Physics:Plutonium-241
| General | |
|---|---|
| Symbol | 241Pu |
| Names | plutonium-241, Pu-241 |
| Protons | 94 |
| Neutrons | 147 |
| Nuclide data | |
| Natural abundance | 0 (synthetic) |
| Half-life | 14.33 years[1] |
| Decay products | 241Am 237U |
| Isotope mass | 241.056850[2] u |
| Decay modes | |
| Decay mode | Decay energy (MeV) |
| β− | 0.0208[3] |
| α | 5.140[3] |
| Isotopes of Chemistry:plutonium Complete table of nuclides | |
Plutonium-241 (The element Chemistry:Plutonium does not exist., Pu-241) is an isotope of plutonium formed when plutonium-240 captures a neutron. Like some other plutonium isotopes (especially 239Pu), 241Pu is fissile, with a neutron absorption cross section about one-third greater than that of 239Pu, and a similar probability of fissioning on neutron absorption, around 73%. In the non-fission case, neutron capture produces plutonium-242. In general, isotopes with an odd number of neutrons are both more likely to absorb a neutron and more likely to undergo fission on neutron absorption than isotopes with an even number of neutrons.
Decay properties
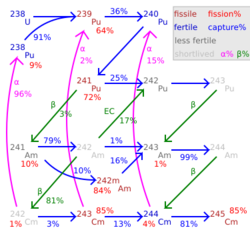
Plutonium-241 is a beta emitter with a half-life of 14.33 years, corresponding to a decay of about 5% of 241Pu nuclei over a one-year period. This decay has a Q-value of only 20.8 keV, and does not emit gamma rays.[3] The longer spent nuclear fuel waits before reprocessing, the more 241Pu decays to americium-241, which is nonfissile (although fissionable by fast neutrons) and an alpha emitter with a half-life of 432.6 years; 241Am, which does emit gamma rays, is a major contributor to the radioactivity of nuclear waste on a scale of hundreds to thousands of years.[citation needed] In its fully ionized state, the beta-decay half-life of 241Pu94+ decreases to 4.2 days, and only bound-state beta decay is possible.[4]
Plutonium-241 also has a rare alpha decay branch to uranium-237, occurring in about 0.0025% of decays. Unlike its usual beta decay, this can emit gamma rays, X-rays, and associated electrons.[1]
Actinides and fission products by half-life
| ||||||||
|---|---|---|---|---|---|---|---|---|
| Actinides[5] by decay chain | Half-life range (y) |
Fission products of 235U by yield<ref>Specifically from thermal neutron fission of U-235, e.g. in a typical nuclear reactor.</ref> | ||||||
| 4n | 4n+1 | 4n+2 | 4n+3 | |||||
| 4.5–7% | 0.04–1.25% | <0.001% | ||||||
| 228Ra№ | 4–6 | † | 155Euþ | |||||
| 244Cmƒ | 241Puƒ | 250Cf | 227Ac№ | 10–29 | 90Sr | 85Kr | 113mCdþ | |
| 232Uƒ | 238Puƒ | 243Cmƒ | 29–97 | 137Cs | 151Smþ | 121mSn | ||
| 248Bk[6] | 249Cfƒ | 242mAmƒ | 141–351 |
No fission products | ||||
| 241Amƒ | 251Cfƒ[7] | 430–900 | ||||||
| 226Ra№ | 247Bk | 1.3 k – 1.6 k | ||||||
| 240Pu | 229Th | 246Cmƒ | 243Amƒ | 4.7 k – 7.4 k | ||||
| 245Cmƒ | 250Cm | 8.3 k – 8.5 k | ||||||
| 239Puƒ | 24.1 k | |||||||
| 230Th№ | 231Pa№ | 32 k – 76 k | ||||||
| 236Npƒ | 233Uƒ | 234U№ | 150 k – 250 k | ‡ | 99Tc₡ | 126Sn | ||
| 248Cm | 242Pu | 327 k – 375 k | 79Se₡ | |||||
| 1.53 M | 93Zr | |||||||
| 237Npƒ | 2.1 M – 6.5 M | 135Cs₡ | 107Pd | |||||
| 236U | 247Cmƒ | 15 M – 24 M | 129I₡ | |||||
| 244Pu | 80 M |
... nor beyond 15.7 M years[8] | ||||||
| 232Th№ | 238U№ | 235Uƒ№ | 0.7 G – 14.1 G | |||||
|
Legend for superscript symbols | ||||||||
References
- ↑ 1.0 1.1 Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties". Chinese Physics C 45 (3). doi:10.1088/1674-1137/abddae. https://www-nds.iaea.org/amdc/ame2020/NUBASE2020.pdf.
- ↑ Template:AME2020 II
- ↑ 3.0 3.1 3.2 Basunia, M. S. (1 August 2006). "Nuclear Data Sheets for A = 237". Nuclear Data Sheets 107 (8): 2323–2422. doi:10.1016/j.nds.2006.07.001.
- ↑ Takahashi, K.; Boyd, R. N.; Mathews, G. J.; Yokoi, K. (1 October 1987). "Bound-state beta decay of highly ionized atoms". Physical Review C 36 (4): 1522–1528. doi:10.1103/PhysRevC.36.1522. https://www.researchgate.net/publication/13335547_Bound-state_beta_decay_of_highly_ionized_atoms.
- ↑ Plus radium (element 88). While actually a sub-actinide, it immediately precedes actinium (89) and follows a three-element gap of instability after polonium (84) where no nuclides have half-lives of at least four years (the longest-lived nuclide in the gap is radon-222 with a half life of less than four days). Radium's longest lived isotope, at 1,600 years, thus merits the element's inclusion here.
- ↑ Milsted, J.; Friedman, A. M.; Stevens, C. M. (1965). "The alpha half-life of berkelium-247; a new long-lived isomer of berkelium-248". Nuclear Physics 71 (2): 299. doi:10.1016/0029-5582(65)90719-4. Bibcode: 1965NucPh..71..299M.
"The isotopic analyses disclosed a species of mass 248 in constant abundance in three samples analysed over a period of about 10 months. This was ascribed to an isomer of Bk248 with a half-life greater than 9 y. No growth of Cf248 was detected, and a lower limit for the β− half-life can be set at about 104 y. No alpha activity attributable to the new isomer has been detected; the alpha half-life is probably greater than 300 y." - ↑ This is the heaviest nuclide with a half-life of at least four years before the "Sea of Instability".
- ↑ Excluding those "classically stable" nuclides with half-lives significantly in excess of 232Th; e.g., while 113mCd has a half-life of only fourteen years, that of 113Cd is nearly eight quadrillion years.
 |
