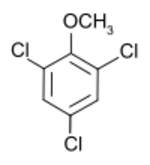
Chemistry:2,4,6-Trichloroanisole
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
1,3,5-Trichloro-2-methoxybenzene | |
| Other names
2,4,6-Trichloroanisole
TCA 2,4,6-Trichloromethoxybenzene | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| C7H5Cl3O | |
| Molar mass | 211.47 g·mol−1 |
| Melting point | 60 to 62 °C (140 to 144 °F; 333 to 335 K) |
| Boiling point | 140 °C (284 °F; 413 K) at 28 Torr |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H302, H319, H413 | |
| P264, P270, P273, P280, P301+312, P305+351+338, P330, P337+313, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
This article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these template messages)
(Learn how and when to remove this template message)
|
2,4,6-Trichloroanisole (TCA) is a chemical compound that represents one of the strongest of off-flavors, substances "generated naturally in foods/beverages [that considerably] deteriorate the quality" of such products.[1][2] As of 2000, TCA was considered the primary chemical compound responsible for the phenomenon of cork taint in wines,[3][1] and it has an unpleasant earthy, musty and moldy smell.[2]
Chemically, TCA is a chlorinated derivative of anisole, and it is a formed by the biomethylation of 2,4,6-trichlorophenol.[4][5] More generally, it may be produced when naturally occurring airborne fungi and bacteria are presented with chlorinated phenolic compounds, which they then convert into chlorinated anisole derivatives.[6] Species implicated include those of the genera Aspergillus, Penicillium, Actinomycetes, Botrytis (e.g. Botrytis cinerea), Rhizobium, or Streptomyces.[7][8][6]
The chlorophenol precursor, 2,4,6-trichlorophenol, is used as a fungicide; more generally, related compounds can originate as contaminants found in some pesticides and wood preservatives, or as by-products of the chlorine bleaching process used to sterilize or bleach wood, paper, and other materials.[9]
TCA has also been suggested as cause of the "Rio defect" in coffees from Brazil and other parts of the world,[10] which refers to a taste described as "medicinal, phenolic, or iodine-like".[5] In investigation of the mechanism of its role in producing off-flavor effects, it was found to "attenuate olfactory transduction by suppressing cyclic nucleotide-gated channels, without evoking odorant responses."[1]
Further reading
- Marsili, R. (2000). "Solid-Phase Microextraction: Food Technology Applications". in Wilson, Ian D.. Encyclopedia of Separation Science. New Yor, NY: Academic Press. pp. 4178–4190. doi:10.1016/B0-12-226770-2/06791-0. ISBN 9780122267703. https://www.sciencedirect.com/science/article/abs/pii/B0122267702067910. "Over the last two decades, the incidence of mouldy and musty off-flavours in cork-sealed wines has increased significantly. 2,4,6-Trichloroanisole (TCA) has been identified as the primary chemical responsible for cork taint. The human olfactometry threshold for TCA is 4–10 ng L−1 in white wine and 50 ng L−1 in red wine. In the case of wine, a worldwide loss of roughly US$1 billion per year is attributed to cork taint."
- Science Direct Staff (June 2023). "2-4-6-Trichloroanisole" (Science Direct citation sample/listing). https://www.sciencedirect.com/topics/chemistry/2-4-6-trichloroanisole.
- Buser, H.R.; Zanier, C. & Tanner, H. (1982). "Identification of 2,4,6-Trichloroanisole as a Potent Compound Causing Cork Taint in Wine.". Journal of Agricultural and Food Chemistry 30 (2): 359–362. doi:10.1021/jf00110a037. An early primary research report on the role of TCA in cork taint.
See also
References
- ↑ 1.0 1.1 1.2 Takeuchi, Hiroko; Kato, Hiroyuki & Kurahashi, Takashi (2013-09-16). "2,4,6-Trichloroanisole is a Potent Suppressor of Olfactory Signal Transduction". Proceedings of the National Academy of Sciences 110 (40): 16235–16240. doi:10.1073/pnas.1300764110. ISSN 1091-6490. PMID 24043819. Bibcode: 2013PNAS..11016235T.[non-primary source needed]
- ↑ 2.0 2.1 Jackson, Ron S. (2009). "Chapter 3: Olfactory Sensations" (in En). Wine tasting: a professional handbook. Food science and technology international series (2nd ed.). Academic Press. ISBN 978-0-12-374181-3.
- ↑ Marsili, R. (2000). "Solid-Phase Microextraction: Food Technology Applications". in Wilson, Ian D.. Encyclopedia of Separation Science. New Yor, NY: Academic Press. pp. 4178–4190. doi:10.1016/B0-12-226770-2/06791-0. ISBN 9780122267703. https://www.sciencedirect.com/science/article/abs/pii/B0122267702067910. "Over the last two decades, the incidence of mouldy and musty off-flavours in cork-sealed wines has increased significantly. 2,4,6-Trichloroanisole (TCA) has been identified as the primary chemical responsible for cork taint. The human olfactometry threshold for TCA is 4–10 ng L−1 in white wine and 50 ng L−1 in red wine. In the case of wine, a worldwide loss of roughly US$1 billion per year is attributed to cork taint."
- ↑ Pereira, Helena (2007-01-01), Pereira, Helena, ed., "Chapter 14 - Wine and cork", Cork (Amsterdam: Elsevier Science B.V.): pp. 305–327, ISBN 978-0-444-52967-1, https://www.sciencedirect.com/science/article/pii/B9780444529671500169, retrieved 2024-01-14
- ↑ 5.0 5.1 Spadone, Jean Claude; Takeoka, Gary & Liardon, Remy (1990). "Analytical Investigation of Rio Off-Flavor in Green Coffee". Journal of Agricultural and Food Chemistry 38: 226–233. doi:10.1021/jf00091a050. Note, at best, this source states that 2,4,6-trichlorophenol is "the probable precursor of TCA".
- ↑ 6.0 6.1 Cravero, Maria Carla; Bonello, Federica; Pazo Alvarez, Maria del Carmen; Tsolakis, Christos; Borsa, Daniela (24 June 2015). "The sensory evaluation of 2,4,6-trichloroanisole in wines: The sensory evaluation of 2,4,6-trichloroanisole in wines" (in en). Journal of the Institute of Brewing 121 (3): 411–417. doi:10.1002/jib.230. https://onlinelibrary.wiley.com/doi/10.1002/jib.230.
- ↑ Crane, Louise (22 March 2019). "Trichloroanisole: Cork taint" (in en). https://www.chemistryworld.com/podcasts/trichloroanisole-cork-taint/3010280.article.
- ↑ With regard to circumstantial evidence, Spodone, et al., op. cit., note that Rio off-flavor is associated with "beans heavily infested with various fungi (Aspergilli, Fusaria, Penicillia, Rhizopus, etc.) and bacteria (Lactobacilli, Streptrococci)".
- ↑ NTP (National Toxicology Program). 2021. "2,4,6-Trichlorophenol", Report on Carcinogens, Fifteenth Edition. Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service. https://ntp.niehs.nih.gov/go/roc15 DOI: https://doi.org/10.22427/NTP-OTHER-1003
- ↑ These include Central and South America.[citation needed]
 |

