2009. https://www.cdc.gov/flu/weekly/weeklyarchives2008-2009/weekly35.htm.
- ↑ "Amantadine and rimantadine for influenza A in children and the elderly". The Cochrane Database of Systematic Reviews 2014 (11). November 2014. doi:10.1002/14651858.CD002745.pub4. PMID 25415374.
- ↑ 20.0 20.1 20.2 20.3 20.4 20.5 20.6 "Gocovri- amantadine capsule, coated pellets". 26 December 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=82ff865f-78f3-4c94-b6a9-727a54944b28.
- ↑ "Amantadine for fatigue in multiple sclerosis". The Cochrane Database of Systematic Reviews 2007 (1). January 2007. doi:10.1002/14651858.CD002818.pub2. PMID 17253480.
- ↑ "WITHDRAWN: Interventions for fatigue and weight loss in adults with advanced progressive illness". The Cochrane Database of Systematic Reviews (Chichester, UK: John Wiley & Sons, Ltd) 4 (4). April 2017. doi:10.1002/14651858.cd008427. PMID 28387447.
- ↑ "Amantadine: multiple sclerosis-related fatigue". Hospital Pharmacy 49 (8): 710–2. September 2014. doi:10.1310/hpj4908-710. PMID 25477595.
- ↑ "Symptomatic treatment of multiple sclerosis. Multiple Sclerosis Therapy Consensus Group (MSTCG) of the German Multiple Sclerosis Society". European Neurology 56 (2): 78–105. 2006. doi:10.1159/000095699. PMID 16966832. https://www.karger.com/Article/FullText/95699.
- ↑ "Recommendations | Multiple sclerosis in adults: Management | Guidance | NICE". 22 June 2022. https://www.nice.org.uk/guidance/ng220/chapter/recommendations#ms-symptom-management-and-rehabilitation.
- ↑ 26.0 26.1 26.2 "Amantadine and memantine: a comprehensive review for acquired brain injury". Brain Injury 34 (3): 299–315. February 2020. doi:10.1080/02699052.2020.1723697. PMID 32078407.
- ↑ "The use of amantadine in patients with unresponsive wakefulness syndrome after severe cerebral hemorrhage". Brain Injury 34 (8): 1084–1088. July 2020. doi:10.1080/02699052.2020.1780315. PMID 32552090.
- ↑ "Practice guideline update recommendations summary: Disorders of consciousness: Report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology; the American Congress of Rehabilitation Medicine; and the National Institute on Disability, Independent Living, and Rehabilitation Research". Neurology 91 (10): 450–460. September 2018. doi:10.1212/WNL.0000000000005926. PMID 30089618.
- ↑ 29.0 29.1 "Placebo-controlled trial of amantadine for severe traumatic brain injury". The New England Journal of Medicine 366 (9): 819–26. March 2012. doi:10.1056/NEJMoa1102609. PMID 22375973.
- ↑ "Study of the neuroprotective effects of memantine in patients with mild to moderate ischemic stroke". Iranian Journal of Pharmaceutical Research 13 (2): 591–8. 2014. PMID 25237355.
- ↑ 31.0 31.1 31.2 Cite error: Invalid
<ref>tag; no text was provided for refs namedTGA - ↑ "Symmetrel (Amantadine) Prescribing Information". Endo Pharmaceuticals. May 2003. http://www.symmetrel.com/PDF/symmetrel_pack_insert.pdf.
- ↑ "Amantadine: a review of use in child and adolescent psychiatry". Journal of the Canadian Academy of Child and Adolescent Psychiatry 22 (1): 55–60. February 2013. PMID 23390434.
- ↑ Amantadine Hydrochloride package leaflet, Manx Healthcare, 1/24; Common possible side effects (may affect up to one in 10 people)
- ↑ "Cutaneous Adverse Effects of Neurologic Medications". CNS Drugs 30 (3): 245–67. March 2016. doi:10.1007/s40263-016-0318-7. PMID 26914914. http://link.springer.com/10.1007/s40263-016-0318-7.
- ↑ "Livedo reticularis during amantadine treatment". British Medical Journal 2 (5762): 627–8. June 1971. doi:10.1136/bmj.2.5762.627. PMID 5580722.
- ↑ "Some medicines increase serum creatinine without affecting glomerular function". https://medsafe.govt.nz/profs/PUArticles/December2019/Some-medicines-increase-serum-creatinine-without-affecting-glomerular-function.htm.
- ↑ Gocovri (amantadine) extended-release capsules [prescribing information]. Emeryville, CA: Adamas Pharma, LLC; August 2017
- ↑ Reglan (metoclopramide) [prescribing information]. Baudette, MN: ANI Pharmaceuticals Inc; August 2017
- ↑ "Metoclopramide and pimozide in Parkinson's disease and levodopa-induced dyskinesias". Journal of Neurology, Neurosurgery, and Psychiatry 38 (4): 331–335. April 1975. doi:10.1136/jnnp.38.4.331. PMID 1095689.
- ↑ "Amantadine – MeSH". NCBI. https://www.ncbi.nlm.nih.gov/mesh/68000547.
- ↑ Jasek, W, ed (2007) (in de). Austria-Codex (62nd ed.). Vienna: Österreichischer Apothekerverlag. p. 3962. ISBN 978-3-85200-181-4.
- ↑ "Effects of the 1-amino-adamantanes at the MK-801-binding site of the NMDA-receptor-gated ion channel: a human postmortem brain study". European Journal of Pharmacology 206 (4): 297–300. April 1991. doi:10.1016/0922-4106(91)90113-v. PMID 1717296.
- ↑ "Amantadine inhibits NMDA receptors by accelerating channel closure during channel block". The Journal of Neuroscience 25 (13): 3312–22. March 2005. doi:10.1523/JNEUROSCI.4262-04.2005. PMID 15800186.
- ↑ "Affinity of 1-aminoadamantanes for the sigma binding site in post-mortem human frontal cortex". Neurosci Lett 163 (2): 129–131. December 1993. doi:10.1016/0304-3940(93)90362-o. PMID 8309617.
- ↑ 46.0 46.1 46.2 "Involvement of the sigma 1 receptor in the modulation of dopaminergic transmission by amantadine". The European Journal of Neuroscience 19 (8): 2212–20. April 2004. doi:10.1111/j.0953-816X.2004.03297.x. PMID 15090047.
- ↑ "Further studies on the mode of action of amantadine". Acta Pharmacologica et Toxicologica 30 (3): 161–71. November 1971. doi:10.1111/j.1600-0773.1971.tb00646.x. PMID 5171936.
- ↑ 48.0 48.1 "(-)-1-(Benzofuran-2-yl)-2-propylaminopentane enhances locomotor activity in rats due to its ability to induce dopamine release". Eur J Pharmacol 421 (3): 181–189. June 2001. doi:10.1016/s0014-2999(01)01040-8. PMID 11516435.
- ↑ "The lipophilic bullet hits the targets: medicinal chemistry of adamantane derivatives". Chem Rev 113 (5): 3516–3604. May 2013. doi:10.1021/cr100264t. PMID 23432396.
- ↑ "The noradrenaline transporter as site of action for the anti-Parkinson drug amantadine". Neuropharmacology 62 (4): 1708–1716. March 2012. doi:10.1016/j.neuropharm.2011.11.017. PMID 22155208.
- ↑ "Structure-activity studies leading to (-)1-(benzofuran-2-yl)-2-propylaminopentane, ((-)BPAP), a highly potent, selective enhancer of the impulse propagation mediated release of catecholamines and serotonin in the brain". Bioorg Med Chem 9 (5): 1197–1212. May 2001. doi:10.1016/s0968-0896(01)00002-5. PMID 11377178.
- ↑ "(-)1-(Benzofuran-2-yl)-2-propylaminopentane, [(-)BPAP, a selective enhancer of the impulse propagation mediated release of catecholamines and serotonin in the brain"]. Br J Pharmacol 128 (8): 1723–1732. December 1999. doi:10.1038/sj.bjp.0702995. PMID 10588928.
- ↑ "High-resolution structures of the M2 channel from influenza A virus reveal dynamic pathways for proton stabilization and transduction". Proceedings of the National Academy of Sciences of the United States of America 112 (46): 14260–5. November 2015. doi:10.1073/pnas.1518493112. PMID 26578770. Bibcode: 2015PNAS..11214260T.
- ↑ "Amantadine". PubChem. U.S. Library of Medicine. https://pubchem.ncbi.nlm.nih.gov/compound/2130.
- ↑ "Drug resistance in influenza A virus: the epidemiology and management". Infection and Drug Resistance 10: 121–134. 20 April 2017. doi:10.2147/idr.s105473. PMID 28458567.
- ↑ 56.0 56.1 "Amantadine Hydrochloride Monograph for Professionals". https://www.drugs.com/monograph/amantadine-hydrochloride.html.
- ↑ 57.0 57.1 "Amantadine – FDA prescribing information, side effects and uses". https://www.drugs.com/pro/amantadine.html.
- ↑ "Clinical pharmacokinetic and pharmacodynamic properties of drugs used in the treatment of Parkinson's disease". Clinical Pharmacokinetics 41 (4): 261–309. 2002. doi:10.2165/00003088-200241040-00003. PMID 11978145.
- ↑ "Amantadine". https://www.drugbank.ca/drugs/DB00915.
- ↑ "Rimantadine hydrochloride (CHEBI:8865)". https://www.ebi.ac.uk/chebi/searchId.do?chebiId=CHEBI:8865.
- ↑ Science and Corporate Strategy: Du Pont R&D, 1902–1980 John. Cambridge University Press. 1988. p. 469. ISBN 978-0521327671. https://books.google.com/books?id=6ld0K9VNpmIC.
- ↑ "Sales of flu drug by du Pont unit a 'disappointment'". The New York Times (Wilmington, Delaware). 5 October 1982. https://query.nytimes.com/gst/fullpage.html?sec=health&res=9C02E0D71F38F936A35753C1A964948260.
- ↑ "Panel urges wide use of antiviral drug". Science 206 (4422): 1058–60. November 1979. doi:10.1126/science.386515. PMID 386515. Bibcode: 1979Sci...206.1058M.
- ↑ "Amantadine: an alternative for prevention of influenza". Science 192 (4235): 130–1. April 1976. doi:10.1126/science.192.4235.130. PMID 17792438.
- ↑ "International Review of Neurobiology", Recent advances in the use of Drosophila in neurobiology and neurodegeneration, International Review of Neurobiology, 99, Elsevier, 2011, pp. i–iii, doi:10.1016/b978-0-12-387003-2.00010-0, ISBN 978-0-12-387003-2, http://dx.doi.org/10.1016/b978-0-12-387003-2.00010-0, retrieved 11 November 2020
- ↑ 66.0 66.1 "The emerging influenza virus threat: status and new prospects for its therapy and control". Archives of Virology 163 (4): 831–844. April 2018. doi:10.1007/s00705-018-3708-y. PMID 29322273.
- ↑ "Antiviral Drug Resistance among Influenza Viruses". 17 April 2019. https://www.cdc.gov/flu/professionals/antivirals/antiviral-drug-resistance.htm.
- ↑ "Amantadine in the treatment of Parkinson's disease". JAMA 208 (7): 1168–1170. May 1969. doi:10.1001/jama.1969.03160070046011. PMID 5818715.
- ↑ "Amantadine in Parkinson's disease. Review of more than two years' experience". JAMA 222 (7): 792–5. November 1972. doi:10.1001/jama.222.7.792. PMID 4677928.
- ↑ "NDA 208944 Approval Letter". https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2017/208944orig1s000ltr.pdf.
- ↑ "Drug Approval Package: Gocovri (amantadine extended-release)". 29 June 2018. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2017/208944Orig1s000TOC.cfm.
- ↑ "Amantadine (International database)". 5 August 2024. https://www.drugs.com/international/amantadine.html.
- ↑ "Amantadine: MedlinePlus Drug Information". https://medlineplus.gov/druginfo/meds/a682064.html.
- ↑ 74.0 74.1 74.2 74.3 "From PCP to MXE: a comprehensive review of the non-medical use of dissociative drugs". Drug Test Anal 6 (7–8): 614–632. 2014. doi:10.1002/dta.1620. PMID 24678061.
- ↑ 75.0 75.1 75.2 "Амантадин" (in ru). https://aipsin.com/newsubstance/258/.
- ↑ "Evaluating the abuse potential of psychedelic drugs as part of the safety pharmacology assessment for medical use in humans". Neuropharmacology 142: 89–115. November 2018. doi:10.1016/j.neuropharm.2018.01.049. PMID 29427652.
- ↑ "Evaluation of the reinforcing and discriminative stimulus properties of the low-affinity N-methyl-D-aspartate channel blocker memantine". Behav Pharmacol 9 (3): 231–243. May 1998. PMID 9832937.
- ↑ 78.0 78.1 "Bird Flu Drug Rendered Useless". The Washington Post. 18 June 2005. https://www.washingtonpost.com/wp-dyn/content/article/2005/06/17/AR2005061701214_pf.html.
- ↑ "Influenza virus susceptibility and resistance to oseltamivir". Antiviral Therapy 12 (4 Pt B): 603–16. 2007. doi:10.1177/135965350701200S04.1. PMID 17944268.
- ↑ "Enforcement Report – Week of September 23, 2015". U.S. Food and Drug Administration (FDA). http://www.accessdata.fda.gov/scripts/enforcement/enforce_rpt-Product-Tabs.cfm?action=select&recall_number=V-261-2015&w=09232015&lang=eng.
- ↑ 81.0 81.1 81.2 "Role of calcium, glutamate and NMDA in major depression and therapeutic application". Prog Neuropsychopharmacol Biol Psychiatry 64: 325–33. January 2016. doi:10.1016/j.pnpbp.2015.02.015. PMID 25747801.
- ↑ 82.0 82.1 82.2 "Ketamine and Other Glutamate Receptor Modulating Agents for Treatment-Resistant Depression: A Systematic Review of Randomized Controlled Trials". Iran J Psychiatry 17 (3): 320–340. July 2022. doi:10.18502/ijps.v17i3.9733. PMID 36474699.
- ↑ 83.0 83.1 "Efficacy of off-label augmentation in unipolar depression: A systematic review of the evidence". Eur Neuropsychopharmacol 27 (5): 423–441. May 2017. doi:10.1016/j.euroneuro.2017.03.003. PMID 28318897.
- ↑ "Emerging drugs for the treatment of attention-deficit hyperactivity disorder (ADHD)". Expert Opin Emerg Drugs 25 (4): 395–407. December 2020. doi:10.1080/14728214.2020.1820481. PMID 32938246.
- ↑ "Amantadine versus methylphenidate in children and adolescents with attention deficit/hyperactivity disorder: a randomized, double-blind trial". Human Psychopharmacology 25 (7–8): 560–565. November 2010. doi:10.1002/hup.1154. PMID 21312290.
- ↑ "Amantadine for the treatment of childhood and adolescent psychiatric symptoms". Proceedings 34 (5): 566–570. September 2021. doi:10.1080/08998280.2021.1925827. PMID 34456474.
- ↑ "Przeciwzapalne działanie amantadyny i memantyny w zakażeniu SARS-CoV-2" (in Polish). Pol Merkur Lekarski 49 (289): 67–70. February 2021. PMID 33713098. https://medpress.com.pl/pubmed.php?article=28967. Retrieved 13 August 2024.
- ↑ "SARS-CoV-2 infection in patients with serious mental illness and possible benefits of prophylaxis with Memantine and Amantadine". Rom J Morphol Embryol 61 (4): 1007–1022. 2020. doi:10.47162/RJME.61.4.03. PMID 34171050.
External links
 |
