Biology:Ferredoxin
Ferredoxins (from Latin ferrum: iron + redox, often abbreviated "fd") are iron–sulfur proteins that mediate electron transfer in a range of metabolic reactions. The term "ferredoxin" was coined by D.C. Wharton of the DuPont Co. and applied to the "iron protein" first purified in 1962 by Mortenson, Valentine, and Carnahan from the anaerobic bacterium Clostridium pasteurianum.[1][2]
Another redox protein, isolated from spinach chloroplasts, was termed "chloroplast ferredoxin".[3] The chloroplast ferredoxin is involved in both cyclic and non-cyclic photophosphorylation reactions of photosynthesis. In non-cyclic photophosphorylation, ferredoxin is the last electron acceptor thus reducing the enzyme NADP+ reductase. It accepts electrons produced from sunlight-excited chlorophyll and transfers them to the enzyme ferredoxin: NADP+ oxidoreductase EC 1.18.1.2.
Ferredoxins are small proteins containing iron and sulfur atoms organized as iron–sulfur clusters. These biological "capacitors" can accept or discharge electrons, with the effect of a change in the oxidation state of the iron atoms between +2 and +3. In this way, ferredoxin acts as an electron transfer agent in biological redox reactions.
Other bioinorganic electron transport systems include rubredoxins, cytochromes, blue copper proteins, and the structurally related Rieske proteins.
Ferredoxins can be classified according to the nature of their iron–sulfur clusters and by sequence similarity.
Bioenergetics of ferredoxins
Ferredoxins typically carry out a single electron transfer.
- Fd0ox + e− [math]\ce{ <=> }[/math] Fd−red
However a few bacterial ferredoxins (of the 2[4Fe4S] type) have two iron sulfur clusters and can carry out two electron transfer reactions. Depending on the sequence of the protein, the two transfers can have nearly identical reduction potentials or they may be significantly different.[4][5]
- Fd0ox + e− [math]\ce{ <=> }[/math] Fd−red
- Fd−red + e− [math]\ce{ <=> }[/math] Fd2−red
Ferredoxins are one of the most reducing biological electron carriers. They typically have a mid point potential of -420 mV.[6] The reduction potential of a substance in the cell will differ from its midpoint potential depending on the concentrations of its reduced and oxidized forms. For a one electron reaction, the potential changes by around 60 mV for each power of ten change in the ratio of the concentration. For example, if the ferredoxin pool is around 95% reduced, the reduction potential will be around -500 mV.[7] In comparison, other biological reactions mostly have less reducing potentials: for example the primary biosynthetic reductant of the cell, NADPH has a cellular redox potential of -370 mV (E0 = -320 mV).
Depending on the sequence of the supporting protein ferredoxins have reduction potential from around -500mv[6][8] to -340 mV.[9] A single cell can have multiple types of ferredoxins where each type is tuned to optimally carry out different reactions.[10]
Reduction of ferredoxin
The highly reducing ferredoxins are reduced either by using another strong reducing agent, or by using some source of energy to "boost" electrons from less reducing sources to the ferredoxin.[11]
Direct reduction
Reactions that reduce Fd include the oxidation of aldehydes to acids like the glyceraldehyde to glycerate reaction (-580 mV), the carbon monoxide dehydrogenase reaction (-520 mV), and the 2-oxoacid:Fd Oxidoreductase reactions (-500 mV)[12][8] like the reaction carried out by pyruvate synthase.[7]
Membrane potential coupled reduction
Ferredoxin can also be reduced by using NADH (-320 mV) or H2 (-414 mV), but these processes are coupled to the consumption of the membrane potential to power the "boosting" of electrons to the higher energy state.[6] The Rnf complex is a widespread membrane protein in bacteria that reversibly transfers electrons between NADH and ferredoxin while pumping Na+ or H+ ions across the membrane. The chemiosmotic potential of the membrane is consumed to power the unfavorable reduction of Fdox by NADH. This reaction is an essential source of Fd−red in many autotrophic organisms. If the cell is growing on substrates that provide excess Fd−red, the Rnf complex can transfer these electrons to NAD+ and store the resultant energy in the membrane potential.[13] The energy converting hydrogenases (Ech) are a family of enzymes that reversibly couple the transfer of electrons between Fd and H2 while pumping H + ions across the membrane to balance the energy difference.[14]
- Fd0ox + NADH + Na+outside [math]\ce{ <=> }[/math] Fd2−red + NAD+ + Na+inside
- Fd0ox + H2 + H+outside [math]\ce{ <=> }[/math] Fd2−red + H+ + H+inside
Electron bifurcation
The unfavourable reduction of Fd from a less reducing electron donor can be coupled simultaneously with the favourable reduction of an oxidising agent through an electron bifurcation reaction.[6] An example of the electron bifurcation reaction is the generation of Fd−red for nitrogen fixation in certain aerobic diazotrophs. Typically in oxidative phosphorylation the transfer of electrons from NADH to Ubiquinone(Q) is coupled to charging the proton motive force. In Azotobacter the energy released by transferring one electron from NADH to Q is used to simultaneously boost the transfer of one electron from NADH to Fd.[15][16]
Direct reduction of high potential ferredoxins
Some ferredoxins have a sufficiently high redox potential that they can be directly reduced by NADPH. One such ferredoxin is adrenoxin (-274mV) which takes part in the biosynthesis of many mammalian steroids.[17] The ferredoxin Fd3 in the roots of plants that reduces nitrate and sulfite has a midpoint potential of -337mV and is also reduced by NADPH.[10]
Fe2S2 ferredoxins
| 2Fe-2S iron-sulfur cluster binding domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
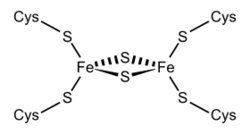 Structural representation of an Fe2S2 ferredoxin. | |||||||||
| Identifiers | |||||||||
| Symbol | Fer2 | ||||||||
| Pfam | PF00111 | ||||||||
| Pfam clan | CL0486 | ||||||||
| InterPro | IPR001041 | ||||||||
| PROSITE | PDOC00642 | ||||||||
| SCOP2 | 3fxc / SCOPe / SUPFAM | ||||||||
| OPM protein | 1kf6 | ||||||||
| |||||||||
Members of the 2Fe–2S ferredoxin superfamily (InterPro: IPR036010) have a general core structure consisting of beta(2)-alpha-beta(2), which includes putidaredoxin, terpredoxin, and adrenodoxin.[18][19][20][21] They are proteins of around one hundred amino acids with four conserved cysteine residues to which the 2Fe–2S cluster is ligated. This conserved region is also found as a domain in various metabolic enzymes and in multidomain proteins, such as aldehyde oxidoreductase (N-terminal), xanthine oxidase (N-terminal), phthalate dioxygenase reductase (C-terminal), succinate dehydrogenase iron–sulphur protein (N-terminal), and methane monooxygenase reductase (N-terminal).
Plant-type ferredoxins
One group of ferredoxins, originally found in chloroplast membranes, has been termed "chloroplast-type" or "plant-type" (InterPro: IPR010241). Its active center is a [Fe2S2] cluster, where the iron atoms are tetrahedrally coordinated both by inorganic sulfur atoms and by sulfurs of four conserved cysteine (Cys) residues.
In chloroplasts, Fe2S2 ferredoxins function as electron carriers in the photosynthetic electron transport chain and as electron donors to various cellular proteins, such as glutamate synthase, nitrite reductase, sulfite reductase, and the cyclase of chlorophyll biosynthesis.[22] Since the cyclase is a ferredoxin dependent enzyme this may provide a mechanism for coordination between photosynthesis and the chloroplasts need for chlorophyll by linking chlorophyll biosynthesis to the photosynthetic electron transport chain. In hydroxylating bacterial dioxygenase systems, they serve as intermediate electron-transfer carriers between reductase flavoproteins and oxygenase.
Thioredoxin-like ferredoxins
The Fe2S2 ferredoxin from Clostridium pasteurianum (Cp2FeFd; P07324) has been recognized as distinct protein family on the basis of its amino acid sequence, spectroscopic properties of its iron–sulfur cluster and the unique ligand swapping ability of two cysteine ligands to the [Fe2S2] cluster. Although the physiological role of this ferredoxin remains unclear, a strong and specific interaction of Cp2FeFd with the molybdenum-iron protein of nitrogenase has been revealed. Homologous ferredoxins from Azotobacter vinelandii (Av2FeFdI; P82802) and Aquifex aeolicus (AaFd; O66511) have been characterized. The crystal structure of AaFd has been solved. AaFd exists as a dimer. The structure of AaFd monomer is different from other Fe2S2 ferredoxins. The fold belongs to the α+β class, with first four β-strands and two α-helices adopting a variant of the thioredoxin fold.[23] UniProt categorizes these as the "2Fe2S Shethna-type ferredoxin" family.[24]
Adrenodoxin-type ferredoxins
| ferredoxin 1 | |
|---|---|
 Crystal structure of human ferredoxin-1 (FDX1).[25] | |
| Identifiers | |
| Symbol | FDX1 |
| Alt. symbols | FDX |
| NCBI gene | 2230 |
| HGNC | 3638 |
| OMIM | 103260 |
| RefSeq | NM_004109 |
| UniProt | P10109 |
| Other data | |
| Locus | Chr. 11 q22.3 |
Adrenodoxin (adrenal ferredoxin; InterPro: IPR001055), putidaredoxin, and terpredoxin make up a family of soluble Fe2S2 proteins that act as single electron carriers, mainly found in eukaryotic mitochondria and Pseudomonadota. The human variant of adrenodoxin is referred to as ferredoxin-1 and ferredoxin-2. In mitochondrial monooxygenase systems, adrenodoxin transfers an electron from NADPH:adrenodoxin reductase to membrane-bound cytochrome P450. In bacteria, putidaredoxin and terpredoxin transfer electrons between corresponding NADH-dependent ferredoxin reductases and soluble P450s.[26][27] The exact functions of other members of this family are not known, although Escherichia coli Fdx is shown to be involved in biogenesis of Fe–S clusters.[28] Despite low sequence similarity between adrenodoxin-type and plant-type ferredoxins, the two classes have a similar folding topology.
Ferredoxin-1 in humans participates in the synthesis of thyroid hormones. It also transfers electrons from adrenodoxin reductase to CYP11A1, a CYP450 enzyme responsible for cholesterol side chain cleavage. FDX-1 has the capability to bind to metals and proteins.[29] Ferredoxin-2 participates in heme A and iron–sulphur protein synthesis.[30]
Fe4S4 and Fe3S4 ferredoxins
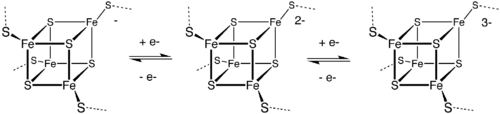
The [Fe4S4] ferredoxins may be further subdivided into low-potential (bacterial-type) and high-potential (HiPIP) ferredoxins.
Low- and high-potential ferredoxins are related by the following redox scheme:
The formal oxidation numbers of the iron ions can be [2Fe3+, 2Fe2+] or [1Fe3+, 3Fe2+] in low-potential ferredoxins. The oxidation numbers of the iron ions in high-potential ferredoxins can be [3Fe3+, 1Fe2+] or [2Fe3+, 2Fe2+].
Bacterial-type ferredoxins
| 3Fe-4S binding domain | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
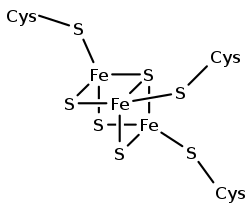 Structural representation of an Fe3S4 ferredoxin. | |||||||||||
| Identifiers | |||||||||||
| Symbol | Fer4 | ||||||||||
| Pfam | PF00037 | ||||||||||
| InterPro | IPR001450 | ||||||||||
| PROSITE | PDOC00176 | ||||||||||
| SCOP2 | 5fd1 / SCOPe / SUPFAM | ||||||||||
| OPM protein | 1kqf | ||||||||||
| |||||||||||
A group of Fe4S4 ferredoxins, originally found in bacteria, has been termed "bacterial-type". Bacterial-type ferredoxins may in turn be subdivided into further groups, based on their sequence properties. Most contain at least one conserved domain, including four cysteine residues that bind to a [Fe4S4] cluster. In Pyrococcus furiosus Fe4S4 ferredoxin, one of the conserved Cys residues is substituted with aspartic acid.
During the evolution of bacterial-type ferredoxins, intrasequence gene duplication, transposition and fusion events occurred, resulting in the appearance of proteins with multiple iron–sulfur centers. In some bacterial ferredoxins, one of the duplicated domains has lost one or more of the four conserved Cys residues. These domains have either lost their iron–sulfur binding property or bind to a [Fe3S4] cluster instead of a [Fe4S4] cluster[31] and dicluster-type.[32]
3-D structures are known for a number of monocluster and dicluster bacterial-type ferredoxins. The fold belongs to the α+β class, with 2-7 α-helices and four β-strands forming a barrel-like structure, and an extruded loop containing three "proximal" Cys ligands of the iron–sulfur cluster.
High-potential iron–sulfur proteins
High potential iron–sulfur proteins (HiPIPs) form a unique family of Fe4S4 ferredoxins that function in anaerobic electron transport chains. Some HiPIPs have a redox potential higher than any other known iron–sulfur protein (e.g., HiPIP from Rhodopila globiformis has a redox potential of ca. -450 mV). Several HiPIPs have so far been characterized structurally, their folds belonging to the α+β class. As in other bacterial ferredoxins, the [Fe4S4] unit forms a cubane-type cluster and is ligated to the protein via four Cys residues.
Human proteins from ferredoxin family
References
- ↑ "An electron transport factor from Clostridium pasteurianum". Biochemical and Biophysical Research Communications 7 (6): 448–52. June 1962. doi:10.1016/0006-291X(62)90333-9. PMID 14476372.
- ↑ "Bacterial Ferredoxin". Bacteriological Reviews 28 (4): 497–517. December 1964. doi:10.1128/MMBR.28.4.497-517.1964. PMID 14244728.
- ↑ "Ferredoxins as electron carriers in photosynthesis and in the biological production and consumption of hydrogen gas". Nature 195 (4841): 537–43. August 1962. doi:10.1038/195537a0. PMID 14039612. Bibcode: 1962Natur.195..537T.
- ↑ "Parsing redox potentials of five ferredoxins found within Thermotoga maritima". Protein Science 28 (1): 257–266. January 2019. doi:10.1002/pro.3547. PMID 30418685.
- ↑ "Discovery of a novel ferredoxin from Azotobacter vinelandii containing two [4Fe-4S] clusters with widely differing and very negative reduction potentials". The Journal of Biological Chemistry 273 (10): 5514–9. March 1998. doi:10.1074/jbc.273.10.5514. PMID 9488675.
- ↑ 6.0 6.1 6.2 6.3 "Flavin-Based Electron Bifurcation, Ferredoxin, Flavodoxin, and Anaerobic Respiration With Protons (Ech) or NAD+ (Rnf) as Electron Acceptors: A Historical Review". Frontiers in Microbiology 9: 401. 2018. doi:10.3389/fmicb.2018.00401. PMID 29593673.
- ↑ 7.0 7.1 "One-megadalton metalloenzyme complex in Geobacter metallireducens involved in benzene ring reduction beyond the biological redox window". Proceedings of the National Academy of Sciences of the United States of America 116 (6): 2259–2264. February 2019. doi:10.1073/pnas.1819636116. PMID 30674680. Bibcode: 2019PNAS..116.2259H.
- ↑ 8.0 8.1 "The Catalytic Bias of 2-Oxoacid:ferredoxin Oxidoreductase in CO2: Evolution and reduction through a ferredoxin-mediated electrocatalytic assay". Electrochimica Acta 199: 349–356. 2016. doi:10.1016/j.electacta.2016.02.119.
- ↑ "A two [4Fe-4S]-cluster-containing ferredoxin as an alternative electron donor for 2-hydroxyglutaryl-CoA dehydratase from Acidaminococcus fermentans". Archives of Microbiology 179 (3): 197–204. March 2003. doi:10.1007/s00203-003-0517-8. PMID 12610725.
- ↑ 10.0 10.1 "A post genomic characterization of Arabidopsis ferredoxins". Plant Physiology 134 (1): 255–64. January 2004. doi:10.1104/pp.103.032755. PMID 14684843.
- ↑ "Bioenergetic constraints on the origin of autotrophic metabolism". Philosophical Transactions. Series A, Mathematical, Physical, and Engineering Sciences 378 (2165): 20190151. February 2020. doi:10.1098/rsta.2019.0151. PMID 31902344. Bibcode: 2020RSPTA.37890151B.
- ↑ "A structural phylogeny for understanding 2-oxoacid oxidoreductase function". Current Opinion in Structural Biology 41: 54–61. December 2016. doi:10.1016/j.sbi.2016.05.011. PMID 27315560.
- ↑ "The Rnf Complex Is an Energy-Coupled Transhydrogenase Essential To Reversibly Link Cellular NADH and Ferredoxin Pools in the Acetogen Acetobacterium woodii". Journal of Bacteriology 200 (21). November 2018. doi:10.1128/JB.00357-18. PMID 30126940.
- ↑ "Energy-converting hydrogenases: the link between H2 metabolism and energy conservation". Cellular and Molecular Life Sciences 77 (8): 1461–1481. April 2020. doi:10.1007/s00018-019-03329-5. PMID 31630229.
- ↑ "The Electron Bifurcating FixABCX Protein Complex from Azotobacter vinelandii: Generation of Low-Potential Reducing Equivalents for Nitrogenase Catalysis". Biochemistry 56 (32): 4177–4190. August 2017. doi:10.1021/acs.biochem.7b00389. PMID 28704608.
- ↑ "Electron Transfer to Nitrogenase in Different Genomic and Metabolic Backgrounds". Journal of Bacteriology 200 (10). May 2018. doi:10.1128/JB.00757-17. PMID 29483165.
- ↑ "Adrenodoxin--a versatile ferredoxin". IUBMB Life 64 (6): 506–12. June 2012. doi:10.1002/iub.1029. PMID 22556163.
- ↑ "Crystallization and preliminary X-ray diffraction analysis of a [2Fe-2S] ferredoxin (FdVI) from Rhodobacter capsulatus". Acta Crystallographica. Section D, Biological Crystallography 57 (Pt 2): 301–3. February 2001. doi:10.1107/S0907444900017832. PMID 11173487.
- ↑ "Redox-dependent structural reorganization in putidaredoxin, a vertebrate-type [2Fe-2S] ferredoxin from Pseudomonas putida". Journal of Molecular Biology 347 (3): 607–21. April 2005. doi:10.1016/j.jmb.2005.01.047. PMID 15755454.
- ↑ "A model for the solution structure of oxidized terpredoxin, a Fe2S2 ferredoxin from Pseudomonas". Biochemistry 38 (17): 5666–75. April 1999. doi:10.1021/bi983063r. PMID 10220356.
- ↑ "A new electron transport mechanism in mitochondrial steroid hydroxylase systems based on structural changes upon the reduction of adrenodoxin". Biochemistry 41 (25): 7969–78. June 2002. doi:10.1021/bi0160361. PMID 12069587.
- ↑ "Aerobic Barley Mg-protoporphyrin IX Monomethyl Ester Cyclase is Powered by Electrons from Ferredoxin". Plants 9 (9): 1157. September 2020. doi:10.3390/plants9091157. PMID 32911631.
- ↑ "High resolution crystal structures of the wild type and Cys-55-->Ser and Cys-59-->Ser variants of the thioredoxin-like [2Fe-2S] ferredoxin from Aquifex aeolicus". The Journal of Biological Chemistry 277 (37): 34499–507. September 2002. doi:10.1074/jbc.M205096200. PMID 12089152.
- ↑ family:"2fe2s shethna type ferredoxin family"
- ↑ PDB: 3P1M; "Crystal structure of human ferredoxin-1 (FDX1) in complex with iron-sulfur cluster". Worldwide Protein Data Bank. 2010. doi:10.2210/pdb3p1m/pdb.
- ↑ "Putidaredoxin reductase and putidaredoxin. Cloning, sequence determination, and heterologous expression of the proteins". The Journal of Biological Chemistry 265 (11): 6066–73. April 1990. doi:10.1016/S0021-9258(19)39292-0. PMID 2180940.
- ↑ "Cytochrome P-450terp. Isolation and purification of the protein and cloning and sequencing of its operon". The Journal of Biological Chemistry 267 (20): 14193–203. July 1992. doi:10.1016/S0021-9258(19)49697-X. PMID 1629218.
- ↑ "Genetic analysis of the isc operon in Escherichia coli involved in the biogenesis of cellular iron-sulfur proteins". Journal of Biochemistry 130 (1): 63–71. July 2001. doi:10.1093/oxfordjournals.jbchem.a002963. PMID 11432781.
- ↑ "Entrez Gene: FDX1 ferredoxin 1". https://www.ncbi.nlm.nih.gov/sites/entrez?Db=gene&Cmd=ShowDetailView&TermToSearch=2230.
- ↑ "FDX2 ferredoxin 2 [Homo sapiens (human) - Gene - NCBI"]. https://www.ncbi.nlm.nih.gov/gene/112812.
- ↑ "Structure of [4Fe-4S] ferredoxin from Bacillus thermoproteolyticus refined at 2.3 A resolution. Structural comparisons of bacterial ferredoxins". Journal of Molecular Biology 210 (2): 383–98. November 1989. doi:10.1016/0022-2836(89)90338-0. PMID 2600971.
- ↑ "Refined crystal structure of the 2[4Fe-4S] ferredoxin from Clostridium acidurici at 1.84 A resolution". Journal of Molecular Biology 243 (4): 683–95. November 1994. doi:10.1016/0022-2836(94)90041-8. PMID 7966291.
Further reading
- "Structure, function and evolution of bacterial ferredoxins". FEMS Microbiology Reviews 4 (2): 155–75. 1988. doi:10.1111/j.1574-6968.1988.tb02741.x. PMID 3078742.
- "High potential iron-sulfur proteins and their role as soluble electron carriers in bacterial photosynthesis: tale of a discovery". Photosynthesis Research 85 (1): 115–31. 2005. doi:10.1007/s11120-004-6556-4. PMID 15977063.
- "Structure and function of plant-type ferredoxins". Photosynthesis Research 81 (3): 289–301. 2004. doi:10.1023/B:PRES.0000036882.19322.0a. PMID 16034533.
- "Adrenodoxin: structure, stability, and electron transfer properties". Proteins 40 (4): 590–612. September 2000. doi:10.1002/1097-0134(20000901)40:4<590::AID-PROT50>3.0.CO;2-P. PMID 10899784.
- "Structure-function studies of [2Fe-2S] ferredoxins". Journal of Bioenergetics and Biomembranes 26 (1): 67–88. February 1994. doi:10.1007/BF00763220. PMID 8027024.
- "Ferredoxins of the third kind". FEBS Letters 509 (1): 1–5. November 2001. doi:10.1016/S0014-5793(01)03049-6. PMID 11734195.
External links
- InterPro: IPR006057 - 2Fe–2S ferredoxin subdomain
- InterPro: IPR001055 - Adrenodoxin
- InterPro: IPR001450 - 4Fe–4S ferredoxin, iron–sulfur binding
- InterPro: IPR000170 - High potential iron–sulfur protein
- PDB: 1F37 - X-ray structure of thioredoxin-like ferredoxin from Aquifex aeolicus (AaFd)
 |